* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 5 Kingdoms of Life - Cellular
Polyadenylation wikipedia , lookup
Endomembrane system wikipedia , lookup
Molecular evolution wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Western blot wikipedia , lookup
Non-coding RNA wikipedia , lookup
Gel electrophoresis of nucleic acids wikipedia , lookup
Non-coding DNA wikipedia , lookup
Gel electrophoresis wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Epitranscriptome wikipedia , lookup
Protein moonlighting wikipedia , lookup
Expanded genetic code wikipedia , lookup
Gene expression wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Genetic code wikipedia , lookup
Protein structure prediction wikipedia , lookup
Protein adsorption wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Proteolysis wikipedia , lookup
List of types of proteins wikipedia , lookup
Deoxyribozyme wikipedia , lookup
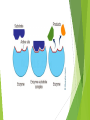
Biological Molecules – BZ 21 - 27 + Water H2O BZ 21 Biological Molecules Water, carbohydrates, lipids and nucleic acids are the molecules that make up all living things. Start BZ 21 Structures of Biological Molecules Construct a map of the terms only BZ 21 - Water Water is required for life to exist because it is: Universal solvent – metabolic reactions occur in it easily. Liquid at room temperature + low viscosity (flows easily and animals can swim through it easily) Thermally very stable and absorbs a lot of heat before changing states. Ice floats in water so the underlying water is insulated High surface tension – forms droplets easily and flows easily through narrow spaces – capillary action. Colourless and allows high transmission of light. Complete BZ 21 Do not draw BZ 21 Ans BZ 22 – Carbohydrates - CHO’s – saccharides CHO’s are a family of organic (containing Carbon) molecules made up of only carbon, hydrogen and oxygen. created by photosynthesis in chloroplasts • consumed by mitochondria to release energy in cellular respiration • Monosaccharides - ribose, glucose, fructose • Disaccharides – Maltose, Lactose • Polysaccharide molecules • cellulose – wood/cell walls • starch – stored energy in plants, • chitin – arthropod exoskeletons, fungi cell walls • glycogen – stored energy for animals BZ 22 Ans BZ 23 - Lipids Lipids are non-polar (non-charged), hydrophobic organic molecules, that have many important biological functions. Characteristics Oily, greasy or waxy. Relatively insoluble in water Higher concentration of energy than CHO’s or protein Functions Protection of organs and insulation Energy storage Plasma membranes Hormones - steroids BZ 23 Ans BZ 24 – Nucleotides and Nucleic acids Nucleotides are the building blocks of DNA and RNA. Nucleic acids are long chains of nucleotides that store and transmit genetic information. Nucleotide Structure Base + sugar + phosphate group DNA - Base + deoxyribose + phosphate group RNA - Base + ribose + phosphate group Functions of nucleotides Building blocks of DNA or RNA (nucleic acids) Nucleotide ATP derivatives (adenosine tri-phosphate) – high energy bonds DNA BZ 24 Ans DNA and RNA DNA – double helix DNA replication - with animations BBC Bitesize, RNA - protein synthesis - BBC Bitesize RNA – single helix BZ 25 Amino Acids Amino acids can be joined together to form polypeptides. Proteins are made up of one of more polypeptide molecules. Characteristics 2 or more amino acids form peptides Long chains of amino acids form proteins Don’t Draw Glutamine BZ 25 Ans BZ 26 – Globular proteins – structure = function Protein structure is related to its biological function. Properties: 3 Don’t draw Structural levels of Proteins – stop around 5 mins Soluble in water Geometry is critical to its function Folded into a spherical shape Functions: Catalytic - Enzymes Regulatory – Hormones Transport – Haemoglobin Protective - Immunoglobulins BZ 26 Fibrous proteins – structure = function Fibrous proteins Properties: Don’t draw Insoluble Tough physically Parallel in water polypeptide chains Functions: Structural - collagen (connective tissue), spider silk. Contractile – myosin and actin filaments in muscles. BZ 26 Ans BZ 27 Enzymes – All about enzymes youtube Enzymes are biological catalysts that speed up reactions but are not consumed by them and therefore can be re-used repeatedly. They are effective in low concentrations very specific (only catalyze one type of reaction) deactivated or denatured by extremes of heat and pH Properties: The lock and key model (image over page) Active site – specific shape (drugs used to kill parasites can either block or alter the active site on their enzymes) Substrate – the specific chemical it works on Fully re-usable Explaining the shape of an enzyme graph Heating a reaction makes the molecules move faster therefore collide more often. More collisions in a given time means a faster reaction rate. Over heating the enzyme will denature it - alter its shape so it no longer fits the substrate. Changes in pH have similar effects. Saliva contains Amylase, an enzyme that converts starches into simple sugars. If you added amylase to a solution of starch, all of the starch is broken down – regardless of the quantities - why? BZ 27 Ans