* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Algebra 1 - Edublogs
Extracellular matrix wikipedia , lookup
Endomembrane system wikipedia , lookup
Cell growth wikipedia , lookup
Cytokinesis wikipedia , lookup
Tissue engineering wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell culture wikipedia , lookup
Cell encapsulation wikipedia , lookup
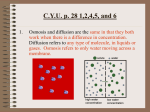
Cells OSMOSIS Aims: Compare hypotonic and hypertonic solutions Describe and differentiate types of passive transport Predict and explain the effects of osmosis on cells Agenda: T&T Diagram Debrief Concept Map Test Wizard Passive Transport Check Egg Demo Activity 5.5 Extension (free-response and POS 5.5) Test Wizard Engage: IV/Salty food/Finger…Elicit that all relate to osmosis, a type of passive transport in cells. Explain: Debrief diagrams in Do Now for following points Difference between iso/hypo/hyper Similarities and differences in types of passive transport Show water and food color as a quick demo to elicit high to low concentration and no energy. Concept map Poll Explore Effects of Osmosis on Egg Cells Explain that now we will focus on osmosis and how it affects cells Students observe demos and explain after seeing exemplar Students use Mybiology Activity 5.5 or below links to explain o http://www.biology.ualberta.ca/facilities/multimedia/uploads/alberta/dif.swf o http://www.coolschool.ca/lor/BI12/unit4/U04L06/rbc.html Remind that often water moves rather than large solute which can’t pass through membrane. Show pp. 1-2 of MyBiology 5.5 if further explanation needed Extend Predicting effects on cells in table Osmosis scenarios POS 5.5 Exit Slip: MC questions Homework: Active Transport Annotation Cells OSMOSIS Do Now 1. Which area, X or Y, is more concentrated? Explain. X Y 2. Label each SOLUTION as hypo-, iso- or hypertonic compared to the cell. Briefly explain your choices. Solution Solution Cell Solution Cell Cell 3. Give a caption to and annotate each diagram below. Solute 4. What do all of the processes shown above have in common? Cells OSMOSIS Exploring & Explaining Osmosis In this demo, one egg was placed in a hypotonic solution (0.0 M, or pure water) and another egg was placed in a hypertonic solution (0.6 M sucrose). Working in pairs, explain what occurred to each egg cell using your knowledge of osmosis. Watch MyBiology Activity: Osmosis and Water Balance in Cells (5.5) pp.3-4 for help. Solution in Beaker What happened to the egg cell? Why did this happen? Isotonic (0.3 M) The egg cell did not change in In an isotonic environment the concentration size . of water and solutes inside the cell is the same as the solution outside. This is equilibrium, and no overall water movement by osmosis occurs. Hypotonic (0.0 M) Hypertonic (0.6 M) If time, check out the following links for more effects of osmosis on plants and animals: o http://www.coolschool.ca/content/showcase.php?type=science o http://www.biology.ualberta.ca/facilities/multimedia/uploads/alberta/dif.swf Rules of Osmosis Cells OSMOSIS Extending Osmosis 1. Complete the table below. Environment Plant cell Animal cell Plant cell Animal cell Plant cell Plant cell Animal cell Water concentration is highest in… Water will… Cell will … Hypotonic Hypertonic Hypertonic Hypotonic Isotonic Hypertonic Hypotonic 2. In the winter when roads are salted, plants that grow along the road often die. Explain why using concentration, cells, water, low, high, osmosis. _________________________________________________________________________ _________________________________________________________________________ _________________________________________________________________________ _________________________________________________________________________ _________________________________________________________________________ 3. Explain why you feel thirsty after eating very salty or very sweet foods using the terms concentration, cheek cells, low, water, high, osmosis in your explanation. _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ 4. Hospital patients that are hooked up to an IV receive a slightly saline (salty) solution rather than pure water. Explain why using the terms concentration, cells, low, water, high, osmosis in your explanation. _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ 5. When a person soaks in a bath tub for a long time, their fingers swell and wrinkle. Explain why using the terms concentration, cells, low, water, high, osmosis. ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ 6. Complete Process of Science: How Does Osmosis Affect Cells? (5.5) at MyBiology.com Cells Exit Slip OSMOSIS Cells OSMOSIS Animation Guided Questions Go to http://www.northland.cc.mn.us/biology/Biology1111/animations/passive1.swf 1. What is passive transport, in your own words? 2. What are three types of passive transport? Tour the Cell membrane… 3. Why is it important that the membrane is fluid? 4. What kinds of molecules don’t pass easily through the lipid bilayer? 5. What kinds of molecules pass easily through the lipid bilayer? Continue to Simple Diffusion… 6. What is diffusion? 