* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download File - Mrs. LeCompte
Lactate dehydrogenase wikipedia , lookup
Magnesium in biology wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Butyric acid wikipedia , lookup
Biosynthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Mitochondrion wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Phosphorylation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Electron transport chain wikipedia , lookup
Biochemistry wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
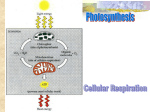
8-2: Glycolysis Cellular Respiration: An Overview 3 Stages: 1) Glycolysis Occurs in the cytoplasm Splits glucose (6 C) into two pyruvate (3 C) molecules 2) Krebs Cycle Occurs in the mitochondrial matrix Breaks down a pyruvate derivative (Acetyl CoA) into CO2 Both glycolysis and the Krebs Cycle produce: o A small amount of ATP o NADH by reducing NAD+ o Use SUBSTRATE-LEVEL PHOSPHORYLATION = ATP production by the direct enzymatic transfer of phosphate from an intermediate substrate in catabolism to ADP. 3) Electron Transport Chain Occurs along the inner membrane of the mitochondria Accepts energized electrons (e-s) from reduced coenzymes (NADH and FADH2) Couples the exergonic slide of e-s to ATP synthesis or… OXIDATIVE PHOSPHORYLATION = ATP production that is coupled to the exergonic transfer of e-s from food to oxygen _______________________________________________________________________ GLYCOLYSIS = catabolic pathway during which 6-C glucose is split into two 3-C sugars, called pyruvate Occurs in the cytoplasm No CO2 is released Occurs whether or not oxygen is present Energy-Investment Phase: 1) 2 ATP are used to activate glucose by phosphorylating it This is endergonic 2) Glucose is split into two 3-C molecules known as Glyceraldehyde-3-Phosphate (G3P). After this, each G3P goes through the same series of steps Energy-Harvesting Phase: 3) G3P is oxidized e-s are removed, along with two H+ ions These are passed to NAD+ to form NADH as follows: 2 NAD+ + 4 e- + 2 H+ 2 NADH 2 NADH’s are made total, one from each G3P This is very exergonic G3P is phosphorylated by inorganic phosphate, increasing the energy of the molecule again 4) ATP is produced by substrate-level phosphorylation Highly exergonic 5) Molecule is rearranged 6) ATP is produced again by substrate-level phosphorylation Highly exergonic In Summary: Glucose + 2 ATP + 2 NAD+ 2 pyruvic acid + 4 ATP + 2 NADH + 2 H+ Net Gain: 2 ATP + 2 NADH FERMENTATION Cellular respiration is an aerobic process, but some organisms can oxidize their food under anaerobic conditions = existing in the absence of free oxygen Fermentation = anaerobic catabolism of organic nutrients Begins with Glycolysis Still gets a net gain of 2 ATP Instead of being further oxidized (as it is in respiration), the NADH hands the two electrons back to the organic molecule they came from, reducing it again. Two Main Types: 1) Alcohol Fermentation Final product is ethanol Releases CO2 Many yeasts and bacteria carry out alcoholic fermentation under anaerobic conditions 2) Lactic Acid Fermentation Final product is lactate (lactic acid) When O2 is scarce (a.k.a. oxygen debt), vertebrate muscle cells switch from aerobic respiration to lactic acid fermentation Lactate accumulates and the lower pH it provides: o Causes pain o Slows muscle contractions (a.k.a. muscle fatigue), allowing O2 to build back up again “2nd wind” The lactate is gradually carried to the liver where it is converted back to pyruvate. Advantages: Makes FOOD!! Like cheese, yogurt, sauerkraut, soy sauce, etc. Makes Chemicals: isopropanol, acetic acid, etc. Alcoholic beverages: wine, beer, etc. Disadvantage: Less ATP per glucose The 2 ATP net gain in fermentation (from glycolysis) = 14.6 kcal. Complete breakdown of glucose to CO2 and H2O = 686 kcal. Efficiency rate of fermentation = 14.6 / 686 (x100%) = 2.1% Fermentation will only produce 2 ATP per glucose, but aerobic respiration can produce 36-38 ATP per glucose! 