* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Wasp Voodoo Rituals, Venom-Cocktails, and the Zombification of Cockroach Hosts SYMPOSIUM Frederic Libersat
Nervous system network models wikipedia , lookup
Behaviorism wikipedia , lookup
Neuroplasticity wikipedia , lookup
Neuroanatomy wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Central pattern generator wikipedia , lookup
Neuroethology wikipedia , lookup
Development of the nervous system wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Optogenetics wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
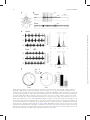
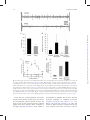
Integrative and Comparative Biology Advance Access published April 4, 2014 Integrative and Comparative Biology Integrative and Comparative Biology, pp. 1–14 doi:10.1093/icb/icu006 Society for Integrative and Comparative Biology SYMPOSIUM Wasp Voodoo Rituals, Venom-Cocktails, and the Zombification of Cockroach Hosts Frederic Libersat1 and Ram Gal Department of Life Sciences, Ben-Gurion University of the Negev, P.O. Box 653, Be’er Sheva 84105, Israel 1 E-mail: [email protected] Synopsis The parasitoid Jewel Wasp uses cockroaches as a live food supply for its developing larvae. The adult wasp uses mechanoreceptors on its stinger to locate the host’s cerebral ganglia and injects venom directly into the cockroach’s ‘‘brain,’’ namely in the subesophageal ganglion and in and around the central complex in the supraesophageal ganglion. As a result, the cockroach first engages in continuous grooming for roughly 30 min. Dopamine identified in the wasp’s venom is likely to cause this grooming, as injecting a dopamine-receptor antagonist into the cockroach hemolymph prior to a wasp’s sting greatly reduced the venom-induced, excessive grooming. Conversely, injecting a dopamine-receptor agonist into the brain induces excessive grooming in normal cockroaches. A second effect of the head-sting is the induction of a long-lasting lethargic state, during which the cockroach demonstrates a dramatically reduced drive to self-initiate locomotion. Unlike most paralyzing venoms, Ampulex’s venom seems to affect the ‘‘motivation’’ of its host to initiate locomotion, rather than affecting the motor centers directly. In fact, the venom specifically increases thresholds for the initiation of walking-related behaviors and, once such behaviors are initiated, affects their maintenance without affecting the walking-pattern generators. Thus, the venom manipulates neuronal centers within the cerebral ganglia that are specifically involved in the initiation and maintenance of walking. We have shown that in stung cockroaches focal injection of an octopaminergic receptor agonist around the central complex area in the brain partially restores walking. Another likely candidate target of the venom is the opioid system, which is known to affect responsiveness to stimuli in insects. Opioid receptor agonists increase startle threshold in control cockroaches and using a bioassay for opioid receptors, we found that the venom blocks opioid-like receptors. This effect is reversed with naloxone, an opioid antagonist. Introduction Many parasitoid wasps use insects or spiders as a food supply for their developing larvae (Quicke 1997; O’Neill 2001). To this end, the adult female typically stings the host, penetrating its cuticle with the ovipositor and injecting a venom cocktail into the hemolymph. In most cases the venom blocks the neuromuscular junction and paralyzes the host. The wasp then lays one or more eggs on, or inside, the body of the host, then conceals the host in a nest or a burrow for protection (for review, see Libersat and Gal 2007). Some parasitoid wasp species, however, exert a more sophisticated modulation of their host. These wasps may affect the central nervous system (CNS) of their hosts and induce or repress specific behaviors that accommodate the developmental constraints of their larvae (Libersat et al. 2009). For example, the braconid endoparasitoid wasp Glyptapanteles coerces its caterpillar host to behave as a bodyguard to protect its larvae (Grosman et al. 2008), and the larva of the Ichneumonid wasp Hymenoepimecis manipulates its spider host to stop building its normal orb-shaped web and to begin building a shelter-like web that will protect the larvae during metamorphosis (Eberhard 2000). These, and other, examples (Libersat and Gal 2007) provide unique insights not only into the evolutionary processes involved in the establishment of such exquisite predator–prey or parasite–host adaptations, but also into the mechanisms underlying both the ß The Author 2014. Published by Oxford University Press on behalf of the Society for Integrative and Comparative Biology. All rights reserved. For permissions please email: [email protected]. Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 From the symposium ‘‘Parasitic Manipulation of Host Phenotype, or How to Make a Zombie’’ presented at the annual meeting of the Society for Integrative and Comparative Biology, January 3–7, 2014 at Austin, Texas. 2 feed on nutritious hemolymph for the next few days. When ready to pupate, the larva penetrates through the cockroach’s cuticle, feeds on its internal organs and pupates inside the abdominal cavity. There, well satiated and safe from predators, the larva metamorphoses and emerges roughly a month later as an adult, ready to restart this unique life cycle. The second sting into the head thus evokes two different and specific behavioral manipulations in the cockroach prey: a relatively short phase of excessive grooming and a significantly longer inhibition of the cockroach’s escape behavior and spontaneous locomotion. These behavioral observations raise a number of interesting questions: does the wasp inject its venom directly into the CNS of its host? If so, to which CNS region(s) is the venom injected and how does the wasp identify the target? What neuronal modulations account for the excessive grooming behavior, and which circuits are affected, and how, to induce the long-lasting decrease in escape responses and locomotion? These are the main questions that we intend to answer in this review. The head-stinging process The site of the Jewel Wasp’s sting, as well as the unique effects of wasp’s venom on the behavior of its cockroach host, suggest that the wasp uses its stinger—a modified ovipositor—to inject venom directly inside the head ganglia of its host. However, until 2003, a direct proof of such injection of venom into the host’s CNS was lacking. Furthermore, it is only recently that the sensory mechanisms allowing the wasp to inflict this ‘‘stereotactic’’ sting have been identified. The Jewel Wasp injects venom directly inside the cockroach’s CNS To test the specific location of injection, Haspel et al. (2003) injected Jewel Wasps with a mixture of C14 radiolabeled amino acids that were incorporated into the venom. In cockroaches stung by such ‘‘hot’’ wasps, most of the radioactive signal was localized around the midline of the sub-esophageal ganglion (SEG) and in the central part of the supra-esophageal ganglion (SupEG), posterior to the central complex (CC) and around the mushroom bodies (Fig. 2A). This means that the wasp’s stinger penetrates through the protective ganglionic sheath of these two head ganglia (the cockroach’s ‘‘brain’’), such that venom is injected directly onto the neuronal targets. However, how can such a precise venom injection be achieved? Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 wasp’s and its host’s behavior. Unfortunately, however, the mechanisms used by most behavior-manipulating parasitoid wasps, namely the chemicals involved, their means of delivery, their neuronal targets, and their methods of action within the host’s CNS, are today little understood. One exception to this and probably the best and only well-understood example of underlying neuronal mechanisms of a wasp-induced behavioral manipulation is the relationship between the ampulicine Jewel Wasp (Ampulex compressa Fabricius) and its cockroach host (Libersat 2003) (Fig. 1). To supply its larvae with live cockroaches (Periplaneta americana) as a food supply, this tropical wasp first attacks a cockroach by clamping its mandibles on the pronotum or on the base of the wing and inflicting a sting into the host’s thorax. This sting temporarily inactivates central motor circuits to block motor output in the cockroach’s forelegs for 1–2 min (Moore et al. 2006), facilitating a second and longer sting, through the host’s neck and into its head. Then, after the head-sting is complete, the wasp leaves for roughly 30 min in search of a nest suitable for the development of its young; during this time, the stung cockroach shows a peculiar behavioral change: instead of escaping the scene, it engages in excessive grooming behavior and thoroughly cleans its entire body with its legs and mandibles (Weisel-Eichler et al. 1999). However, it is only when the wasp returns to the grooming cockroach that the full and most exquisite extent of the stinginduced behavioral modulation can be observed. At this stage, between 30 and 40 min after the sting and lasting a few days, the cockroach becomes what can be figuratively described as a docile ‘‘zombie’’ that is completely compliant with the wasp’s needs. Upon returning to its stung prey the wasp first uses its strong jaws to break the cockroach’s antennae close to their base and then sucks up the exuding fresh hemolymph from the stumps. The stung cockroach, nevertheless, neither flees nor fights off the wasp, allowing its predator to grab one of its antennal stumps and escort it in a long walk to the preselected nest. Guided by the wasp, the docile cockroach enters its ‘‘tomb’’ and remains immobile as the wasp lays an egg and glues it onto the mid-leg cuticle (Fouad et al. 1994). Although not paralyzed, the ‘‘zombie’’ cockroach does not escape the nest as the wasp searches around for small items (leaves, pebbles etc.) with which to seal the entrance, before flying away. Entombed inside the sealed nest, the stung cockroach then serves as living food storage for the developing larva that hatches a couple of days later and perforates the host’s leg cuticle to F. Libersat and R. Gal Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 Fig. 1 Life cycle of the parasitoid Jewel Wasp and the effects of its venom on cockroach behavior Upon striking the cockroach, the female Jewel Wasp grabs it by the pronotum and stings it in the thorax. This sting induces a short-term (3–5 min) flaccid paralysis of the front legs, which is mediated by venom compounds that open chloride channels in thoracic neurons. This paralysis facilitates a second, and longer, sting in the head, after which the wasp grooms thoroughly, leaves the cockroach and goes in search for a suitable nest (here, a glass vial). In the meantime, as a result of the head-sting, the cockroach grooms excessively for 30 min. This is presumably due to venom-induced activation of dopamine-related circuits within the cockroach’s brain. The wasp then returns to the cockroach, cuts off both its antennae and drinks the hemolymph oozing from the stumps. At this stage, the cockroach enters the third phase of the envenomation process, namely a ‘‘lethargic’’ state during which it does not self-initiate walking or escape responses. The wasp then transports the cockroach to the nest, holding its antennal stump and walking backwards while the ‘‘lethargic’’ cockroach follows in a docile manner. Next, the wasp glues one egg onto the cockroach’s leg, seals the nest with leaves and pebbles, and then leaves. Under laboratory conditions, the cockroach will remain ‘‘docile’’ for 3–6 days and then resume normal activity. However, in the natural setting, the wasp’s larva hatches from its egg and feeds of the cockroach’s hemolymph. After a few days, it penetrates the host’s abdomen to feed on its internal organs, ultimately killing the cockroach before the venom’s effect dissipates. The larva then pupates inside the cockroach’s abdomen, metamorphoses and hatches as an adult approximately a month later. Wasp ‘zombifies’ cockroach host 3 4 F. Libersat and R. Gal The Jewel Wasp uses sensory cues from its stinger to identify the cockroach’s CNS In a recent study, Gal et al. (2014) hypothesized that specialized sensory organs on the wasp’s stinger (Fig. 2B) allow the wasp to discriminate the host’s brain from other tissues inside the head capsule. In this scenario, the wasp would identify the brain in a manner similar to the mechanism that other parasitic wasps use to locate a hidden host within a surrounding substrate (LeRalec et al. 1996; Vilhelmsen 2000; Vilhelmsen et al. 2001). To test this hypothesis, possible sensory organs on the stinger were ablated with liquid nitrogen and the wasps were allowed to sting normal cockroaches. The treatment prolonged the stinging duration more than 20-fold, suggesting that sensory ablation opened a sensory feedback loop during the stinging process. Next, using a complementary approach, the ultimate target of the stinger, namely the SupEG, was completely removed from the head capsule of cockroaches prior to the wasp’s sting. Indeed, as shown in Fig. 2C, this treatment significantly prolonged the duration of the Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 Fig. 2 The parasitoid Jewel Wasp uses mechanoreceptors to identify the brain of its cockroach host (A) The SupEG (top) and SEG (bottom) of a cockroach stung by a radiolabeled wasp (autoradiographs). Black staining indicates the presence of venom in and around the central complex of the SupEG and throughout the entire SEG. Adapted from Haspel et al. (2003). (B) The tip of the wasp’s stinger (scanning electron micrograph). Each of the three valves that comprise the stinger’s shaft possesses specialized sensilla (arrows). Adapted from Gal et al. (2014). (C) In a set of behavioral experiments, cockroach brains were either removed (No Brain) or replaced with soft or hard agarose pellets prior to a wasp’s sting. Whereas the duration of the head sting was significantly prolonged in ‘‘No Brain’’ and ‘‘Soft Agarose’’ cockroaches compared with ‘‘Control’’ cockroaches, it was not changed in ‘‘Hard Agarose’’ cockroaches. In addition, wasps injected venom inside Hard Agarose pellets but not inside Soft Agarose pellets. The duration of the wasp’s first sting in the cockroach’s thorax (wherein no neuronal tissue was manipulated) remained unchanged in all groups. Bars represent means SD, ***P50.001 (Kruskal–Wallis ANOVA). Adapted from Gal et al. (2014). Wasp ‘zombifies’ cockroach host Venom-induced grooming behavior and the involvement of dopamine Following recovery from the transient paralysis of the front legs, cockroaches stung by the Jewel Wasp groom almost continuously for 30 min (Fig. 1). The sting-evoked grooming is a complex behavior involving coordinated movements of different appendages (Fig. 3A) and exhibits all the components of normal grooming behavior. It occurs only when the wasp inflicts the head-sting and cannot be accounted for by the stress of the attack, contact with the wasp, or mechanical irritation (Fig. 3B), suggesting that the venom recruits central neural networks responsible for grooming (Weisel-Eichler et al. 1999). Studies of the sting-induced grooming suggest that monoaminergic systems within the cockroach’s CNS, and most prominently dopaminergic systems, are involved in activating this excessive grooming. For instance, an excessive grooming behavior that resembles the sting-induced grooming can be induced in un-stung cockroaches by injecting the monoaminereleasing drug reserpine into the SEG, by injecting dopamine (DA) into the hemolymph (Fig. 3B) or by injecting a D1 DA-receptor agonist into the SupEG (Fig. 3C) (Weisel-Eichler et al. 1999; Weisel-Eichler and Libersat 2002). On the other hand, injecting a DA-receptor antagonist [but not octopamine (OA)receptor antagonists] into the cockroach hemolymph prior to a wasp’s sting greatly reduced the venominduced excessive grooming (Weisel-Eichler et al. 1999) (Fig. 3D). Finally, injecting a DA-receptor agonist into the SupEG of stung cockroaches failed to evoke excessive grooming, suggesting a venominduced change in the DA-mediated signal-transduction cascade at, or downstream of, the DA-receptors (Weisel_Eichler and Libersat 2002). A DA-like substance identified in the wasp’s venom (Weisel-Eichler et al. 1999) is likely to be the component that induces this excessive grooming. Interestingly, D1 DAreceptor agonists have been shown to increase grooming behavior in the fruit fly Drosophila melanogaster (Yellman et al. 1997) as well as in mammals (Molloy and Waddington 1984), possibly indicating shared analogous mechanisms for this complex and coordinated behavior in evolutionary-distant organisms. A grooming-inducing venom cocktail has been reported only in the Jewel Wasp. However, is there an evolutionary advantage for the wasp in inducing this excessive grooming behavior, or is it simply a consequence, or a side-effect, of the venom injected into the head ganglia? Although this is obviously a hard question to answer, some alternative (but not necessarily mutually exclusive) possibilities come to mind. First, considering the wasp’s life cycle, a crucial function of the sting is the production of a long-lasting hypokinetic effect in the cockroach. For reasons yet to be determined, this effect commences only within Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 head-sting, indicating that sensory organs on the stinger are involved in identifying the cockroach’s SupEG during the stinging process. The same treatment did not affect the duration of the thoracic sting, as no neuronal tissue was removed from the cockroach’s thorax. What sensory modality does the wasp use to identify the cockroach’s SupEG? Possible candidates include mechanical, chemical, electrical, or behavioral cues associated with the presence of the SupEG inside the cockroach’s head capsule. When the SupEG of cockroaches was replaced with a small pellet of agarose prior to a wasp’s sting, the duration of stinging in normal wasps depended on the concentration (density) of the agarose (Gal et al. 2014); replacing the ganglion with a softer agarose pellet induced a significantly prolonged head-sting, whereas a harder agarose pellet induced a normal head-sting (Fig. 2C). In addition, a pH indicator mixed with these agarose pellets indicated that venom was injected into harder but not into softer pellets, suggesting that softer but not harder agarose pellets triggered the wasp’s sensory feedback necessary for injection of venom. These results demonstrate that mechanical cues are at least sufficient to induce a successful head-sting, whereas other experimental manipulations ruled out electrical, chemical, or behavioral cues (Gal et al. 2014). Indeed, the wasp’s stinger possesses several sensory organs (Fig. 2B), among which are campaniform sensilla that are known to work as mechanoreceptive strain gages measuring bending of the cuticle, which may mediate this process. Moreover, in vitro stimulation of the tip of the stinger with an agarose pellet increased the firing rate of afferent neurons ascending from the stinger to the wasp’s ventral nerve cord in a density-dependent manner, further strengthening the hypothesis that the wasp uses mechanoreceptors to differentiate the head ganglia from other tissues within the cockroach’s head capsule (Gal et al. 2014). Interestingly, the stinger also possesses contactchemoreceptive ‘‘dome-shaped sensilla;’’ however, the function of these sensilla is yet unknown. It is possible, for example, that these sensilla monitor the amount of injected venom or mediate the host’s acceptance, as was previously demonstrated in other parasitoid wasps (e.g., Nettles et al. 1982; Benedet et al. 2002). 5 6 F. Libersat and R. Gal 30 min after the sting (Fouad et al. 1994), during which the wasp searches for a suitable burrow to conceal the host (Fig. 1). Grooming is known to depress locomotion and increase the escape threshold of cockroaches (Camhi and Nolen 1981; Hoganwarburg et al. 1995) and thus the venominduced excessive grooming behavior, which lasts 30 min, may act to keep the cockroach in the stinging site and enable the wasp to easily locate its prey before transporting it to the nest. A similar phenomenon can be observed in cone snails, which use a combination of venom components that induce hyperactivity followed by flaccid paralysis (Olivera 1999). Apparently, this uncoordinated and frantic motor excitation immediately immobilizes the prey, such that it cannot escape its predator until the slower acting paralysis begins. The wasp may thus use a similar strategy. Another possibility is that the excessive grooming behavior acts to remove ectoparasitic and fungal infections from the cockroach’s cuticle, which may be harmful for the wasp’s egg or its developing larva (Nielsen et al. 2010; Zhukovskaya et al. 2013). Recently, using behavioral observations and chemical analyses, Herzner et al. (2013) showed that once the larva penetrates into the cockroach’s abdomen, it impregnates the cockroach from inside with large amounts of an oral secretion containing a blend of antimicrobials. This