* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lecture 03, NEW - terpenes + polyketides
Microbial metabolism wikipedia , lookup
Photosynthesis wikipedia , lookup
Peptide synthesis wikipedia , lookup
Mitogen-activated protein kinase wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Genetic code wikipedia , lookup
Paracrine signalling wikipedia , lookup
Citric acid cycle wikipedia , lookup
Proteolysis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Metabolomics wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biochemical cascade wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Natural product wikipedia , lookup
Biochemistry wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Isotopic labeling wikipedia , lookup
Amino acid synthesis wikipedia , lookup
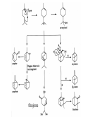
(photosynthesis)
Polysaccharides
Glycosides
Nucleic Acids
phosphoenol
pyruvate
Shikimate pathway
Aromatic Compounds
Lignans
Shikimate
pyruvate
acetyl
CoA CH3COSCoA
aromatic amino acids
aliphatic amino acids
CITRIC
ACID
CYCLE
Alkaloids
Peptides
Penicillins
Cyclic Peptides
CH3COSCoA
-O
2CCH2COSCoA
CH3COCH2COSCoA
CH3COSCoA
CH3COSCoA
(3)
Isoprenoids (terpenes,
steroids, carotenoids)
mevalonate
Polyketides, Fatty Acids
Prostaglandins, Macrocyclic Antibiotics
(3) Isoprene pathway: Terpenes
(3)
Mevalonate
Terpenes, Steroids
-- made from 5-carbon units
- C6 compound
that loses CO2
to form C5 units
phytane (C20)
a-pinene (C10)
cholesterol
(missing 3 C’s)
Isoprene Biosynthesis
decarboxylates
DMAP
to yield IPP
IPP
- Result: two isomeric 5-carbon molecules, IPP + DMAP
- Pyrophosphates: high-energy group powers biosynthetic rxns..
“nature’s leaving group”
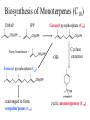
Biosynthesis of Monoterpenes (C10)
DMAP
IPP
Geranyl pyrophosphate (C10)
Prenyl transferase +
-OR-
Cyclase
enzymes
Farnesyl pyrophosphate (C15)
rearranged to form
sesquiterpenes (C15)
cyclic monoterpenes (C10)
Biosynthesis of Monoterpenes (C10)
(1) DMAP ionizes to form
electrophilic carbocation
(2) Nucleophilic attack by
IPP forms geranyl-PP
(3) Stereospecific loss of
HR, forming double bond
(4) Geranyl-PP ionizes,
rearranges to form a
carbocation intermediate
- Cyclic monoterpenes then form via enzyme-catalyzed
stereospecific rearrangements, functionalizations
thujone
Most cyclic monoterpenes have a distinctive odor
- basis of perfume & flavor industries
Stereoisomers have different characteristic smells
- demonstrates that smell receptors are 3D proteins,
i.e. chiral environments that can distinguish enantiomers
(+)-carvone = caraway seed
(-)-carvone = spearmint
(+)-limonene = oranges
(-)- limonene = lemons
Biosynthesis of Sesquiterpenes (C15)
DMAP
IPP
Prenyl transferase +
Farnesyl pyrophosphate (C15)
rearranged to form
sesquiterpenes (C15)
Geranyl pyrophosphate (C10)
Biosynthesis of Sesquiterpenes (C15)
DMAP
IPP
Geranyl pyrophosphate (C10)
Prenyl transferase +
Farnesyl pyrophosphate (C15)
rearranged to form
sesquiterpenes (C15)
- if you introduce a
labeled carbon in the
precursor, you can see
where it ends up in the
final natural product
Biosynthesis of Sesquiterpenes (C15)
DMAP
IPP
Geranyl pyrophosphate (C10)
Prenyl transferase +
Farnesyl pyrophosphate (C15)
rearranged to form
sesquiterpenes (C15)
- if you introduce a
labeled carbon in the
precursor, you can see
where it ends up in the
final natural product
Labeling studies in Biosynthesis 1
Using isotopically enriched precursors, it is possible to trace
the incorporation of building blocks into complex metabolites
- follow the label and see which atoms in the natural
product end up labeled
Originally done with radioactive labels (14C, 3H)
- feed cells a precursor with one 14C
- use chemical degradation to break natural product apart,
see which pieces contain the radioactivity
- drawback: sloppy, usually not possible to completely
determine biosynthetic route
Labeling studies in Biosynthesis 2
Now done by NMR using non-radioactive isotope 13C
