* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Application Note - Horizon Discovery
Signal transduction wikipedia , lookup
Tissue engineering wikipedia , lookup
Extracellular matrix wikipedia , lookup
Biochemical switches in the cell cycle wikipedia , lookup
Spindle checkpoint wikipedia , lookup
Cell growth wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
List of types of proteins wikipedia , lookup
Microtubule wikipedia , lookup
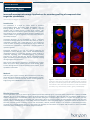
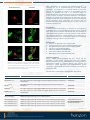
HORIZON DISCOVERY Application Note Immunofluorescent microscopy: Applications for secondary profiling of compounds that target the cytoskeleton David Sorrell, Ceri Wiggins and Rebecca Foster Introduction Fluorescent microscopy is the main tool used for examining the cytoskeleton. Horizon Discovery offers custom immunofluorescence imaged-based assays to support the secondary cellular profiling of compounds originating from cytoskeletal-targeting drug discovery programs (e.g. Aurora and Polo-like kinases, microtubule-binding agents, or kinesins) or where a compound series is suspected of having off-targets against the cytoskeleton. Here the use of fluorescence microscopy to characterize the on-target effects of an Aurora kinase inhibitor and two tubulin-binding agents, and the off-target effects of a broad-spectrum kinase inhibitor, on the cytoskeleton is described. Methods Cells were grown on glass coverslips. After treatment they were fixed, stained with tubulin antibodies or Alexa Fluor 488-phalloidin to visualize F-actin, mounted in anti-fade mounting medium containing DAPI and imaged using a Zeiss LSM500 Meta confocal microscope. Control Interphase VX680 * * * Paclitaxel Unintended disruption of the cytoskeleton is also a commonly encountered off-target effect of small molecules, which can result in undesirable cytotoxity3. This is most likely due to the promiscuous nature of tubulin in binding a structurally diverse range of molecules and because the cytoskeleton is regulated by a complex network of upstream regulators. Mitosis Nocodazole The cytoskeleton is a target for several classes of important pharmacological agents. Anti-mitotics target microtubules, either by binding directly to tubulin, or indirectly by inhibiting upstream regulators such as mitotic kinases or kinesin motor proteins1. This results in disruption of the bipolar spindle and mitotic arrest. Similarly, actin filaments can be modulated by direct binding of inhibitors or by indirect inhibition of upstream regulators, which can result in effects on cell motility and shape2. Figure 1. Differential phenotypes resulting from anti-mitotic treatment. HeLa cells were treated with VX680 (0.5 µM), paclitaxel (0.1 µM) or nocodazole (1 µM) for 24h. Mini-spindles are indicated with asterisks. Cells were fixed and stained to reveal microtubules (α-tubulin; red), centrosomes (ϒ-tubulin; green) and DNA (blue). Results & Discussion The on-target effects of three anti-mitotic agents, paclitaxel (microtubule stabilizing agent), nocodazole (tubulin polymerization inhibitor) and VX680 (MK-0457; pan-Aurora kinase inhibitor) on mitotic spindle microtubules were investigated in HeLa cells after 24h of treatment. VX680 induced a number of spindle defects consistent with previous reports4. Most striking was a novel phenotype in which bi- or tripolar mini-spindles, that were typically separated from the main chromosomal mass, were formed (Figure 1). Increased numbers of multinucleated interphase cells were also apparent, which is consistent with the known ability of Aurora B inhibition to induce polyploidy4 (Figure 1). Nocodazole induced the formation of abnormal multipolar spindles consisting of short thick microtubules, whereas paclitaxel resulted in the formation of well-developed ‘bushy’ monopolar and multipolar spindles (Figure 1). These agents also distorted the microtubules of interphase cells compared to control and VX680treated cells, which exhibited long and relatively straight microtubules. Control Staurosporine α-tubulin α-tubulin These observations are consistent with published literature5,6. To investigate the use of immunofluorescence to identify off-target phenotypes, we examined the non-specific effects of the broadspectrum kinase inhibitor staurosporine, on microtubules and actin filaments in MCF10A cells after a 2h treatment (Figure 2). Staurosporine-treated cells adopted a striking star-like morphology due to the presence of multiple cytoplasmic extensions. Staurosporine treatment also resulted in the disappearance of stress fibers that were present in control cells and the accumulation of F-actin at the periphery of the extensions. These phenotypes are consistent with the uncoordinated lamellipodial activity reported for other cell lines in response to staurosporine7. Conclusion Fluorescent confocal microscopy is an important tool for studying the cytoskeleton since it can extract detailed information at the single cell level, which cannot be achieved with Western blot, flow cytometry or plate-reader-based approaches commonly favored for drug discovery. Two important applications of these methods are to confirm the ontarget mechanism-of-action of anti-mitotic agents and for identifying unintended ‘off-target’ effects on the cytoskeleton. F-actin F-actin References 1. 2. 3. 4. 5. 6. 7. Merge + DAPI Merge + DAPI Figure 2. Staurosporine induces an altered morphology and F-actin redistribution. MCF10A cells were treated for 2 h with 2 µM staurosporine, fixed and stained to reveal microtubules (α-tubulin; red), F-actin (Alexa Fluor 488-phalloidin; green) and DNA (blue). Jackson et al., Nature Rev Cancer 2007 (7) p107 Fenteany and Zhu, Curr Topics in Med Chem 2003 (3) p593 Ross-Macdonald et al., Mol Cancer Ther 2008 (7) p3490 Tyler et al., Cell Cycle 2007 (6) p2846 Ywon et al., Mol Biol Cell 1999 (10) p947 Jordan et al., J Cell Sci 1992 (102) p401 Mannherz et al., Eur J Cell Biol 2006 (85) p785 Horizon Support Horizon supplies genetically-defined cell lines, custom cell line generation, in vivo models, reporter gene assay kits, molecular reference standards and assay development and screening services to organizations engaged in academic research; drug discovery and development; clinical diagnostics; and biopharmaceutical process optimization. Please contact us to learn more about how Horizon can support your work. Cell lines similar to those listed in this Application Note include: Name Description Catalogue No. p53 (R175HInducible) DLD-1 Over-expression of GFP-tagged mutant (R175H) p53 induced in the absence of tetracycline HD R00-010 p53 (wtInducible) DLD-1 Over-expression of GFP-tagged wild type p53 induced in the absence of tetracycline HD R00-012 PUMA (wtInducible) DLD-1 Over-expression of GFP-tagged PUMA induced in the absence of tetracycline HD R00-014 TGFβRII (wtInducible) DLD-1 Over-expression of HA and GFP dual tagged TGFβRII induced in the absence of tetracycline Over-expression lines HD R00-016 HaloTag® reporter kits K-Ras (+Halotag/+) SW48 Heterozygous HaloTag fusion to wild type K-Ras K-Ras (G12CHaloTag®/+) SW48 Heterozygous HaloTag reporter fusion to K-Ras activating mutation (G12C) K-Ras (G12DHaloTag®/+) SW48 Heterozygous HaloTag reporter fusion to K-Ras activating mutation (G12D) K-Ras (G12VHaloTag®/+) SW48 Heterozygous HaloTag reporter fusion to K-Ras activating mutation (G12V) K-Ras (G13DHaloTag®/+) SW48 Heterozygous HaloTag reporter fusion to K-Ras activating mutation (G13D) Contact Horizon or go to www.horizondiscovery.com for more information. © 2015 v-01 t + 44 (0)1223 655580 f + 44 (0)1223 655581 e [email protected] w www.horizondiscovery.com Horizon Discovery, 7100 Cambridge Research Park, Waterbeach, Cambridge, CB25 9TL, United Kingdom