* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download APOPTOSIS
Synaptic gating wikipedia , lookup
Metastability in the brain wikipedia , lookup
Multielectrode array wikipedia , lookup
Haemodynamic response wikipedia , lookup
Signal transduction wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Electrophysiology wikipedia , lookup
Subventricular zone wikipedia , lookup
Nervous system network models wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Development of the nervous system wikipedia , lookup
Optogenetics wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Biochemistry of Alzheimer's disease wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Neuroanatomy wikipedia , lookup
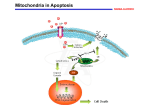
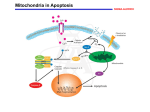
Today Feb 27… Apoptosis Aging of the Nervous System APOPTOSIS What is it? Why is it important? How is it controlled? What is its role in age-related disease? APOPTOSIS Programmed cell death Orderly cellular self destruction Process: as crucial for survival of multi-cellular organisms as cell division MULTIPLE FORMS??? Forms of cell death Necrosis Passive Pathological Swelling, lysis "Classic" Apoptosis Active Physiological or pathological Condensation, cross-linking Mitotic catastrophe Passive Pathological Swelling, lysis Dissipates Phagocytosed Dissipates Inflammation No inflammation Inflammation Externally induced Internally or externally induced Internally induced STAGES OF CLASSIC APOPTOSIS Healthy cell DEATH SIGNAL (extrinsic or intrinsic) Commitment to die (reversible) EXECUTION (irreversible) Dead cell (condensed, crosslinked) ENGULFMENT (macrophages, neighboring cells) DEGRADATION APOPTOSIS: important in embryogenesis Morphogenesis (eliminates excess cells): Selection (eliminates non-functional cells): APOPTOSIS: important in embryogenesis Immunity (eliminates dangerous cells): Self antigen recognizing cell Organ size (eliminates excess cells): APOPTOSIS: important in adults Tissue remodeling (eliminates cells no longer needed): Apoptosis Virgin mammary gland Late pregnancy, lactation - Testosterone Apoptosis Prostate gland Involution (non-pregnant, non-lactating) APOPTOSIS: important in adults Tissue remodeling (eliminates cells no longer needed): Apoptosis Resting lymphocytes + antigen (e.g. infection) - antigen (e.g. recovery) Steroid immunosuppressants: kill lymphocytes by apoptosis Lymphocytes poised to die by apoptosis APOPTOSIS: important in adults Maintains organ size and function: Apoptosis X + cell division Cells lost by apoptosis are replaced by cell division (remember limited replicative potential of normal cells restricts how many times this can occur before tissue renewal declines) APOPTOSIS: control Receptor pathway (physiological): FAS ligand Death receptors: (FAS, TNF-R, etc) TNF Death domains Adaptor proteins Pro-caspase 8 (inactive) Pro-execution caspase (inactive) MITOCHONDRIA Caspase 8 (active) Execution caspase (active) Death APOPTOSIS: control Intrinsic pathway (damage): Mitochondria BAX BAK BOK BCL-Xs BAD BID B IK BIM NIP3 BNIP3 Cytochrome c release Pro-caspase 9 cleavage Pro-execution caspase (3) cleavage BCL-2 BCL-XL BCL-W MCL1 BFL1 DIVA NR-13 Several viral proteins Caspase (3) cleavage of cellular proteins, nuclease activation, etc. Death APOPTOSIS: control Physiological receptor pathway Intrinsic damage pathway MITOCHONDRIAL SIGNALS Caspase cleavage cascade Orderly cleavage of proteins and DNA CROSSLINKING OF CELL CORPSES; ENGULFMENT (no inflammation) APOPTOSIS: Role in Disease TOO MUCH: Tissue atrophy Neurodegeneration Thin skin etc TOO LITTLE: Hyperplasia Cancer Athersclerosis etc APOPTOSIS: Role in Disease Neurodegeneration Neurons are post-mitotic (cannot replace themselves; neuronal stem cell replacement is inefficient) Neuronal death caused by loss of proper connections, loss of proper growth factors (e.g. NGF), and/or damage (especially oxidative damage) Neuronal dysfunction or damage results in loss of synapses or loss of cell bodies (synaptosis, can be reversible; apopsosis, irreversible) PARKINSON'S DISEASE ALZHEIMER'S DISEASE HUNTINGTON'S DISEASE etc. APOPTOSIS: Role in Disease Cancer Apoptosis eliminates damaged cells (damage => mutations => cancer Tumor suppressor p53 controls senescence and apoptosis responses to damage Most cancer cells are