* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Cellular Respiration - Parkway C-2
Fatty acid synthesis wikipedia , lookup
Mitochondrion wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Blood sugar level wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Butyric acid wikipedia , lookup
Photosynthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Electron transport chain wikipedia , lookup
Phosphorylation wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Microbial metabolism wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Biochemistry wikipedia , lookup
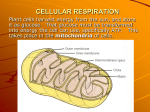
Cellular Respiration What, if anything, is the difference between respiration and breathing? Do fish breathe? Do plants respire? What about anaerobic bacteria like those we find buried in swamps? Energy comes in a lot of different forms: light, heat, electricity, etc. Living things use chemical compounds that are converted within cells to store and release energy. Life requires energy to survive and organisms can either make it themselves (autotrophs) or acquire it from someone else (heterotrophs). The majority of autotrophs are photosythesizers and use energy from the sun to make sugars. Whether you are a photoautotroph or heterotroph, all life must release the energy contained within sugars and other compounds for survival. This process is called respiration. http://www.globalchange.umich.edu/globalchange1/current/lectures/kling/energyflow/energyflow.html https://eapbiofield.wikispaces.com/file/view/CellResp1.gif Fig. 9-2 Light energy ECOSYSTEM Photosynthesis in chloroplasts CO2 + H2O Organic +O molecules 2 Cellular respiration in mitochondria ATP ATP powers most cellular work Heat energy Glucose This chemical is called adenosine triphosphate or ATP. ATP is made up of adenosine – a nitrogenous base, ribose – a five-carbon sugar, and three phosphate groups. The negatively charged phosphate groups are the source of energy within the ATP molecule. When energy is available within a cell, a third phosphate group is added to adenosine diphosphate (ADP) making ATP. Phosphate groups are negatively charged and “do not like being” bonded to each other. When the bonds between phosphates are broken, energy is released. Fig. 8-8 Adenine Phosphate groups Ribose Fig. 8-9 P P P Adenosine triphosphate (ATP) H2O Pi + Inorganic phosphate P P + Adenosine diphosphate (ADP) Energy Fig. 8-12 ATP + H2O Energy from catabolism (exergonic, energy-releasing processes) ADP + P i Energy for cellular work (endergonic, energy-consuming processes) That energy is enough to power many cellular activities like active transport across cell membranes, protein synthesis, and muscle contraction; however, cells are not chock full of ATP molecules, instead they only contain about enough to last a few seconds of activity. A single glucose molecule stores about 90 times more energy than a molecule of ATP. Cells break down glucose effectively “recharging” ATP from ADP. For this process to be most efficient, oxygen is required. We get oxygen from breathing. Fig. 7-UN2 Active transport: ATP Fig. 17-18-4 Amino end of polypeptide E 3′′ mRNA Ribosome ready for next aminoacyl tRNA P A site site 5′′ GTP GDP E E P A P A GDP GTP E P A Fig. 50-27-4 Thick filament Thin filaments Thin filament Myosin head (lowenergy configuration ATP ATP Thick filament Thin filament moves toward center of sarcomere. Actin ADP Myosin head (lowenergy configuration ADP + Pi Pi ADP Pi Cross-bridge Myosin binding sites Myosin head (highenergy configuration http://www.qcc.cuny.edu/BiologicalSciences/Faculty/DMeyer/respiration.html Question of the Day! 1. Can the flame move? 2. Can the flame grow? 3. Can it reproduce? 4. Does the flame respire? 5. Is the flame alive? Think About This !! Do plants respire? Design an experiment that will test your answer? Hypothesis: Plants will give off CO2. Methods: Isolate a plant. Put in the dark. Measure CO2. Note: Made sure plants had carbohydrates (ie sugar) CO2 in water creates carbonic acid (H2CO3). This is the same thing as carbonated water or seltzer. Listen to This !! http://weblogs.ccsd.k12.co.us/cth/brios/?m=20070509 Write !! Any word or concept that you are unaware of or confused about. ANSWER THESE !! 1. How is fermentation different from respiration? 2. Put the three parts of aerobic respirations in order. 3. How many ATP are made during the entire process? 4. How are NADH and NAD+ involved in aerobic respiration? 5. Some organisms, like yeast, can survive in oxygen-rich and oxygen-depleted environments. In which situation would you expect yeast to grow the fastest? Explain. Fig. 9-UN1 becomes oxidized (loses electron) becomes reduced (gains electron) Fig. 9-UN2 becomes oxidized becomes reduced Fig. 9-3 Reactants Products becomes oxidized becomes reduced Methane (reducing agent) Oxygen (oxidizing agent) Carbon dioxide Water Fig. 9-UN3 becomes oxidized becomes reduced Glucose Glycolysis 2 ADP 2 ATP 4 ATP 2 NAD+ Glucose 2 Glyceraldehyde-3phosphate 4 ADP 2 NADH 2 Pyruvic Acid Krebs cycle 1 NADH Enzyme 1 NADH 1 NADH 1 ATP 1 NADH 1 FADH2 Citric Acid Pyruvic Acid Answer These, Too !! 1. What can you say about the relationship between the reactants and products the photosynthesis and respiration equations? 