7. In which direction (hi to low or low to hi) do molecules moves along a concentration gradient? 8. When does diffusion end? 9. What does equilibrium mean? Continue to Facilitated Diffusion 10. Does diffusion require energy (ATP)? 11. Who “helps” certain molecules through the membrane? 12. How is facilitated diffusion similar to simple diffusion? 13. How is facilitated diffusion different from simple diffusion? Continue to Osmosis 14. How is osmosis similar to diffusion? 15. How is it different from diffusion? 16. What is the difference between a hypertonic solution and a hypotonic solution? 17. Water will travel by osmosis from a __________________ environment to a __________________ environment. Why does this happen, based on the principle of diffusion? 18. Water is polar. Why is it able to pass through the nonpolar lipid membrane? Cells OSMOSIS Notes Passive Transport is the movement of molecules from _______ to _______ concentration without _______________. A solution where molecules are evenly distributed is at _________________________. 3 Types of Passive Transport 1. Diffusion is the movement of _________________ from ______ to ______ concentration without ______________. Molecules that are ___________ or ___________ can diffuse across the lipid bilayer. 2. Osmosis is the movement of _________________ from _____ to _____ concentration without _____________. Osmosis is a specific type of _______________. During osmosis, water travels from a _____________________ solution to a ______________________ solution. a. Plant cells in hypertonic solution will _____________ because water ____________ the cell. b. Plant cells in a hypotonic solution will ____________ because water ____________ the cell. c. Plant cells in an isotonic solution will remain _____________ because water ___________ and _______________ the cell. d. Animal cells in a hypertonic solution will ________________ because water ____________ the cell. e. Animals cells in a hypotonic solution will ____________ because water ____________ the cell. f. Animal cells in an isotonic solution will _________________ because water ___________ and _______________ the cell. 3. Faciliated diffusion is the movement of molecules from _________ to ________ concentration with the help of ____________________. Molecules that are _____________ and ____________ cross the membrane this way. Practice 1. Identify the type of passive transport that occurs in each example. _____________________ Water enters plant cells _____________________ The odor of cologne spreads across a room. _____________________ Tea from a tea bag spreads through a cup of hot water. _____________________ Sugar enters the cell without energy but with the help of a protein Cells OSMOSIS _____________________ Water leaves cheek cells when you eat salty food. _____________________ Amino acids enter the cell without energy but with the help of a protein _____________________ Food coloring spread through a glass of water _____________________ Water leaves a red blood cell 2. Complete the Venn diagram using the following terms: Passive Water Proteins No energy Large molecules High to Low Cells Food coloring Diffusion Odor Small molecules Nonpolar molecules Plasmolysis Osmosis 2. In the diagrams below, assume that the solute can pass through the cell membrane. Label the areas of high and low concentration of solute. Then use arrows to show the direction that the solute particle will move during diffusion. 3. In the diagrams below, assume that the solute cannot pass through the cell membrane. Label the areas of high and low concentration of water molecules. Then use arrows to show the direction that water will move during osmosis. Cells OSMOSIS 3. Predict what will happen to each cell in the given conditions. Environment Plant cell Animal cell Plant cell Animal cell Plant cell Plant cell Animal cell Where is water concentration highest? Water will… Cell will … Hypotonic Hypertonic Hypertonic Hypotonic Isotonic Hypertonic Hypotonic 4. Complete the table below. Draw arrows to show the direction of water flow when necessary. What is happening to the Is water entering or Where is water cell? leaving the cell? concentration highest? Cells OSMOSIS 5. Complete the table showing the effects of osmosis on plant cells. Does water enter or leave? What is happening to the cell? Where is the concentration of water the highest? Cells OSMOSIS 6. A laboratory technician places red blood cells into three different solutions. Observations are recorded each minute for five minutes. Cells OSMOSIS A. In solution 1, is the concentration of solvent higher inside or outside the cell initially? B. In solution 1, why did the cells disappear? C. In solution 2, is the concentration of solvent higher inside or outside the cell initially? D. In solution 2, why was there no change in the cells? E. In solution 3, is the concentration of solvent higher inside or outside the cell initially? F. In solution 3, why did the cells disappear? G. What process explains the