- common carbon isotope 12C is invisible to NMR
- rare 13C has nuclear spin I = n/2, so is detectable by NMR
- feed cells a precursor with one position 13C labeled;
then see which positions show exaggerated peaks in the
carbon NMR spectrum
since only 1% of carbons are naturally 13C, the positions
that incorporate the label will give much higher peaks
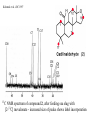
Kubanek et al. JOC 1997
13C
NMR spectrum of compound 2, after feeding sea slug with
[2-13C] mevalonate - increased size of peaks shows label incorporation
- allowed researchers to
infer biosynthetic pathway
for these related C-23 and
C-21 terpenes
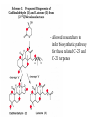
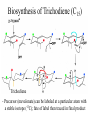
Biosynthesis of Trichodiene (C15)
Trichodiene
- Precursor (mevalonate) can be labeled at a particular atom with
a stable isotope (13C); fate of label then traced in final product
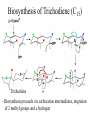
Biosynthesis of Trichodiene (C15)
Trichodiene
- Biosynthesis proceeds via carbocation intermediates, migration
of 2 methyl groups and a hydrogen
Trichodiene
Trichothecenes
Trichodiene is a key precursor of fungal metabolites called
trichothecenes, a major human health problem
- fungal toxins are called mycotoxins
- often present in moldy grain consumed by cattle or people
Winter 1942: thousands of Russians died after eating grains
left in the field over winter, due to the war; grain was infected
with the trichothecene-producing fungus
1981: USA accused Russia of spraying fungal spores in Vietnam,
resulting in foliage contaminated with trichothecenes (false)
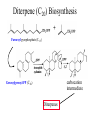
Diterpene (C20) Biosynthesis
+
Farnesyl pyrophosphate (C15)
carbocation
intermediate
Gerenylgerenyl-PP (C20)
Diterpenes
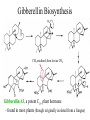
Gibberellin Biosynthesis
CH3 oxidized, then lost as CO2
Gibberellin A3, a potent C19 plant hormone
- found in most plants (though originally isolated from a fungus)
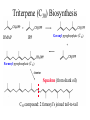
Triterpene (C30) Biosynthesis
DMAP
IPP
Geranyl pyrophosphate (C10)
Farnesyl pyrophosphate (C15)
Squalene (from shark oil)
C30 compound: 2 farnesyl’s joined tail-to-tail
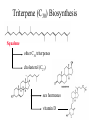
Triterpene (C30) Biosynthesis
Squalene
other C30 triterpenes
cholesterol (C27)
sex hormones
vitamin D
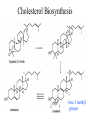
Cholesterol Biosynthesis
- lose 3 methyl
groups
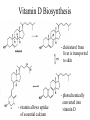
Vitamin D Biosynthesis
- cholesterol from
liver is transported
to skin
- vitamin allows uptake
of essential calcium
- photochemically
converted into
vitamin D
Notes on Terpenes
(1) Oxidation reactions are carried out by the enzyme
cytochrome P450
- activate oxygen to introduce -OH, carboxyl groups
- allow removal of C’s through decarboxylation
(2) Triterpenes form flexible rings (chair, boat conformations)
with many chiral centers {rings usually not aromatic}
- provides a huge number of potential 3D structures
- high degree of biological activity
(3) Pathways can be elucidated using labeled precursors,
such as mevalonate with a 13C at position 2
- carbon NMR experiments reveal where the label ends
up in the completed molecule
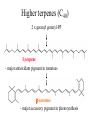
Higher terpenes (C40)
2 x geranyl geranyl-PP
Lycopene
- major antioxidant pigment in tomatoes
b-carotene
- major accessory pigment in photosynthesis
The highest terpenes (Cbig #)
~ 1 % of plants can synthesize cis-polyisoprenoids, like rubber
H3C
Commercially used rubber plants can convert nearly 100% of
their mevalonate into rubber
(photosynthesis)
Polysaccharides
Glycosides
Nucleic Acids
phosphoenol
pyruvate
Shikimate pathway
Aromatic Compounds
Lignans
Shikimate
pyruvate
acetyl
CoA CH3COSCoA
aromatic amino acids
aliphatic amino acids
CITRIC
ACID
CYCLE
Alkaloids
Peptides
Penicillins
Cyclic Peptides
CH3COSCoA
-O
2CCH2COSCoA
CH3COCH2COSCoA
CH3COSCoA
CH3COSCoA
Isoprenoids (terpenes,
steroids, carotenoids)
mevalonate
Polyketides, Fatty Acids
(4)