8-3 Notes: Cellular Respiration Recall: Glycolysis involves glucose being broken down into 2 pyruvate molecules. The remainder of Cellular Respiration occurs in the mitochondrion. Review its structure: The Preparatory (Prep) Reaction The 2 pyruvates cannot directly enter the next phase (nor the mitochondrion), but must be modified first. The molecule is oxidized by transferring two e-s to NAD+ NADH One carbon is removed, forming CO2 Coenzyme A is added, making the final product acetyl CoA This reaction happens twice for each molecule of glucose first entered. Final Tally: 2 pyruvate 2 acetyl CoA + 2 CO2 + 2 NADH The Citric Acid (Krebs) Cycle Occurs in the mitochondrial matrix Requires 2 turns for one glucose molecule Many steps are involved, but here are the ones you need to know: 1) Acetyl CoA (2 C) enters and combines with oxaloacetate (C4) to form Citric Acid (C6) 2) The compound is oxidized a total of 4 times, losing two e-s each time o Forms a total of 3 NADH and 1 FADH2 3) The compound is phosphorylated by an inorganic phosphate, which it then transfers by substrate-level phosphorylation to ATP 4) During the cycle, the compound also loses a total of 2 C’s as CO2 o This completes the breakdown of the original 6-C glucose molecule Net Gain from Citric Acid Cycle (per glucose or 2 turns of the cycle) 2 ATP 6 NADH 2 FADH2 THE ELECTRON TRANSPORT CHAIN Series of electron carrier (protein) molecules embedded in the inner mitochondrial membrane (cristae) of eukaryotes (or the plasma membrane invaginations of prokaryotes) NADH and FADH2 pass their high energy e-s to the E.T.C., becoming oxidized again o They can now be reused Each carrier in the chain is successively more electronegative (pulls on electrons harder) than the one before it, so electrons passed from one to the next in series Most carriers in the chain are forms of cytochrome 1st carrier = FMN (Flavin mononucleotide) becomes reduced when it accepts the electrons, but immediately becomes oxidized when the next carrier takes the electrons from it. This pattern continues the length of the chain until the final electron acceptor – O2 – is reduced. o As molecular oxygen is reduced, it also picks up H+ ions from the matrix to form H2O o For every 2 NADHs, 1 O2 is reduced to 2 H2O o O2 is so important, that if it is not present, the chain does not function and NO ATP is produced by the mitochondria. At any given time, the human body only contains enough ATP to sustain life for ~1 minute! Cellular respiration must be done constantly to prevent death. (That’s why suffocation is quick). Estimated that our mitochondria produce our body weight in ATP every day! The E.T.C. does NOT create ATP. It generates an electrochemical gradient across the inner mitochondrial membrane, which stores potential energy that can be used to phosphorylate ADP ATP. How it works: The Energy-Coupling Mechanism o Chemiosmosis = the coupling of exergonic e- flow down an e- transport chain to endergonic ATP production by the creation of a proton gradient across a membrane o Proposed by Peter Mitchell (1961) o The term chemiosimosis emphasizes the coupling between (1) chemical reactions (phosphorylation) and (2) transport processes (H+ transport) o Site of oxidative phosphorylation = the Inner Mitochondrial Membrane cristae = the infoldings of the inner mitochondrial membrane These increase the surface area available for chemiosmosis to occur Contain ATP synthase = protein complexes that function as enzymes to catalyze the production of ATP o Use the existing proton gradient from the ETC Process: o 3 of the carriers in the chain are also H+ pumps o NADH drops its electrons off at the first one, and they pass through all three on their way to oxygen. o FADH2 drops its e-s off at a different carrier after the first one, so its electrons only pass through two of the pumps. o When each of these three pick up the electrons, they pump a H+ out of the matrix (LOW [H+]) to the intermembrane space (HIGH [H+]). o This creates a proton gradient, which is maintained because the membrane is impermeable to H+ and prevents them from leaking back across the membrane by diffusion o This creates potential energy o ATP Synthase uses the potential energy stored in the proton gradient to make ATP by allowing H+s to diffuse through it back across the membrane o The drop in potential energy can then be captured in the bonds that attach the last phosphate to ADP ATP. The process is similar to a battery: (We’ll draw it in class) The proton gradient that results is called the Proton Motive Force = potential energy stored in the proton gradient created across biological membranes that are involved in chemiosmosis This is an electrochemical gradient with: o Concentration gradient of protons (chemical gradient) o Voltage across the membrane b/c of a higher concentration of positively charged protons on one side (electrical gradient) It tends to drive protons across the membrane into the matrix For every one H+ pumped out in the ETC, one H+ is allowed to diffuse back in through an ATP synthase, creating one ATP Therefore, for every pair of e-s dropped off by: o NADH leads to ATP synthase creating 3 ATP o FADH2 leads to ATP synthase creating 2 ATP Final Tallies of ATP