presumably provides an effective frontline defense against the unpredictable spectrum of pathogens with which the larva may be confronted during its development inside the cockroach host. The excessive grooming behavior may thus serve a similar purpose in the pre-penetration stage, wherein the egg and hatching larva are most vulnerable to cockroach-borne ectoparasites and pathogens. Mechanisms underlying the venom-induced hypokinesia Be the ultimate function of the excessive grooming behavior as it may, this behavior decays in the stung cockroach some 30 min following the head-sting. It is at around this time that the second and more prominent effect of the head-sting commences, wherein the cockroach ceases to initiate normal escape responses and spontaneous locomotion (Fouad et al. 1994; Libersat 2003). As the stung cockroach does not seem to have lost the ability to walk per se, this hypokinetic phase of envenomation has been metaphorically described as a ‘‘lethargic,’’ ‘‘depressive-like’’ or even ‘‘zombie-like’’ state wherein the cockroach appears to have lost its ‘‘will’’ or drive to self-initiate locomotion (Gal and Libersat 2010a). Indeed, considering the extremely welladapted escape system of the American cockroach (Nye and Ritzmann 1992; Schaefer et al. 1994), such specific non-paralytic manipulation of the cockroach behavior in favor of the wasp’s needs appears peculiar. It is interesting to note, however, that this unique state is, in principle, reversible. Although in nature the wasp’s larva kills the cockroach and begins pupating in its abdomen 6 days after the sting (Haspel et al. 2005), under experimental conditions (i.e., when the wasp is not allowed to lay an egg on the cockroach or when the egg or larva are Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 Fig. 3 The wasp induces grooming in its cockroach host through the dopaminergic system (A) A cockroach grooming its antenna and foreleg (arrows in top and bottom images, respectively) following a wasp’s sting. (B) Duration of grooming was significantly increased in cockroaches that received a full sting (head þ thorax) compared with cockroaches that received a thoracic sting only and had their SEG punctured by an experimenter (‘‘sham-sting’’). Dopamine (DA) injected into the hemolymph of an un-stung cockroach similarly increased duration of grooming. (C) When injected into the SupEG, a DA-receptor (DAR) agonist induced a dose-dependent increase in duration of grooming, with a maximal response at 10-8M. (D) Compared with a saline injection, a DAR antagonist but not an octopamine-receptor (OAR) antagonist, prevented venom-induced grooming when injected into the hemolymph prior to a wasp’s sting. Bars and data points represent means SD, **P50.01, ***P50.001 (t-test/ANOVA). Panels A, B, and D adapted from Weisel-Eichler et al. (1999). 7 Wasp ‘zombifies’ cockroach host removed by an experimenter) the cockroach regains normal behavioral activity between 3 and 7 days after the sting (Fouad et al. 1994; Gal and Libersat 2008). Sensory and motor aspects of the venom-induced hypokinesia Central aspects of the venom-induced hypokinesia The Jewel Wasp decreases the drive of its cockroach prey to initiate and maintain walking The hypokinetic state is specific to walking-related behaviors, but does the venom completely prevent the initiation of walking or does it elevate the threshold stimulus required to elicit walking? A recent study (Gal and Libersat 2008) provides answers to some of these questions. Periplaneta are terrestrial insects, and therefore immersion in water provides a reliable and continuous stressful stimulus that typically produces a rigorous walking-like motor pattern (Cocatrezilgien and Delcomyn 1990). Hence, Gal and Libersat (2008) quantified the walking-like behavior of cockroaches immersed in a water-filled container, in a setup similar to the Forced Swim Test paradigm that is traditionally used to evaluate depression-like phenotypes in non-human mammals (McArthur and Borsini 2006). Although stung cockroaches readily initiated normal and coordinated walking-like motor patterns upon immersion (Fig. 4C), their duration of swimming was dramatically lower compared with un-stung cockroaches (Fig. 4D). In fact, stung cockroaches tended to float passively on the water, occasionally moving their antennae about in an exploratory manner. Hence, in addition to increasing the threshold for the initiation of walking-related behaviors, the venom appears to also affect the ability Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 American cockroaches are known for their highly effective escape responses (Camhi 1988). For instance, when a predator approaches the cockroach, wind-sensitive hairs on the cerci (two appendages at the end of the abdomen) detect changes in air pressure resulting from the approaching predator, rapidly convey this information through giant interneurons (GIs) to the thoracic and head ganglia, and induce a fast and reliable escape response in the opposite direction (Camhi 1988). Similarly, through the large descending mechanosensory interneurons (DMIs) reaching from the antennae to the head-ganglia and thoracic ganglia, a tactile stimulus to the antenna also induces a reliable and rapid escape response (Ye and Comer 1996). Both these stimuli, however, consistently fail to evoke such an escape response in a stung cockroach (Fouad et al. 1996). As this behavioral manipulation occurs only after the venom is injected into the cockroach’s head, it cannot directly affect the escape response at the level of the leg muscles or neuromuscular junctions. Instead, it could result from the venom’s effect on sensory, central, or premotor neurons. As a first step, therefore, the venom’s effect on sensory and premotor systems was tested. Studies of the sensory systems in stung cockroaches revealed that sensory cues reliably propagate along sensory neurons and interneurons, e.g. the GIs from the cerci or the DMIs from the antennae (Fouad et al. 1994, 1996). Similarly, the muscles and motor neurons are functional in stung cockroaches and are able to produce normal motor outputs (Fouad et al. 1994, 1996; Gal and Libersat 2008). For instance, the rigorous escape response evoked in normal cockroaches by tactile or wind stimuli are characterized by the rhythmic recruitment of slow and fast leg depressor and extensor muscles acting in coordination (Libersat et al. 1999; Libersat 2003). As these muscles are innervated by specific sets of thoracic motor neurons, electromyographic (EMG) recordings from these muscles, for example, through electrodes implanted in the coxal depressor muscles of behaving cockroaches (Fig. 4A), provide a direct readout of both the slow and fast coxal depressors (Ds and Df, respectively) as well as of their associated motor neurons (Pearson and Iles 1971). Such EMG recordings in stung cockroaches reveal that, although a wind stimulus to the cerci evokes neuronal activity that propagates normally to the CNS, it evokes only a short burst of Ds potentials and no Df potentials (Fig. 4B). Behaviorally, this is associated with a postural reflex (Ds activity) without the typical successive running response that is otherwise normally associated with a rapid phasic Df activity (Libersat et al. 1999). Nevertheless, both Ds and Df potentials can be rhythmically evoked in a walking-like manner when stung cockroaches are immersed in water (Gal and Libersat 2008) (Fig. 4C) and Df potentials can be evoked normally when suspending stung cockroaches in the air and initiating flying behavior, which involves Df activity (Libersat 2003). Hence, the venom must affect central, rather than peripheral, mechanisms to decrease locomotion and induce the ‘‘zombie-like’’ state. In addition, these results imply that the venom does not affect all motor outputs similarly but rather that some aspects of behavior, namely the initiation and/or maintenance of walking-related behaviors, are specifically impaired in stung cockroaches while other motor outputs are spared. 