2. How many molecules of carbon dioxide and how many molecules of water are needed for green plants to synthesize one molecule of glucose and six molecules of oxygen? 3. What does respiration produce and what does it use? 4. Why is the Krebs cycle also called the Citric acid cycle? 5. How much of the ATP produced during aerobic respiration comes from the Krebs cycle and how much comes from glycolysis? Fill This Out !! Question of the Day! Make a flow chart using as many of the following terms as possible: Sun Cytoplasm lactic acid Glucose Yeast Mitocondria Glycolysis CO2 1st order consumers Oxygen ethyl alcohol Water Autotrophs Krebs cycle Photosynthesis Some prokaryotes pyruvic acid FAD/FADH2 2nd order consumers NAD+/NADH ATP Student study School Mr Schmalz Good grade Good College Happy Test No study Bad Grade No College Sad Cellular respiration is the process that releases energy by breaking down glucose and other food molecules. When oxygen is present, it’s aerobic; when oxygen is absent, it’s anaerobic. There are four pathways in cellular respiration (not all function at the same place or at the same time): glycolysis, fermentation (2 types – alcoholic and lactic acid), Krebs cycle, and electron transport chain. Respiration is a process of breaking down glucose in such a way that energy capture is maximized. When sugars are burned, all the energy is released at one time; however, if cells truly “burned” sugars, much of the energy contained within each glucose molecule would be lost as heat and light and cells might even catch fire. Fig. 9-6-1 Electrons carried via NADH Glycolysis Pyruvate Glucose Cytosol ATP Substrate-level phosphorylation Fig. 9-6-2 Electrons carried via NADH and FADH2 Electrons carried via NADH Citric acid cycle Glycolysis Pyruvate Glucose Mitochondrion Cytosol ATP ATP Substrate-level phosphorylation Substrate-level phosphorylation Fig. 9-6-3 Electrons carried via NADH and FADH2 Electrons carried via NADH Citric acid cycle Glycolysis Pyruvate Glucose Oxidative phosphorylation: electron transport and chemiosmosis Mitochondrion Cytosol ATP ATP ATP Substrate-level phosphorylation Substrate-level phosphorylation Oxidative phosphorylation http://www.qcc.cuny.edu/BiologicalSciences/Faculty/DMeyer/respiration.html Glycolysis is the process in which one molecule of glucose is broken into 2 glyceraldehyde 3-phosphates (a 3-carbon compound) “investing” 2 ATP. These are converted into two molecules of pyruvic acid and the ‘investment’ pays out a total of 4 ATP and 2 NADH. The process of glycolysis produces and then captures two pairs of high-energy electrons using the carrier NAD+. NADH carries some of glucose’s energy to a place where it can be converted to ATP. Glycolysis is fast and does not require oxygen; however, when it occurs anaerobically, it hits a roadblock when all the NAD+ get tied up carrying electrons. Fig. 9-8 Energy investment phase Glucose 2 ADP + 2 P 2 ATP used 4 ATP formed Energy payoff phase 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 2 NADH + 2 H+ 2 Pyruvate + 2 H2O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H2O 2 ATP 2 NADH + 2 H+ Fig. 9-UN5 Outputs Inputs 2 ATP Glycolysis + 2 NADH Glucose 2 Pyruvate http://www.science.smith.edu/departments/B iology/Bio231/ When oxygen is absent, glycolysis is followed by fermentation. The two main types of fermentation are alcoholic fermentation and lactic acid fermentation. Both types of fermentation function by passing the high energy electrons back to pyruvic acid, freeing up NAD+ again so glycolysis can continue. In the absence of oxygen, yeast and a few other microorganisms use alcoholic fermentation, forming ethyl alcohol and carbon dioxide as wastes. Animals cannot perform alcoholic fermentation, but some cells, such as human muscle cells, can convert glucose into lactic acid. This is called lactic acid fermentation. Fig. 9-19 Glucose CYTOSOL Glycolysis Pyruvate No O2 present: Fermentation O2 present: Aerobic cellular respiration MITOCHONDRION Ethanol or lactate Acetyl CoA Citric acid cycle Fig. 9-18 2 ADP + 2 Pi Glucose 2 ATP Glycolysis 2 Pyruvate 2 NAD+ 2 NADH + 2 H+ 2 CO2 2 Acetaldehyde 2 Ethanol (a) Alcohol fermentation 2 ADP + 2 Pi Glucose 2 ATP Glycolysis 2 NAD+ 2 NADH + 2 H+ 2 Pyruvate 2 Lactate (b) Lactic acid fermentation Fig. 9-18a 2 ADP + 2 P i Glucose 2 ATP Glycolysis 2 Pyruvate 2 NAD+ 2 Ethanol (a) Alcohol fermentation 2 NADH + 2 H+ 2 CO2 2 Acetaldehyde Fig. 9-18b 2 ADP + 2 P i Glucose 2 ATP Glycolysis 2 NAD+ 2 NADH + 2 H+ 2 Pyruvate 2 Lactate (b) Lactic acid fermentation Sun Autotroph 1st Order consumer 2nd Order Alcohol fermentation Cytoplasm H2O Photosynthesis Yeast Glucose O2 Glycolysis Some CO2 Prokaryotes Pyruvic acid Lactic acid fermentation Krebs cycle NAD+/NADH ATP FADH2 Mitocondria Electron transport chain Fig. 9-10 CYTOSOL MITOCHONDRION NAD+ NADH + H+ 2 1 Pyruvate Transport protein 3 CO2 Coenzyme A Acetyl CoA Fig. 9-11 Pyruvate CO2 NAD+ CoA NADH + H+ Acetyl CoA CoA CoA Citric acid cycle 2 CO2 3 NAD+ FADH2 3 NADH FAD + 3 H+ ADP + P i ATP Fig. 9-UN6 Inputs Outputs S—CoA C 2 ATP O CH3 2 Acetyl CoA 6 NADH O C COO CH2 COO 2 Oxaloacetate Citric acid cycle 2 FADH2