Prostaglandins, Macrocyclic Antibiotics
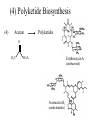
(4) Polyketide Biosynthesis
(4)
Acetate
Polyketides
O
H3C
SCoA
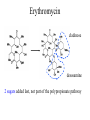
Erythromycin A
(antibacterial)
Avermectin B1
(antihelminthic)
(4) Polyketide Biosynthesis
(4)
Acetate
(1) aromatic compounds, usually
with meta placement of -OH’s
O
H3C
Polyketides:
SCoA
(2) non-aromatic macrolides
Avermectin B1
6-methyl salicylate
(antihelminthic)
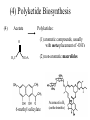
Polyketide Biosynthesis
Compounds constructed by addition of successive 2-carbon units
Ketone may or may not be later reduced
Options in final structure:
OH
O
-orH3C
C
H
H3C
C
-or-
H3C
C
Polyketide Biosynthesis
Important natural producers of polyketide metabolites:
- fungi (= molds)
- bacteria, particularly of the family Actinomycetes
- sea slugs
Polyketide Biosynthesis
Actual pathway starts with an acetyl-coA, but then successively
uses malonyl coA, which loses CO2 thus adding C2 units
- analogous to how mevalonate loses CO2 to add C5 units in
terpenoid biosynthesis
1 acetate, 3 malonyl coA’s
4 carbons are labeled
in the final product
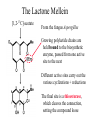
The Lactone Mellein
[1,2-13C]-acetate
From the fungus Aspergillus
Growing polyketide chains are
held bound to the biosynthetic
enzyme, passed from one active
site to the next
Different active sites carry out the
various cyclizations + reductions
The final site is a thioesterase,
which cleaves the connection,
setting the compound loose
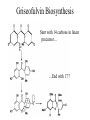
Griseofulvin Biosynthesis
Start with 14 carbons in linear
precursor…
…End with 17 ?
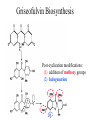
Griseofulvin Biosynthesis
Post-cyclization modifications:
(1) addition of methoxy groups
(2) halogenation
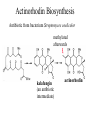
Actinorhodin Biosynthesis
Antibiotic from bacterium Streptomyces coelicolor
methylated
afterwards
kalafungin
(an antibiotic
intermediate)
actinorhodin
Aflatoxin
Isolated in 1960 after thousands of turkeys died of liver failure
from eating moldy peanut meal; culprit was Aspergillus mold
Now evidence that in parts of the world where peanuts are a
dietary staple, much liver disease is due to persistent aflatoxin
consumption
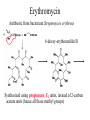
Erythromycin
Antibiotic from bacterium Streptomyces erythreus
6-deoxy-erythononlide B
Synthesized using propionate, C3 units, instead of 2-carbon
acetate units (hence all those methyl groups)
Erythromycin
cladinose
desosamine
2 sugars added last, not part of the polypropionate pathway
Erythromycin
Biosynthetic genes for erythromycin have been cloned:
- made by 3 large, polyfunctional proteins called modular
polyketide synthases
- proteins have a series of similar active sites that carry out
each successive step in the biosynthesis (hence “modular”)
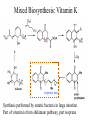
Mixed Biosynthesis: Vitamin K
isoprene unit
Synthesis performed by enteric bacteria in large intestine
Part of vitamin is from shikimate pathway, part isoprene
Rules for identifying pathways
(1) Is there nitrogen? Yes...alkaloid
(2) Count carbons
a) multiple of 5....
terpene
C10 = monoterpene
especially if the rings are
C15 = sesquiterpene
not aromatic
C20 = diterpene
b) another even number... probably polyketide
c) multiple of 3...
(w/ lots of methyls)
polypropionate
Rules for identifying pathways
Hints for carbon-counting:
1) non-skeleton carbons don’t count
- Those attached through oxygen (esters, ethers) are
usually (but not always) added after the main pathway
- For example, in R-OMe, the methoxy carbon would
not be included in your count (secondary modification)
2) sugars do not count; they are often tacked onto shikimate,
polyketide metabolites after the synthesis is complete
Rules for identifying pathways
(3) Aromatic...
a) hydroxyl groups are meta...
b) otherwise...
polyketide
shikimate
You can’t always differentiate these 2 pathways without doing
labeling experiments