defective in apoptotic response (damaged, mutant cells survive) High levels of anti-apoptotic proteins or Low levels of pro-apoptotic proteins ===> CANCER APOPTOSIS: Role in Disease AGING Aging --> both too much and too little apoptosis (evidence for both) Too much (accumulated oxidative damage?) ---> tissue degeneration Too little (defective sensors, signals? ---> dysfunctional cells accumulate hyperplasia (precancerous lesions) OPTIMAL FUNCTION (HEALTH) APOPTOSIS AGING APOPTOSIS Neurodegeneration, cancer, ….. Questions Why is apoptosis important embryologically? Why is apoptosis important in adults? What can too much apoptosis cause? Too little apoptosis? What is the difference between apoptosis and necrosis? Aging of the Nervous System: Structural Changes Brain Plasticity and CNS Regenerative Potential (A review of 2 previous lectures given) From the beginning of the 20th Century until the 1990s, it was stated that neurons DID NOT proliferate. The fact that they COULD NOT proliferate did not exclude the possibility of proliferation under “specific conditions.” In fact, the CNS has a considerable regenerative potential depending on the special conditions of the neuronal environment. Neurons that may proliferate into adulthood include: Progenitor “precursor” neurons lining the cerebral ventricules Neurons in the hippocampus Neurons usually “dormant” with potential for neuron and glia proliferation Neuroglia (astrocytes, oligodentrocytes) and microglia (immune cells) with the ability to perpetually self renew and produce the three types of neural cells Regenerative potential depends on changes in whole body and neural microenvironment Whole body changes: Physical exercise Appropriate nutrition Good circulation Education Stress others • Neural microenvironment changes: –Brain metabolism (oxygen consumption, free radicals, circulatory changes) –Hormonal changes (estrogens, growth factors, others) –others Major Function of the Nervous System The major function of the CNS is to communicate & to connect: with other CNS cells with peripheral tissues (outside CNS) with the external environment (including physical and social environments) This communication regulates: Mobility Sensory information Cognition Affect and mood Functions of whole-body systems Aging of the Nervous System Structural Changes 1. Changes in Brain Weight 2. Denudation 3. Loss of Neurons 4. Neuropatholog ical Markers Biochemical Changes (will be in a lecture YET TO COME) 1. Neurotransmitt ers 2. CNS Synapses 3. Neurotransmitt er Imbalance and Brain Disorders Fig. 7-2: Changes in brain weight with aging in human males and females Fig. 7-4: “Denudation” of the neurons. Changes in pyramidal neurons of the aging human cerebral cortex In normal aging, the loss of neurons is moderate & occurs in specific brain areas: Locus ceruleus (catecholaminergic neurons) Substantia nigra (dopaminergic neurons) Nucleus basalis of Meynert (cholinergic neurons) Hippocampus (cholinergic neurons) Neuropathologies Lipofuscin By-product of cellular autophagia Linear increase with normal aging Function in disease unkown Lewy Bodies Present in normal aging (60+) Increased accumulation in Parkinson’s Disease Neurofibrillary Tangles Tangled masses of fibrous elements Present in normal aging in hippocampus Accumulation in cortex is sign of Alzheimer’s Young Fig. 7-6: Neuron from 605 day old rat Old Fig. 7-5: Free radical accumulation (lipofuscin) in young rats and old rats Fig. 7-7: Magnification of lipofuscin granules of Fig. 7-6 Fig. 7-8: Lewy Bodies. Aggregation of filaments, vesicular profiles and poorly resolved granular material C A & B: Fibrillary tangles Alterations of tau protein and microtubule assembly? Paired Helical Filaments (PHF) C: Neuritic plaque Accumulation of amyloid broken down PHFs Pathological and Cellular Changes with Normal Aging Increased intracellular deposits of lipofuscin Intracellular formation of PHFs Accumulation of amyloid deposits in the neuritic plaques and surrounding the cerebral blood vessels Accumulation of Lewy bodies Cell death (apoptosis, necrosis) Questions What are the pathological/cellular changes that occur with normal aging? What are the normal structural changes with aging? What are some examples of the areas of the brain where there is loss of neurons? What conditions favor neuroregeneration?