8 F. Libersat and R. Gal Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 Fig. 4 The motor behavior of cockroaches during the ‘‘zombie-like’’ state induced by the wasp’s sting (A) A diagram indicating positioning of EMG electrodes (black circles). Numbers represent leg pairs (1, 2, and 3 for the prothoracic, mesothroacic, and metathoracic legs, respectively) and letters represent lateralization (L: left, R: right). (B) EMG recordings from leg R3 in one control and one stung cockroach standing on a solid surface. A wind stimulus applied to the cerci (bottom line in each trace) evoked rhythmic fast and slow potentials (Df and Ds, respectively) in the depressor muscle of the coxa of a control cockroach, but only a burst of Ds potentials (associated with a postural change without displacement) in a stung cockroach. Voltage scale is different for the top and bottom traces. Adapted from Libersat (2003). (C) EMG recordings from legs R2, L3, and R3, and respective phase histograms, in one control and one stung cockroach immersed in water. In both the control and the stung cockroach, a normal interleg coordination can be observed during active bouts of swimming, such that legs R2 and L3 move in together (black bars in histograms) and alternate with leg R3 (gray bars in histogram). Adapted from Gal and Libersat (2008). (D) Swimming trajectory (left) of one control and one stung cockroach, and cumulative duration of swimming in a modified 60-s ‘‘forced swim test’’. Arrows and asterisks indicate location at the beginning and end of the test, respectively. Bold lines indicate the spatial location during which periods of immobility were observed. Bars represent means SD, ***P50.001 (t-test). Adapted from Gal and Libersat (2008). 9 Wasp ‘zombifies’ cockroach host The Jewel Wasp decreases ongoing and evoked neuronal activity in the cockroach’s SEG The Jewel Wasp inserts its stinger through the cockroach’s soft neck cuticle in close proximity to the location of the SEG, and venom is found in large quantities specifically inside the SEG and SupEG of stung cockroaches (Haspel et al. 2003). Both these head ganglia are generally known to exert a ‘‘top– down’’ regulation of the thoracic Central pattern generators (CPGs), which in turn produce the local spatiotemporal patterns required for locomotion (Altman and Kien 1987; Strausfeld 1999; Strauss 2002; Gal and Libersat 2006). It is thus probable that neuronal ensembles within either one, or both, of these ganglia determine the ‘‘internal state’’ of the cockroach, and thus its ‘‘motivation’’ to engage in specific motor patterns such as walking. Compared with un-stung cockroaches, spontaneous and stimulus-evoked neuronal spiking activity in the core of the SEG was found to be dramatically decreased in stung cockroaches (Gal and Libersat 2010b) (Fig. 5A and B), concomitant with the decrease in spontaneous and stimulus-evoked locomotion. This observation suggests that the SEG may be an important part of the decision-making system and may therefore play a central role in determining the ‘‘rest state’’ and the drive for walking in cockroaches. Indeed, in cockroaches, the SEG is known generally to exert a tonic permissive effect on walking-related thoracic CPGs (Gal and Libersat 2006). Moreover, intracellular recordings from neurons in the SEG and SupEG of locusts indicate that the spontaneous initiation of walking is accompanied by changes in the firing pattern of several neurons descending from both the SEG and SupEG. However, while SEG and SupEG interneurons both fire during walking, and are thus both involved in maintaining walking behavior, predominantly SEG interneurons fire during the preparatory phase of walking. These observations suggest a prime role for SEG neuronal circuits in determining the motivational level of insects to engage in walking (Kien and Altman 1992). Indeed, some evidence also suggests that a venom-induced decrease in SEG neuronal activity may be involved in the ‘‘apathetic’’ state induced in crickets by the parasitoid wasp Liris niger (Ferber et al. 2001). The exact role of the SupEG in the venominduced manipulation of the cockroach motor behavior is currently under investigation. Several possibilities come to mind, for example, a role in evoking the excessive grooming behavior (Libersat 2003) or a role in the venom-induced changes in cockroach metabolism (Haspel et al. 2005). Because the SupEG is known to regulate motor behavior in insects (Roeder 1937; Ridgel and Ritzmann 2005; Gal and Libersat 2006), it is also possible that the SupEG, in concert with the SEG, plays a role in inducing certain aspects of venom-induced hypokinesia. For instance, the venom could affect the SupEG directly by affecting specific circuitries in this ganglion, or indirectly by affecting ascending SEG interneurons which, in turn, modulate SupEG circuitries that control motor behavior. Several recent studies highlight the role of a specific region within the SupEG, namely the CC, in mediating numerous aspects of walking behavior (Bender et al. 2010; Harley and Ritzmann 2010; Guo and Ritzmann 2013). In fact, the CC of arthropods shares numerous homologies with the basal ganglia of vertebrates, implicating its role in the regulation and release of adaptive behaviors (Strausfeld and Hirth 2013). Most of the venom found in the SupEG of stung cockroaches is, indeed, concentrated in and around the CC (Haspel et al. 2003). Involvement of neuromodulatory systems in venom-induced hypokinesia Prime candidates for the hypokinetic manipulation are neuromodulatory interneurons, and in particular monoaminergic interneurons ascending from the SEG to the SupEG. With respect to motivation, the role of monoaminergic systems may have been conserved throughout evolution. In the CNS of insects and mammals, DA and noradrenaline (or its invertebrate analog, OA) have been shown to profoundly affect motivation, arousal, and locomotion (Libersat and Pflueger 2004). Moreover, in insects as in mammals, it is the interplay between different neuromodulators that seems to affect behavioral choices. Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 of the cockroach to maintain such behaviors over a long period of time. This, in turn, suggests that stung cockroaches have a specific deficit in reaching the decision to walk, or in their ‘‘drive’’ to initiate and maintain walking, rather than in their ability to walk per se. Understanding the mechanism underlying such a manipulation may be beneficial in the investigation of key questions in the study of the neurobiology of behavior (Gal and Libersat 2010a), such as which circuits determine the internal state of the insect and regulate the decision to engage in a specific motor behavior, and what are the neuronal substrates responsible for determining the ‘‘motivation’’ specifically for walking? Although these questions are at the core of current ongoing research, some answers have already begun to emerge owing to the study of this unique parasitoid–host system. 10 F. Libersat and R. Gal In the SEG, one specific population of monoaminergic neurons that may be affected by the venom is the octopaminergic unpaired median neurons. The axons of some of these neurons innervate segmental ganglia while others innervate major neuropiles in the SupEG (Braunig and Burrows 2004). Similar to noradrenaline in mammals, OA in insects has been previously implicated in regulating locomotion (Sombati and Hoyle 1984; Bacon et al. 1995; Pfluger and Duch 2000; Saraswati et al. 2004) and activity in SEG-OA neurons in Manduca larvae has been correlated with fictive locomotion (Cholewa Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 Fig. 5 Possible targets of the wasp’s venom (A) Representative traces of extracellular neuronal activity in the SEG of one control and one stung cockroach. Adapted from Gal and Libersat (2010b). (B) Spontaneous neuronal activity in the SEG of stung cockroaches is significantly lower than in control cockroaches. Bars represent means SEM, **P50.05 (t-test). Adapted from Gal and Libersat (2010b). (C) Injection of an octopamine receptor (OAR) agonist into the SupEG increased spontaneous walking both in un-stung and stung cockroaches. Adapted from Rosenberg et al. (2007). Bars represent means SD, ***P50.001 (ANOVA). (D) The foot-shock voltage required to elicit an escape response, namely a ‘‘startle’’ response followed by locomotion, was reversibly increased following the sting. Bars represent means SD, different letters represent statistically significant differences (P50.001, Mann–Whitney Rank Sum Test). Adapted from Gal and Libersat (2008). (E) The ‘‘startle’’ threshold was similarly increased in stung cockroaches and in un-stung cockroaches treated with an opioid receptor (OpR) agonist. Bars represent means SD. Adapted from Gavra and Libersat (2011). 11 Wasp ‘zombifies’ cockroach host escaping the electric foot-shock (Gal and Libersat 2008) (Fig. 5D). This indicates that not only is the threshold for initiation of walking elevated in stung cockroaches, but also that their basic ability to recruit walking-related CPGs in a coordinated manner upon reaching this threshold is preserved. In search for a candidate neuromodulator to account for this change in the threshold for initiation of startle, Gavra and Libersat (2011) showed that injections of the opioid agonist loperamide induce a significant and dose-dependent increase in the avoidance threshold of cockroaches in a shuttle box assay (Fig. 5E). Moreover, in the cockroach ‘‘antenna-heart’’ preparation [a well-known preparation for testing the pharmacology of peptides in insects, and which contains opioid fibers in cockroaches (Gavra and Libersat 2011)], application of crude venom completely inhibited contractions of the myogenic dilator muscle (Gavra and Libersat 2011). Application of the opioid antagonist naloxone (which, by itself, has no effect on muscle contractions) to the preparation partly reversed this effect by normalizing the amplitude of the contractions but not their frequency. Whether an opiate agonist in the venom is directly responsible for the change in escape threshold to noxious stimulus remains to be proven. Conclusions and future directions In this article we unfold the mechanisms by which a parasitoid wasp ‘‘hijacks’’ the CNS of its cockroach host to control the host’s behavior. In a process involving mechanosensory inputs arriving from its stinger, the Jewel Wasp identifies the cockroach’s brain inside the head capsule, penetrates through its ganglionic sheath (the insect’s blood–brain– barrier) and injects a venom cocktail directly inside the two ganglia of the head. This ‘‘brain-sting’’ modulates specific motor behaviors in the host, which first grooms excessively and then shows a dramatic decrease in the drive to self-initiate locomotion. Unlike motivational deficits in mammals, however, the venom-induced hypokinesia can be traced to specific neuronal structures within the CNS of stung cockroaches; namely, it seems that the venom modulates neuronal circuitries within (at least) the cockroach’s SEG and/or CC in the SupEG, which may therefore be involved in determining the ‘‘motivation’’ of the insect to engage in spontaneous and evoked walking. Although some candidates have been suggested, the specific neuronal ensembles that are manipulated by the venom have not been identified yet. Given the Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 and Pfluger 2009), further highlighting these neurons as major candidates for the venom-induced hypokinesia. Accordingly, the OA receptor agonist chlordimeform has been shown to induce a significant increase in spontaneous walking when injected into the SupEG of either stung or un-stung cockroaches (Rosenberg et al. 2007) (Fig. 5C). This correlates well with the decrease in spontaneous neuronal spiking activity in the core of the SEG in stung cockroaches (as discussed above; Gal and Libersat 2010b), as OA neurons are characterized by a baseline rate of firing (Rosenberg et al. 2006). Thus, the Jewel Wasp’s venom may interfere with octopaminergic modulation of the initiation of walking in central structures of the cockroach SupEG, which receive inputs from the SEG. A similar role for OA in depressing host’s locomotion following a wasp’s sting has been also proposed for the wasp Cotesia, a parasitoid of Manduca (Adamo and Shoemaker 2000). Notably, numerous studies have shown a dense innervation of OA-immunoreactive neurons within the CC of insects (Sinakevitch et al. 2005, 2011; Busch et al. 2009; Homberg et al. 2013); as mentioned above, most venom injected by the Jewel Wasp inside the cockroach’s SupEG is found in and around the CC (Haspel et al. 2003). Another possible target of the wasp’s venom is the opioid system, which is known to affect responsiveness to stimuli not only in mammals but also in insects (Ford et al. 1986; Gritsai et al. 2004). Similar to the effect of the Jewel Wasp’s venom, opioid-like substances are known to have an ‘‘analgesic’’-like effect in insects and therefore modulate the insects’ threshold for escape (Stefano et al. 1990). Further, in fruit flies, enkephalin-like immunoreactive neurons are found in the CC and SEG (Thorpe and Duve 1990), both implicated in the control of walking