* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cell Physiology
Vectors in gene therapy wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Biochemical cascade wikipedia , lookup
Magnesium in biology wikipedia , lookup
Photosynthesis wikipedia , lookup
Lipid signaling wikipedia , lookup
Western blot wikipedia , lookup
Signal transduction wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Microbial metabolism wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Mitochondrion wikipedia , lookup
Biochemistry wikipedia , lookup
Electron transport chain wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Citric acid cycle wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
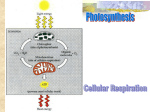
Ch. 2 Cell Physiology Objectives • Understand what the Cell Theory states • Identify organelles and state their function • Know how Mitochondria produce ATP The Cell Theory • All living things are composed of cells • Cell are the smallest unit of life • All cells come from pre-existing cells • The cell is the structural and functional unit of life Cells • Cells vary in size • Small size of cells makes them efficient • Focus on eukaryotic cells Two major kinds of cell • Eukaryotic cell – Complex organization – True nucleus present • Stores DNA – Many membrane bound organelles • Specialized for a particular function • Not all membrane bound – Cell wall present in some organisms – Size: 10 – 100 μm Eukaryotic Cell Structure • Nucleus – Control center of the cell – Bound by nuclear membrane – Stores hereditary information • Nucleolus – Within nucleus – Responsible for making rRNA • Ribosome subunits • Ribosomes – Sites of protein synthesis in a cell • Two subunits – Three binding sites where mRNA and tRNA interact – Free floating or attached to membranes Eukaryotic Cell Structure • Endoplasmic reticulum (ER) – Two types • Rough ER – Synthesis of proteins for export and membrane construction – Ribosomes present • Smooth ER – Synthesis of lipids – Detoxification of drugs and poisons – produces vesicles for transport Eukaryotic Cell Structure • Golgi Complex – UPS of the cell – Receive molecules from the ER • Modify, sort, and deliver – Vesicles do the delivering – Vesicles are specific in the cargo they carry Specificity of Vesicles • Finished proteins contain sorting signals – Bind to specific recognition sites • Coatomers bind to vesicle membrane surface – Cause bulging to occur – Eventually buds off • V-SNARE bind to t-SNARE on plasma membrane – Ensures that content of vesicle is secreted out of the cell Eukaryotic Cell Structure • Lysosomes – Digestive compartments • Hydrolysis • Breakdown old or damaged organic matter – Autophagy • Foreign material – Endocytosis » Phagocytosis » Pinocytosis » Receptor-mediated – Also function in programmed cell death – Defects may lead to storage disorders • Pompe’s disease • Tay-Sachs disease Eukaryotic Cell Structure • Peroxisomes – Contain oxidative enzymes – Enzymes remove H from various substrates and add it to O, producing hydrogen peroxide • catalase – Have many functions • Metabolism of fatty acids • Detoxification Mitochondria and ATP Production • Mitochondria – The powerhouse of the cell – Site of cellular respiration and ATP production • Endosymbiotic theory – Believed to have been prokaryotes that formed a symbiotic relationship with precursor eukaryotic cells • Cellular respiration – – – – Glycolysis Acetyl CoA production Citric acid cycle Electron transport chain Overview of ATP Production Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Key Glucose Carbon atoms ATP Phosphate groups 1 Phosphorylation ADP Glucose 6-phosphate Glycogen Fat Fructose 6-phosphate ATP 2 Priming ADP Fructose 1,6-diphosphate 3 Cleavage 2 PGAL 2 Pi 2 NAD+ 2 NADH + 2 H+ 4 Oxidation 2 2 ADP 2 H2O 2 ATP 2 5 Dephosphorylation 2 ADP 2 ATP 2 2 pyruvic acid 2 NADH + 2 H+ 2 NAD+ 2 2 lactic acid Anaerobic fermentation Aerobic respiration End-products of glycolysis are: 2 pyruvic acid + 2 NADH + 2 ATP + 2 H+ Aerobic Respiration • Most ATP generated in mitochondria – Oxygen required as final electron acceptor • Pyruvate decarboxylated – Combines with coenzyme A to enter matrix • Occurs in two principal steps: – Matrix reactions – controlling enzymes are in the fluid of the mitochondrial matrix – Membrane reactions - controlling enzymes are bound to the membranes of the mitochondrial cristae Mitochondrial Matrix Reactions Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Pyruvic acid (C3) 6 CO2 NAD+ 7 NADH + H+ Acetyl group (C2) 8 Acetyl-Co A Coenzyme A H2O 9 Citric acid (C6) Oxaloacetic acid (C4) H2O 10 NADH + H+ NAD+ (C6) Citric acid