From Glycolysis: 2 ATP (net gain) From Citric Acid Cycle: 2 ATP ATP yield from the ETC is often slightly lower than predicted because the electrons from NADH created in glycolysis must be transported across the membrane by one of several “shuttle” carriers. Depending on which is working, they are either transferred to NAD+ or FAD. If FAD is used, the yield is lower. (Complete on your own or wait until we do this in class…) Process ATP Produced Directly by Substrate-Level Phosphorylation Reduced Coenzyme ATP Produced by Oxidative Phosphorylation ATP NADH ATP Prep Step ATP NADH ATP Citric Acid Cycle ATP NADH ATP FADH2 ATP Glycolysis (Net) TOTAL: Total ATP 8-4 Notes We learned yesterday how ATP is made through Oxidative Phosphorylation in the mitochondrion. The key is the coupling of the proton gradient created by the ETC and the exergonic diffusion of protons back into the matrix by the ATP synthase complex. Even with this awesome process, only about 34% of the energy released from the proton gradient is captured in the bonds of ATP. The rest is lost to the cell/organism as heat. Occasionally, it is beneficial to actually decouple the creation of ATP from the diffusion of H+s back down their electrochemical gradient. If that happens, none of the energy is captured in the bonds of ATP and all of the potential energy is converted to heat. This is beneficial in thermoregulation of animals, as seen in: o Brown fat in newborns and other species o Hibernating mammals If they didn’t do this, they would generate so much ATP, cellular respiration would shut down completely CATABOLISM We don’t eat just glucose, so how do we get energy (ATP) from other food sources? Things our body does: Carbohydrate digestion: o Starch is hydrolyzed into individual glucose molecules in digestion o In between meals, glycogen is hydrolyzed into individual glucose molecules by glucagon o In the small intestine, carbohydrates are broken down into glucose Protein digestion: o Proteins are hydrolyzed into amino acids in the stomach and small intestine Amino acids are used as monomers to make new proteins OR Deaminated (have the amino group removed) to a carbon skeleton, which can be converted into acetyl groups (like acetyl CoA) or can enter the citric acid cycle at some juncture Fat digestion: o Fats can be hydrolyzed in the small intestine into glycerol and fatty acids o Glycerol can be converted to pyruvate and enter at the end of glycolysis o Fatty acids can be broken down into 2-C acetyl CoA’s that enter the citric acid cycle Ex. A single 18-C fatty acid chain 9 acetyl CoA molecules These can enter the Citric Acid Cycle and form: o 27 NADH o 9 FADH2 o 9 ATP in the Citric Acid Cycle The NADH can create a total of 27 x 3 = 81 ATP in the ETC The FADH2 can create a total of 9 x 2 = 18 ATP in the ETC Added together with the ATP from the Citric Acid Cycle = 108 ATP!!! o This is why fats are an efficient form of stored energy: one triglyceride can produce a lot of ATP when needed ANABOLISM Building new molecules often requires the use (breakdown) of ATP. These catabolic pathways we’ve discussed explain how to replace those ATP molecules. Biosynthetic Needs: Photosynthesis uses ATP to help build glucose from CO2 and H2O (next unit). Excessive carbohydrate intake results in fat formation o Extra G3P molecules from glycolysis glycerol o Extra acetyl groups from glycolysis fatty acids o Put together = FAT Some substrates from the citric acid cycle can be transaminated into amino acids o 9 amino acids are “essential” we can’t make them o The other 11 we can created by altering the others through metabolic pathways Control of Cellular Respiration The process of cellular respiration, like all other pathways of the cell, must be finely controlled. Uncontrolled, your body would burn through all your food very quickly, without anywhere to put the excess energy. o This energy would then be converted to heat and lost from the cell. By shutting down the pathway at times, it allows the cell to control the amount of ATP produced and not waste precious food energy. Although there are several control points in the process, the one you need to know the best is an enzyme in the glycolysis pathway called phosphofructokinase. o It is involved with the third step in the glycolytic pathway, in which it acts as an enzyme that phosphorylates the original glucose molecule for the 2nd time by transferring a phosphate from ATP to the substrate. o It is an ALLOSTERIC enzyme It is activated by the presence of AMP It is inhibited by excess ATP and by citric acid (from the citric acid cycle) This is a form of feedback inhibition