in insects (Altman and Kien 1987; Strausfeld 1999; Strauss 2002; Gal and Libersat 2006). To test the threshold for the initiation of startle in stung cockroaches, a modified shuttle box was used in which escapable foot shocks were administered to cockroaches’ legs without previous training. The voltage required to reliably elicit an escape response, which consists of a ‘‘startle’’ (‘‘nociceptive’’) response followed by a rigorous and rhythmic recruitment of the motor neurons and muscles associated with walking in thoracic ganglion circuits, was noted at different time-points after the sting. Compared with sham-treated cockroaches, this threshold voltage gradually increased in stung cockroaches such that, at the peak of the venom’s effect (2–4 h after the sting), stung cockroaches endured voltages 48-fold higher before 12 Acknowledgments All experiments presented in this review comply with Principles of Animal Care, NIH publication no. 86-23, revised in 1985, and also with the current laws of the State of Israel. We thank Gal Haspel for his wonderful illustrations of the venom’s behavioral effects, and Dieter Schulten from the Aquazoo Löbbecke Museum (Düsseldorf, Germany) for his kind gift of wasps. Funding This work is currently supported by The Israel Academy of Sciences and Humanities (1181/11). References Adamo SA, Shoemaker KL. 2000. Effects of parasitism on the octopamine content of the central nervous system of Manduca sexta: a possible mechanism underlying host behavioural change. Can J Zool 78:1580–7. Altman J, Kien J. 1987. Functional organization of the subesophageal ganglion in arthropods. In: Gupta AP, editor. Arthropod brain; its evolution, development, structure, and functions. New York: Wiley. p. 265–301. Bacon JP, Thompson KS, Stern M. 1995. Identified octopaminergic neurons provide an arousal mechanism in the locust brain. J Neurophysiol 74:2739–43. Bender JA, Pollack AJ, Ritzmann RE. 2010. Neural activity in the central complex of the insect brain is linked to locomotor changes. Curr Biol 20:921–6. Benedet F, Leroy T, Gauthier N, Thibaudeau C, Thibout E, Renault S. 2002. Gustatory sensilla sensitive to protein kairomones trigger host acceptance by an endoparasitoid. P Roy Soc B-Biol Sci 269:1879–86. Braunig P, Burrows M. 2004. Projection patterns of posterior dorsal unpaired median neurons of the locust subesophageal ganglion. J Comp Neurol 478:164–75. Busch S, Selcho M, Ito K, Tanimoto H. 2009. A map of octopaminergic neurons in the Drosophila brain. J Comp Neurol 513:643–67. Camhi JM. 1988. Escape behavior in the cockroach - distributed neural processing. Experientia 44:401–8. Camhi JM, Nolen TG. 1981. Properties of the escape system of cockroaches during walking. J Comp Physiol 142:339–46. Cholewa J, Pfluger HJ. 2009. Descending unpaired median neurons with bilaterally symmetrical axons in the suboesophageal ganglion of Manduca sexta larvae. Zoology 112:251–62. Cocatrezilgien JH, Delcomyn F. 1990. Fast axon activity and the motor pattern in cockroach legs during swimming. Physiol Entomol 15:385–92. Eberhard WG. 2000. Spider manipulation by a wasp larva. Nature 406:255–6. Ferber M, Horner M, Cepok S, Gnatzy W. 2001. Digger wasp versus cricket: mechanisms underlying the total paralysis caused by the predator’s venom. J Neurobiol 47:207–22. Ford R, Jackson DM, Tetrault L, Torres JC, Assanah P, Harper J, Leung MK, Stefano GB. 1986. A behavioral role for enkephalins in regulating locomotor-activity in the insect Leucophaea-Maderae - evidence for high-affinity kappa-like opioid binding-sites. Comp Biochem Phys C 85:61–6. Fouad K, Libersat F, Rathmayer W. 1994. The venom of the cockroach-hunting wasp Ampulex-Compressa changes motor thresholds - a novel tool for studying the neural control of arousal. Zoolog Anal Complex Syst 98:23–34. Fouad K, Libersat F, Rathmayer W. 1996. Neuromodulation of the escape behavior of the cockroach Periplaneta americana by the venom of the parasitic wasp Ampulex compressa. J Comp Physiol A-Sens Neural Behav Physiol 178:91–100. Gal R, Libersat F. 2006. New vistas on the initiation and maintenance of insect motor behaviors revealed by specific lesions of the head ganglia. J Comp Physiol A 192:1003–20. Gal R, Libersat F. 2008. A parasitoid wasp manipulates the drive for walking of its cockroach prey. Curr Biol 18:877–82. Gal R, Libersat F. 2010a. On predatory wasps and zombie cockroaches: Investigations of ‘‘free will’’ and spontaneous behavior in insects. Commun Integr Biol 3:458–61. Gal R, Libersat F. 2010b. A wasp manipulates neuronal activity in the sub-esophageal ganglion to decrease the drive for walking in its cockroach prey. PLoS One 5:e10019. Gal R, Kaiser M, Haspel G, Libersat F. 2014. Sensory arsenal on the stinger of the parasitoid jewel wasp and its possible role in identifying cockroach brains. PLoS One 9: e89683. Gavra T, Libersat F. 2011. Involvement of the opioid system in the hypokinetic state induced in cockroaches by a parasitoid wasp. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 197:279–91. Gritsai OB, Dubynin VA, Pilipenko VE, Petrov OP. 2004. Effects of peptide and non-peptide opioids on protective reaction of the cockroach Periplaneta americana in the ‘‘hot camera’’. J Evol Biochem Phys 40:153–60. Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 rich content of chemical compounds in the venom (including monoamines, amino acids, peptides, and proteins) it appears plausible that several neuromodulatory systems, such as the octopaminergic and enkephalinergic ones, are targeted. Post-genomic approaches may be used to answer this question, as advances in this field have substantially increased our understanding of the proximate mechanisms mediating hosts’ behavioral changes. For instance, hosts’ brain proteins can be extracted and their mass and amino acid sequence determined via mass-spectrophotometry and aligned against available databases. Given the time scale of the cockroach’s manipulation by the Jewel Wasp, the venom may have genomic effects on the cockroach’s CNS. Proteomics techniques can thus help identify the molecular targets of different venom components within the cockroach’s brain. These lines of investigation hopefully will provide useful information regarding the neuronal underpinnings of behavioral spontaneity, which is a prime issue in behavioral neurobiology. F. Libersat and R. Gal Wasp ‘zombifies’ cockroach host Moore EL, Haspel G, Libersat F, Adams ME. 2006. Parasitoid wasp sting: a cocktail of GABA, taurine, and beta-alanine opens chloride channels for central synaptic block and transient paralysis of a cockroach host. J Neurobiol 66:811–20. Nettles WC, Morrison RK, Xie ZN, Ball D, Shenkir CA, Vinson SB. 1982. Synergistic action of KCl and magnesium-sulfate on parasitoid wasp oviposition. Science 218:164–6. Nielsen C, Agrawal AA, Hajek AE. 2010. Ants defend aphids against lethal disease. Biology Lett 6:205–8. Nye SW, Ritzmann RE. 1992. Motion analysis of leg joints associated with escape turns of the cockroach, Periplaneta americana. J Comp Physiol A Sens Neural Behav Physiol 171:183–94. O’Neill KM. 2001. Solitary wasps: behavior and natural history. Ithaca, N.Y: Comstock Pub. Associates. Olivera BM. 1999. Conus venom peptides: correlating chemistry and behavior. J Comp Physiol A Sens Neural Behav Physiol 185:353–9. Pearson KG, Iles JF. 1971. Innervation of coxal depressor muscles in the cockroach, Periplaneta americana. J Exp Biol 54:215–32. Pfluger HJ, Duch C. 2000. The functional role of octopaminergic neurons in insect motor behavior. Acta Biol Hung 51:343–8. Quicke DLJ. 1997. Parasitic wasps. 