* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download acetyl CoA
Lactate dehydrogenase wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Mitochondrion wikipedia , lookup
Metalloprotein wikipedia , lookup
Specialized pro-resolving mediators wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Phosphorylation wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Butyric acid wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Electron transport chain wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
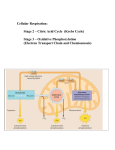
Autotroph Heterotroph (Producers) (Consumers) Make their own food • Photosynthesis • Chemosynthesis - Plants - Some bacteria & protists Energy obtained by eating Cellular Respiration Oxygen + Glucose Carbon Dioxide + Water + ATP (6O2 + C6H12O6 6CO2 + 6H2O + ATP) • Cellular respiration is an aerobic process because it requires oxygen (aerobic respiration) • There are three steps in cellular respiration – Glycolysis – Krebs Cycle (a.k.a. “Citric Acid Cycle”) – Electron Transport Chain (ETC) • In the absence of oxygen, glycolysis is followed by fermentation. This is called anaerobic respiration. Breathing v. Cellular Respiration Electrons “falling” to oxygen provides cellular energy LEO the lion says GER •Lose electrons – oxidation •Gain electrons - reduction Structure of the Mitochondria Overview of Cellular Respiration Mitochondrion Cytoplasm Glycolysis • a single molecule of glucose is enzymatically cut in half through a series of steps, • two molecules of pyruvate are produced, • two molecules of NAD+ are reduced to two molecules of NADH • a net of two molecules of ATP is produced. 2 ATP 2 ADP 4 ADP 4 ATP Substrate-Level Phosphorylation Enzyme P Enzyme ADP ATP P P During glycolysis, the transfer of a phosphate group from a substrate to ADP, produces ATP Formation of Acetyl CoA • Pyruvate does not enter the citric acid cycle, but undergoes some chemical grooming in which – a carboxyl group is removed and given off as CO2, – the two-carbon compound remaining is oxidized while a molecule of NAD+ is reduced to NADH, – coenzyme A joins with the two-carbon group to form acetyl coenzyme A, abbreviated as acetyl CoA, and – acetyl CoA enters the citric acid cycle. NAD NADH H 2 CoA Pyruvate Acetyl coenzyme A 1 CO2 3 Coenzyme A The Citric Acid Cycle • Also called the Krebs cycle (after the German-British researcher Hans Krebs, who worked out much of this pathway in the 1930s) • completes the oxidation of organic molecules • generates many NADH and FADH2 molecules. • the two-carbon group of acetyl CoA is added to a four-carbon compound, forming citrate, • citrate is degraded back to the four-carbon compound, • two CO2 are released, and 1 ATP, 3 NADH, and 1 FADH2 are produced. Kreb’s Cycle Citric Acid Cycle Totals • Remember that the citric acid cycle processes two molecules of acetyl CoA for each initial glucose. • Thus, after two turns of the citric acid cycle, the overall yield per glucose molecule is – 2 ATP, – 6 NADH, and – 2 FADH2. Oxidative Phosphorylation (The E.T.C.) • Involves electron transport and chemiosmosis • Requires an adequate supply of oxygen. • Electrons from NADH and FADH2 travel down the electron transport chain to O2. • Oxygen picks up H+ to form water. • Energy released by these redox reactions is used to pump H+ from the mitochondrial matrix into the intermembrane space. • In chemiosmosis, the H+ diffuses back across the inner membrane through ATP synthase complexes, driving the synthesis of ATP. Electron Transport Chain Oxidative phosphorylation: energy to create ATP is provided by the oxidation of glucose (pyruvic acid & NADH/FADH2) Electron Transport Inhibition • Numerous poisons are deadly because they interfere with cellular respiration, particularly the electron transport chain • Some examples include: • Rotenone (pesticide): prevents electrons from going beyond the first carrier • Cyanide & Carbon Monoxide: prevents passage of electrons past 4th protein complex • DNP: uncoupler (makes mitochondrial membrane leaky to H+ ions) that results in enormous increase in metabolism and therefore increase in body temperature that can be extreme enough to be fatal • Oligomycin (antibiotic): blocks flow of H+ through ATP synthase • Note that toxins can be useful as pesticides, antibiotics and for biochemical research. Energy Totals • Glycolysis produces just 2 net ATP molecules per molecule of glucose. • Krebs Cycle & ETC produce up to 36 additional ATP. • The complete breakdown of glucose through cellular respiration, including glycolysis, results in the production of up to 38 molecules of ATP (net). Fermentation 1. Alcoholic Fermentation: - Performed by yeasts and a few other microorganisms pyruvic acid + NADH → alcohol + CO2 + NAD+ 2. Lactic Acid Fermentation: - in cells, such as muscle cells, the pyruvic acid from glycolysis is converted to lactic acid - pyruvic acid + NADH → lactic acid + NAD+ **Fermentation regenerates NAD+ so that glycolysis can continue Lactic Acid & Alcoholic Fermentation Uses of Fermentation • Lactate is carried by the blood to the liver, where it is converted back to pyruvate and oxidized in the mitochondria of liver cells. • The dairy industry uses lactic acid fermentation by bacteria to make cheese and yogurt. • Other types of microbial fermentation turn – soybeans into soy sauce and – cabbage into sauerkraut. • The baking and winemaking industries have used alcohol fermentation for thousands of years. • In this process yeasts (single-celled fungi) – oxidize NADH back to NAD+ and – convert pyruvate to CO2 and ethanol. Food Molecules and Biosynthesis Carbohydrates Fats Glycerol Fatty Acids Sugars Glucose Proteins G3P Glycolysis Pyruvate Pyruvate Oxidation Acetyl CoA ATP Amino Acids Citric Acid Cycle Oxidative Phosphorylation Food Molecules and Biosynthesis ATP Citric Acid Cycle ATP needed to drive biosynthesis Glucose Synthesis Pyruvate Oxidation Pyruvate G3P Glucose Acetyl CoA Amino groups Amino acids Fatty acids Glycerol Proteins Cells, tissues, organisms Fats Sugars Carbohydrates