cycle 18 H2O NAD+ 11 NADH + H+ (C4) 12 CO2 17 (C5) H2O NAD+ 13 Occurs in mitochondrial matrix (C4) 14 16 FADH2 NADH + H+ (C4) CO2 FAD (C4) Pi 15 GTP ADP GDP ATP Membrane Reactions • Membrane reactions have two purposes: – to further oxidize NADH and FADH2 and transfer their energy to ATP – to regenerate NAD+ and FAD and make them available again to earlier reaction steps • Mitochondrial electron-transport chain – series of compounds that carry out this series of membrane reactions Members of the Transport Chain • Flavin mononucleotide (FMN) – derivative of riboflavin similar to FAD – bound to a membrane protein FMN accepts electrons from NADH • Iron-sulfur (Fe-S) centers – complexes of iron and sulfur atoms bound to membrane proteins • Coenzyme Q (CoQ) – accepts electrons from FADH2 – small mobile molecule that moves about in the membrane • Copper (Cu) ions – bound to two membrane proteins • Cytochromes – five enzymes with iron cofactors – brightly colored in pure form – in order of participation in the chain, b, c1, c, a, a3 Electron Transport • hydrogen atoms are spilt apart as they transfer from coenzymes to the chain • protons pumped into the intermembrane space • electrons travel in pairs (2 e-) along the transport chain • each electron carrier becomes reduced when it receives an electron pair and oxidized again when it passes the electrons along to the next carrier • oxygen is the final electron acceptor – each oxygen atom accepts two electrons from cytochrome a3 and two protons from the mitochondrial matrix forming water • body’s primary source of metabolic water – water synthesized in the body – this reaction explains the body’s oxygen requirement – no oxygen, cell produces too little ATP to sustain life Electron Transport Chain Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 50 NADH + H+ Relative free energy (kcal/mole) 40 NAD+ FADH2 Enzyme complex 1 F AD 30 1 20 Enzyme complex 2 10 ½ O2 + 2 H+ Enzyme complex 3 0 Reaction progress H2O Chemiosmotic Mechanism • electron transport chain energy fuels respiratory enzyme complexes – pump protons from matrix into space between inner and outer mitochondrial membranes – creates steep electrochemical gradient for H+ across inner mitochondrial membrane • inner membrane is permeable to H+ at channel proteins called ATP synthase • chemiosmotic mechanism - H+ current rushing back through these ATP synthase channels drives ATP synthesis Chemiosmotic ATP Synthesis Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Intermembrane space Matrix Cristae Figure 26.6 Inner membrane Outer membrane NADH + H+ ½ O2 + 2 H+ NAD+ 6 H+ H2O Matrix 2e– Inner membrane Enzyme complex 1 CoQ 2e– 2e– Enzyme complex 2 Enzyme complex 3 Cyt c Intermembrane space Outer membrane 2 H+ 2 H+ 2 H+ 3 ADP + 3 Pi 3 ATP ATP synthase Overview of ATP Production • NADH releases an electron pair to electron transport system and H+ to prime pumps – enough energy to synthesize 3 ATP • FADH2 releases its electron pairs further along electron-transport system – enough energy to synthesize 2 ATP • complete aerobic oxidation of glucose to CO2 and H2O produces 36-38 ATP – efficiency rating of 40% - 60% is lost as heat ATP Generated by Oxidation of Glucose Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Glucose 2 Glycolysis ATP (net) 2 NADH + 2 H+ Cytosol 2 pyruvate Mitochondria 2 NADH + 2 H+ CO2 6 NADH + 6 H+ Citric acid cycle 2 ATP Figure 26.7 2 FADH2 Electron-transport chain O2 H2O 4 ATP 28–30 Total 36–38 ATP ATP Vaults and Cytosol • Vaults – May function as cellular transporters – True function unknown • Cytosol – Gel-like semiliquid that occupies most of the cell volume – involved with • Intermediary metabolism • Ribosomal protein synthesis • Inclusions The Cytoskeleton • Structural support of the cell • Composed of three types of protein fibers – Microtubules • Tubulin • Maintain shape of asymmetrical cells • Also functions in cellular locomotion and movement – Microfilaments • Actin • Cellular contractile systems • Mechanical stiffeners – microvilli – Intermediate filaments • Resist mechanical stress The Cytoskeleton The Cytoskeleton • Centrioles – Assembly of microtubules – Function in cell divison • Other functions – Cellular movement • Cilia and flagella – Transport of materials • Dynein-kinesin motor proteins The Plasma Membrane • Surrounds all living cells • Composed of a phospholipid bilayer – Polar hydrophilic heads on the outside, nonpolar hydrophobic tails on the inside • Fluid mosaic model – Proteins and cholesterol embedded Beyond the Plasma Membrane • Extra Cellular Matrix – Surrounds plasma membrane of animal cells • Protection and regulation