1st edition. London, New York: Chapman & Hall. Ridgel AL, Ritzmann RE. 2005. Effects of neck and circumoesophageal connective lesions on posture and locomotion in the cockroach. J Comp Physiol A Neuroethol, Sens Neural Behav Physiol 191:559–73. Roeder KD. 1937. The control of tonus and locomotor activity in the praying mantis (Mantis religiosa L.). J Exp Zool 76:353–74. Rosenberg LA, Glusman JG, Libersat F. 2007. Octopamine partially restores walking in hypokinetic cockroaches stung by the parasitoid wasp Ampulex compressa. J Exp Biol 210:4411–7. Rosenberg LA, Pfluger HJ, Wegener G, Libersat F. 2006. Wasp venom injected into the prey’s brain modulates thoracic identified monoaminergic neurons. J Neurobiol 66:155–68. Saraswati S, Fox LE, Soll DR, Wu CF. 2004. Tyramine and octopamine have opposite effects on the locomotion of Drosophila larvae. J Neurobiol 58:425–41. Schaefer PL, Kondagunta GV, Ritzmann RE. 1994. Motion analysis of escape movements evoked by tactile stimulation in the cockroach Periplaneta americana. J Exp Biol 190:287–94. Sinakevitch I, Mustard JA, Smith BH. 2011. Distribution of the octopamine receptor AmOA1 in the honey bee brain. PLoS One 6:e14536. Sinakevitch I, Niwa M, Strausfeld NJ. 2005. Octopamine-like immunoreactivity in the honey bee and cockroach: comparable organization in the brain and subesophageal ganglion. J Comp Neurol 488:233–54. Sombati S, Hoyle G. 1984. Central nervous sensitization and dishabituation of reflex action in an insect by the neuromodulator octopamine. J Neurobiol 15:455–80. Stefano G, Scharrer B, Lung M. 1990. Neurobiology of opioids in Leucophaea maderae. In: Huber I, Masler EP, Rao BR, editors. Cockroaches as models for Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 Grosman AH, Janssen A, de Brito EF, Cordeiro EG, Colares F, Fonseca JO, Lima ER, Pallini A, Sabelis MW. 2008. Parasitoid increases survival of its pupae by inducing hosts to fight predators. PLos One 3:e2276. Guo PY, Ritzmann RE. 2013. Neural activity in the central complex of the cockroach brain is linked to turning behaviors. J Exp Biol 216:992–1002. Harley CM, Ritzmann RE. 2010. Electrolytic lesions within central complex neuropils of the cockroach brain affect negotiation of barriers. J Exp Biol 213:2851–64. Haspel G, Gefen E, Ar A, Glusman JG, Libersat F. 2005. Parasitoid wasp affects metabolism of cockroach host to favor food preservation for its offspring. J Comp Physiol A 191:529–34. Haspel G, Rosenberg LA, Libersat F. 2003. Direct injection of venom by a predatory wasp into cockroach brain. J Neurobiol 56:287–92. Herzner G, Schlecht A, Dollhofer V, Parzefall C, Harrar K, Kreuzer A, Pilsl L, Ruther J. 2013. Larvae of the parasitoid wasp Ampulex compressa sanitize their host, the American cockroach, with a blend of antimicrobials. Proc Natl Acad Sci USA 110:1369–74. Hoganwarburg AJ, Hogan JA, Ashton MC. 1995. Locomotion and grooming in crickets - competition or time-sharing. Anim Behav 49:531–3. Homberg U, Seyfarth J, Binkle U, Monastirioti M, Alkema MJ. 2013. Identification of distinct tyraminergic and octopaminergic neurons innervating the central complex of the desert locust, Schistocerca gregaria. J Comp Neurol 521:2025–41. Kien J, Altman JS. 1992. Preparation and execution of movement: parallels between insect and mammalian motor systems. Comp Biochem Physiol Comp Physiol 103:15–24. LeRalec A, Rabasse JM, Wajnberg E. 1996. Comparative morphology of the ovipositor of some parasitic hymenoptera in relation to characteristics of their hosts. Can Entomol 128:413–33. Libersat F. 2003. Wasp uses venom cocktail to manipulate the behavior of its cockroach prey. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 189:497–508. Libersat F, Delago A, Gal R. 2009. Manipulation of host behavior by parasitic insects and insect parasites. Annu Rev Entomol 54:189–207. Libersat F, Gal R. 2007. Neuro-manipulation of hosts by parasitoid wasps. In: Rivers D, Yoder J, editors. Recent advances in the biochemistry, toxicity, and mode of action of parasitic wasp venoms. Trivandrum: Research Signpost. p. 93–114. Libersat F, Haspel G, Casagrand J, Fouad K. 1999. Localization of the site of effect of a wasp’s venom in the cockroach escape circuitry. J Comp Physiol A Sens Neural Behav Physiol 184:333–45. Libersat F, Pflueger HJ. 2004. Monoamines and the orchestration of behavior. Bioscience 54:17–25. McArthur R, Borsini F. 2006. Animal models of depression in drug discovery: A historical perspective. Pharmacol Biochem Be 84:436–52. Molloy AG, Waddington JL. 1984. Dopaminergic behavior stereospecifically promoted by the d1 agonist R-Sk-andF-38393 and selectively blocked by the D1 antagonist Sch-23390. Psychopharmacology 82:409–10. 13 14 ovipositor apparatus and associated structures in the Orussidae (Hymenoptera, Insecta). Zoomorphology 121:63–84. Weisel-Eichler A, Libersat F. 2002. Are monoaminergic systems involved in the lethargy induced by a parasitoid wasp in the cockroach prey? J Comp Physiol A 188:315–24. Weisel-Eichler A, Haspel G, Libersat F. 1999. Venom of a parasitoid wasp induces prolonged grooming in the cockroach. J Exp Biol 202(Pt 8):957–64. Ye SP, Comer CM. 1996. Correspondence of escape-turning behavior with activity of descending mechanosensory interneurons in the cockroach, Periplaneta americana. J Neurosci 16:5844–53. Yellman C, Tao H, He B, Hirsh J. 1997. Conserved and sexually dimorphic behavioral responses to biogenic amines in decapitated Drosophila. Proc Natl Acad Sci USA 94:4131–6. Zhukovskaya M, Yanagawa A, Forschler BT. 2013. Grooming behavior as a mechanism of insect disease defense. Insects 4:609–30. Downloaded from http://icb.oxfordjournals.org/ at Ben Gurion University - Aranne Library on April 5, 2014 neurobiology: applications in biomedical research. Boca Raton, FL: CRC Press. p. 285–302. Strausfeld NJ. 1999. A brain region in insects that supervises walking. Prog Brain Res 123:273–84. Strausfeld NJ, Hirth F. 2013. Deep homology of arthropod central complex and vertebrate basal ganglia. Science 340:157–61. Strauss R. 2002. The central complex and the genetic dissection of locomotor behaviour. Curr Opin Neurobiol 12:633–8. Thorpe A, Duve H. 1990. Morphological, biochemical, and physiological-studies on insect enkephalins. Prog Clin Biol Res 342:293–9. Vilhelmsen L. 2000. The ovipositor apparatus of basal Hymenoptera (Insecta): phylogenetic implications and functional morphology. Zool Scr 29:319–45. Vilhelmsen L, Isidoro N, Romani R, Basibuyuk HH, Quicke DLJ. 2001. Host location and oviposition in a basal group of parasitic wasps: the subgenual organ, F. Libersat and R. Gal