* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 3.4 Diffusion and Osmosis - Greenall
Survey
Document related concepts
Cell nucleus wikipedia , lookup
Cytoplasmic streaming wikipedia , lookup
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cell growth wikipedia , lookup
Extracellular matrix wikipedia , lookup
Cytokinesis wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Signal transduction wikipedia , lookup
Cell membrane wikipedia , lookup
Transcript
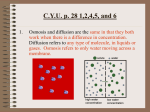
3.4 Diffusion and Osmosis Cells are constantly taking in and sending out substances. If they had to use energy to move every molecule, it would require a massive amount of energy. Some molecules do not have to use energy to move. 3.4 Diffusion and Osmosis Passive transport does not require energy input from a cell. • Molecules can move across the cell membrane through passive transport. • There are two types of passive transport. • diffusion • osmosis 3.4 Diffusion and Osmosis Diffusion is the movement of molecules from a place of higher concentration to a place of lower concentration. It is a natural motion of particles. • Molecules diffuse down a concentration gradient, meaning they move from a higher concentration area, to a lower concentration area. High concentration Low concentration 3.4 Diffusion and Osmosis Diffusion and osmosis are types of passive transport. • The diffusion of water molecules is called osmosis. The process of osmosis is exactly the same as diffusion but refers only to water molecules. Water molecules diffuse across a semipermeable membrane from a place of higher water concentration to a place of lower water concentration. 3.4 Diffusion and Osmosis A Dynamic equilibrium is reached when the concentration is the same throughout the solution. 3.4 Diffusion and Osmosis In cells, small lipids, and other nonpolar molecules, such as carbon dioxide and oxygen, easily diffuse across the membrane 3.4 Diffusion and Osmosis Water is the solvent in our cells, the other molecules are the solute. There are three types of solutions. • isotonic • hypertonic • hypotonic 3.4 Diffusion and Osmosis • A solution is isotonic if it has the same concentration of solutes that the cell has. Equal amount of water enter and exit a cell so size remains constant. • A solution is hypertonic if it has a higher concentration of solutes than a cell. More water diffuses out of the cell, and the cell shrivels. • A solution is hypotonic if it has a lower concentration of solutes than a cell. More water diffuses into the cell, and the cell grows larger. 3.4 Diffusion and Osmosis Some molecules can not diffuse through cells on their own, they need the help of transport proteins to diffuse. • Some molecules cannot easily diffuse across the cell membrane. • Facilitated diffusion is diffusion through transport proteins. • Some transport proteins are simple channels, or tunnels, through with particles such as ions can pass. Others act more like enzymes. When bound, the protein changes shape, allowing the molecule to travel the rest of the way into the cell.