* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Total Synthesis of Spiculoic Acid A
Oligonucleotide synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Metalloprotein wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Biochemistry wikipedia , lookup
Peptide synthesis wikipedia , lookup
Butyric acid wikipedia , lookup
15-Hydroxyeicosatetraenoic acid wikipedia , lookup
Specialized pro-resolving mediators wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
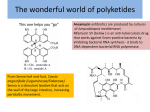
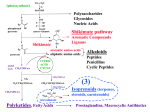
SPICULOIC ACID A Understanding its Biogenesis through its Biomimetic Total Synthesis Atahualpa Pinto and Christopher N. Boddy Department of Chemistry, Syracuse University, Syracuse, NY 13244 Biomimetic Synthesis of Route 2 Linear Precursor Abstract Spiculoic Acid A, a polyketide natural product, was recently isolated from the marine sponge Plakortis angulospiculatus. The specific objective of our research is to investigate the mode of action of dehydratase (DH) domains in polyketide biosynthesis. Two conceivable routes may be hypothesized concerning Spiculoic Acid A's biosynthesis. One containing an unusual dehydration mechanism has been proposed that leads to a β,γ-unsaturated thioester intermediate. A more appealing alternative involves a dehydration intermediate that would resemble the normal enzyme catalyzed product. To examine the latter's viability as a biosynthetic precursor its biomimetic synthesis is being carried out. Introduction H O H HO O Spiculoic Acid A RETROSYNTHESIS Diels-Alder H OTMS OTMS O H HO OEt O O O OEt Extended Enolate Precursor Spiculoic Acid A SYNTHESIS • • • • Marine polyketide from the sponge Plakortis angulospiculatus. Anticancer activity. Highly congested ring system. 2 quaternary carbons. Biosynthesis likely occurs in 2 stages: •Linear chain via Polyketide Synthase (PKS). •Cyclic core via post-PKS tailoring – Diels-Alder reaction. O i ii O 83% OH iii O 81% O E/Z = 17 : 1 Ph 3P i O • Linear chain extension in PKS is analogous to fatty acid biosynthesis: O i 1 iv O iii O OH ii O O O ii Ph 3P O O 2 OH 2 Potential Polyketide Synthase Pathways ROUTE 1 O iii Reagents: (i) 1, toluene, 120oC, overnight; (ii) DIBAL-H, CH2Cl2, -78oC, 1hr; (iii) IBX; (iv) 2. INITIALLY PROPOSED • Abnormal dehydration product. • 2 stereocenters in product. RO O v O O RO O O vi N OR' vii N O O Bn viii R=H R = TBS ix Bn R' = H R' = TFA R = TBS R=H 3 x OTMS OTMS O xiii xi OR O ROUTE 2 xii xiv WE PROPOSE • Normal dehydration product. • Extended enolate is linear precursor to cyclization product. • 1 stereocenter in product. • Favorable electronics for Diels-Alder reaction. Each PKS route leads to different Diels-Alder substrates. OTMS OTFA R =TFA R=H O O xv H O O O O HO H O Spiculoic Acid A Reagents: (v) 3; (vi) TBSCl, Imidazole; (vii) DIBAL-H; (viii) (CF3CO)2, Pyr; (ix) TBAF; (x) LDA; (xi) TMSCl, Imidazole; (xii) LiOH (aq); (xiii) IBX; (xiv) 1; (xv) TBAF, D.