* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Pyrosequencing Technology
Exome sequencing wikipedia , lookup
Transcriptional regulation wikipedia , lookup
Whole genome sequencing wikipedia , lookup
Comparative genomic hybridization wikipedia , lookup
Agarose gel electrophoresis wikipedia , lookup
Eukaryotic transcription wikipedia , lookup
Maurice Wilkins wikipedia , lookup
Molecular evolution wikipedia , lookup
Non-coding DNA wikipedia , lookup
Citric acid cycle wikipedia , lookup
Gel electrophoresis of nucleic acids wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Molecular cloning wikipedia , lookup
DNA sequencing wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Community fingerprinting wikipedia , lookup
DNA supercoil wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Biosynthesis wikipedia , lookup
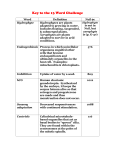
The Principle of PyrosequencingTM Technology TM Pyrosequencing technology is a simple to use technique for accurate and consistent analysis of large numbers of short to medium length DNA sequences. Step 1 A sequencing primer is hybridized to a single stranded, PCR amplified, DNA template, and incubated with the enzymes, DNA polymerase, ATP sulfurylase, luciferase and apyrase, and the substrates, adenosine 5´ phosphosulfate (APS) and luciferin. Step 2 The first of four deoxyribonucleotide triphosphates (dNTP) is added to the reaction. DNA polymerase catalyzes the incorporation of the deoxyribonucleotide triphosphate into the DNA strand, if it is complementary to the base in the template strand. Each incorporation event is accompanied by release of pyrophosphate (PPi) in a quantity equimolar to the amount of incorporated nucleotide. Step 2 Polymerase (DNA)n + dNTP (DNA)n+1 + PPi Step 3 Sulfurylase light APS+PPi ATP luciferin oxyluciferin Luciferase Step 3 ATP sulfurylase quantitatively converts PPi to ATP in the presence of adenosine 5´ phosphosulfate (APS). This ATP drives the luciferase mediated conversion of luciferin to oxyluciferin that generates visible light in amounts that are proportional to the amount of ATP. The light produced in the luciferase-catalyzed reaction is detected by a charge coupled device (CCD) camera and seen as a peak in a Pyrogram™. The height of each peak (light signal) is proportional to the number of nucleotides incorporated. ATP Light time nucleotide incorporation generates light seen as a peak in the Pyrogram Step 4 dNTP Apyrase, a nucleotide degrading enzyme, continuously degrades ATP and unincorporated dNTPs. This switches off the light and regenerates the reaction solution. The next dNTP is then added. dNDP + dNMP + phosphate Apyrase ATP Step 4 Apyrase ADP + AMP + phosphate Step 5 nucleotide sequence G C G C – A GG CC T T A G C T Step 5 Addition of dNTPs is performed one at a time. It should be noted that deoxyadenosine alfa-thio triphosphate (dATPaS) is used as a substitute for the natural deoxyadenosine triphosphate (dATP) since it is efficiently used by the DNA polymerase, but not recognized by the luciferase. As the process continues, the complementary DNA strand is built up and the nucleotide sequence is determined from the signal peaks in the Pyrogram. Technical Note 101 nucleotide added Optimization of Pyrosequencing technology The Pyrosequencing reaction is simple and robust, using an enzyme cascade system that gives unambiguous and easily quantifiable results. Each component and parameter has been optimized to achieve highly consistent results, with accuracy levels of >99% for SNP genotyping, when used routinely in automated systems from Pyrosequencing AB. • Substrate concentrations are optimized. • Apyrase of a specific grade has been selected to ensure that all dNTPs are degraded, including the alfa-thio-dATP which is used instead of dATP. This enzyme also hydrolyzes ATP. • The rate of dNTP degradation by apyrase is slower than the rate of dNTP incorporation by the polymerase, favouring sufficient incorporation of dNTPs. • The rate of ATP synthesis by the sulfurylase is faster than the rate of ATP hydrolysis by apyrase so that ATP concentration and light production are in proportion to the number of dNTPs incorporated. • Reagents are carefully controlled to ensure adequate activity and quality. For further information and the latest applications please visit www.pyrosequencing.com Scientific references Ronaghi, M. et al., (1998) A sequencing method based on real-time pyropohosphate, a review. Science 281, 363-365 Nyrén et al., (1997) Detection of single-base changes using a bioluminometric primer extension assay. Anal Biochem 244, 36-373 Ronaghi, M. et al., (1996) Real-time DNA sequencing using detection of pyrophosphate release. Anal Biochem 242, 84-89 Nyrén, P., (1994) Apyrase immobilized on paramagnetic beads used to improve detection limits in bioluminometric ATP monitoring. J Biolumin Chemilumin 9, 29-34 Nyrén P. et al., (1993) Solid phase DNA minisequencing by enzymatic luminometric inorganic pyrophosphate detection assay. Anal Biochem 208, 171-175 Pyrosequencing, PSQ, Pyrogram and The Solid Phase Sequencing method is covered by patents owned by Pyrosequencing AB and AB Sangtec Medical. The PCR process is covered by several patents owned by Roche Molecular Systems and F. Hoffman-La Roche Ltd. © Copyright 2002 Pyrosequencing AB. are trademarks owned by Pyrosequencing AB. Pyrosequencing technology is covered by patents and patent applications owned by Pyrosequencing AB. HEAD OFFICE: Pyrosequencing AB Vallongatan 1, SE-752 28 Uppsala, Sweden Tel: +46 (0)18 56 59 00 Fax: +46 (0)18 59 19 22 [email protected], www.pyrosequencing.com Pyrosequencing Inc. USA Massachusetts Pyrosequencing Inc. USA California Pyrosequencing Sarl. France Pyrosequencing GmbH. Germany Pyrosequencing AB. Nordic region Pyrosequencing Ltd. UK & Eire (877) 797-6767 (Toll Free) (877) 797-6767 (Toll Free) +33 (0)1 55 94 86 32 +49 (0)40 8195 7566 +46 (0)18 489 70 00 +44 (0)20 8334 8454 Printed in Sweden by Wikströms, Uppsala. For research use only. Not intended for diagnostic purposes. 70-0001-6102 Aug 2002 Nyrén P., (1987) Enzymatic method for continuous monitoring of DNA polymerase activity. Anal Biochem 167, 235-238