* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download cellular respiration
Fatty acid metabolism wikipedia , lookup
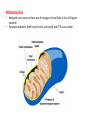
Mitochondrion wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Metalloprotein wikipedia , lookup
Blood sugar level wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Electron transport chain wikipedia , lookup
Phosphorylation wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
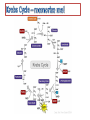
Citric acid cycle wikipedia , lookup
CELLULAR RESPIRATION Glycolysis Pyruvate Oxidation (Anaerobic) (Aerobic ) Krebs cycle Electron Transport Chain Two mechanisms for generating ATP • Substrate level phosphorylation (directly) – responsible for 4 of 36 ATP made • Oxidative phosphorylation (indirectly) – responsible for remaining 32 ATP made much more to come later… C6H12O6 + 6O2 -------> 6CO2 + 6H2O + energy (Δ G = - 2870 KJ/mol glucose) Oxygen is not the only possible electron acceptor in the oxidation of glucose in a cell. obligate anaerobes – micro-organisms that use NO2, SO4, CO2 as final electron acceptors (cannot live in the presence of oxygen) obligate aerobes – most animals, plants, fungi and bacteria require oxygen as the final electron acceptor (most eukaryotes) facultative anaerobes – organisms that can tolerate aerobic and anaerobic conditions (mostly bacteria) Mitochondria • designed to increase surface area for oxygen to bind (why is lots of Oxygen needed) • Pyruvate oxidation, Kreb’s cycle (citric acid cycle) and ETC occur inside Glycolysis (sugar-splitting) • occurs in the cytoplasm • first 10 reactions of cellular respiration • glucose backbone is essentially split in half http://www.accessexcellence.org/RC/VL/GG/ecb/ecb_images/Panel_13_01Glycolysis.pdf Glycolysis is not highly efficient in energy harnessing; 2 ATP x 30.5 KJ/mol = 61 KJ total free energy in 1 mol of glucose = 2870 KJ energy conversion efficiency = 61 KJ x 100% = 2.1% 2870 KJ Glycolysis is most likely the earliest form of metabolism, provides enough energy for smaller simple organisms, but does not provide enough energy for multicellular organisms. 2 ATP molecules are used in first 5 steps these reaction essentially prime the glucose molecule by adding phosphates (costs) Fructose 1,6-bisphosphate is split into dihydroxyacetone phosphate (DHAP) and glyceraldehyde 3phosphate (G3P). (DHAP is quickly converted to G3P) 2 NADH molecules are produced as 2 molecules of G3P processed 2 ATP are produced by substrate level phosphorylation, as 1,3 bisphosphoglycerate (BPG) are processed 2 more ATP are produced by substrate level phosphorylation as 2 molecules of phosphoenolpyruvate (PEP) are converted to pyruvate molecules Overall Equation Glucose + 2ADP + 2Pi + 2NAD+ -----> 2 pyruvate + 2ATP + 2 (NADH + H+) 4 ATP produced 2 ATP used . 2 ATP produced (net) 2 NADH produced (for further processing) Pyruvate Oxidation… Glucose Glycolysis