changes that occurred in these cells? H. Which of the following best explains what is causing the red blood cells in solution 1 to change size over the five-minute period? a. Solvent is entering the cells faster than it is leaving the cells. b. Solute is entering the cells faster than it is leaving the cells. c. The cells are making new protein. d. The cell’s membranes are dissolving. I. The laboratory technician concludes that red blood cells cannot function in any fluid except serum. Which of the following best characterizes this conclusion? a. It is accurate on the basis of the information given. b. It is accurate because the cells changed in all the solutions but one. c. It is inaccurate because the cells were outside the body. d. It cannot be substantiated with the data provided. Exit Slip Under what conditions will a substance be likely to enter a cell through diffusion? a. when the substance is a particle of food b. when a molecule of the substance is very large c. when the concentration of the substance is greater outside the cell than inside d. when the concentration of the substance is greater inside the cell than outside Cells OSMOSIS Do Now 1. Identify three types of diffusion. 2. What do the three types have in common? 3. Label the solute and solvent. Then draw arrows the direction that the solute particles will move during diffusion. 4. Label where the solute is hypotonic and hypertonic. Then label where water concentration is high and low. Then draw arrows to show the direction water would travel during osmosis (assume the solute cannot cross the membrane. Cells OSMOSIS Application Questions Cells OSMOSIS Diffusion Experiment Critical Concepts: 1. What do starch and glucose have in common? 2. How are starch and glucose different? Problem: How does the size of solute affect transport across a semi-permeable membrane? Hypothesis: Materials: Dialysis Tubing Beaker Glucose/Starch solution Water Iodine Pipet Glucose Test Strips Funnel Experiment Design: 1. Obtain a piece of dialysis tubing that has been soaked in water. 2. Roll the tubing between your fingers to open it. 3. Close one end of the tube by knotting it. 4. Using a funnel and graduated cylinder, place 15 mL of glucose/starch solution in the dialysis bag. 5. Smooth the top of the bag to expel air and tie off the open end, leaving enough room to allow for expansion. 6. Measure 160 mL distilled water into a beaker. 7. Add 4 mL of starch indicator solution (iodine) to beaker. 8. Place bag in beaker containing water and iodine. 9. Wait 30 minutes. 10. Test contents of bag and beaker for presence of starch (black color change) and glucose (green color change on test strip. Data: Before After Cells Location Bag Beaker OSMOSIS Starch Initial Starch Final Glucose Initial Glucose Final Results and Conclusions: 1. What represented the cell membrane in this experiment? Why? 2. Which substances (water, iodine, glucose, starch) diffused out of the bag? How do you know? Why did this happen? 3. Which substances (water, iodine, glucose, starch) diffused into the bag? How do you know? Why did this happen? 4. Which substances (water, iodine, glucose, starch) did not diffuse? How do you know? Why did this happen? 5. How do your results compare with your hypothesis? Was it supported or rejected? Use evidence to support your answer. 6. Based on your experiment, what is the relationship between size of solute and ability to diffuse across a semi-permeable membrane? Cells OSMOSIS Do Now 1. A laboratory technician places red blood cells into three different solutions. Observations are recorded each minute for five minutes. A. In solution 1, is the concentration of solvent higher inside or outside the cell initially? B. In solution 1, why did the cells disappear? C. In solution 3, is the concentration of solvent higher inside or outside the cell initially? D. In solution 3, why did the cells disappear? E. What process explains the changes that occurred in these cells? F. Which of the following best explains what is causing the red blood cells in solution 1 to change size over the five-minute period? a. Solvent is entering the cells faster than it is leaving the cells. b. Solute is entering the cells faster than it is leaving the cells. c. The cells are making new protein. d. The cell’s membranes are dissolving. G. The laboratory technician concludes that red blood cells cannot function in any fluid except serum. Which of the following best characterizes this conclusion? a. It is accurate on the basis of the information given. b. It is accurate because the cells changed in all the solutions but one. c. It is inaccurate because the cells were outside the body. d. It cannot be substantiated with the data provided. 2. Under what conditions will a substance be likely to enter a cell through diffusion? a. when the substance is a particle of food b. when a molecule of the substance is very large c. when the concentration of the substance is greater outside the cell than inside d. when the concentration of the substance is greater inside the cell than outside