* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download fatty acids
Peptide synthesis wikipedia , lookup
Point mutation wikipedia , lookup
Signal transduction wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Proteolysis wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Mitochondrion wikipedia , lookup
Genetic code wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Microbial metabolism wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Electron transport chain wikipedia , lookup
Butyric acid wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
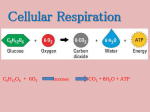
Fig. 17.1 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Dairy Fruits Grains Vegetables Protein ChooseMyPlate.gov Fig. 16.23 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Carbohydrates Lipids Proteins Mouth (salivary glands) Salivary amylase Polysaccharides, Disaccharides Stomach Pepsin Polypeptides Duodenum (pancreas, liver) Bile salts (liver) Pancreatic amylase Lipase (pancreas) Trypsin, chymotrypsin, carboxypeptidase (pancreas) Disaccharides Peptides Epithelium of small intestine Disaccharidases Peptidases Monosaccharides Fatty acids Monoglycerides Amino acids Fig. 2.12 Copyright © McGraw-Hill Education. Permission required for reproduction or display. H H C OH HO O H H C C C C O H C OH H C OH H HO – C HO H H O H H H H H C C– H H C O C C C C C C H H H H H H O H H H H H C C C C C C H H H H H H O H H H H H C C C C C C H H H H H H H H H H H H H H H H H H Enzymes C C C C C H H H H H H O H H H H H C C C C H H C H H C– H H H C O 3 H2O H C O H Fatty acids Glycerol Triglyceride molecule Fig. 2.15 Copyright © McGraw-Hill Education. Permission required for reproduction or display. CH3 CH CH3 OH CH2CH2CH2CH CH3 CH3 CH3 CH3 Cholesterol HO HO Estrogen (estradiol) CH3 OH CH CH3 O CH2CH2 C O NH CH2 C OH O– CH3 CH3 HO CH3 OH Bile salt (glycocholate) O Testosterone Fig. 2.14 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Nitrogen Phosphorus Polar (hydrophilic) region (phosphatecontaining region) Oxygen Carbon Hydrogen Nonpolar (hydrophobic) region (fatty acids) (a) (b) Fig. 2.13 Copyright © McGraw-Hill Education. Permission required for reproduction or display. HO O H H H H H H H H H H H H H H H C C C C C C C C C C C C C C C C H H H H H H H H H H H Palmitic acid (saturated) H H H H O H H H H H H H H C C C C C C C C C H H H H H H H (a) HO Double bond Double bond Linolenic acid (unsaturated) (b) Double bond H Fig. 2.16 Copyright © McGraw-Hill Education. Permission required for reproduction or display. (a) Two examples of amino acids. Each amino acid has an amine group (—NH2) and a carboxyl group (—COOH). Amino acid (alanine) H H CH3 N C C H O H OH H H N C C H O OH Amino acid (glycine) H2O (b) The individual amino acids are joined. H H H CH3 N C C H O H H N C C H O OH H N HO (c) A protein consists of a chain of different amino acids (represented by differentcolored spheres). C H C N C H O C C O H H O C (d) A three-dimensional representation of the amino acid chain shows the hydrogen bonds (dotted red lines) between different amino acids. The hydrogen bonds cause the amino acid chain to become folded or coiled. N O C C N H C C C O H O C N C O H N C C O O H C C N C H N H O H C N C C N C C N N H H C C O C N N O C H N H O C Folded C N N H C O C O H O Coiled C O H O C N C C C C C (e) An entire protein has a complex three-dimensional shape. O C H N C C O N Table 17.2 Table 17.3 Fig. 17.3 Copyright © McGraw-Hill Education. Permission required for reproduction or display. 1 ATP Production The energy released during catabolism can be used to synthesize ATP. P Catabolism Catabolism is the energyreleasing process by which larger molecules are broken down to smaller ones. Ingested food is the source of molecules used in catabolic reactions. P P Anabolism ATP Energy Energy P P P ADP + Pi 2 ATP Breakdown The energy released from the breakdown of ATP can be used during anabolism to synthesize other molecules and to provide energy for cellular processes, such as active transport and muscle contraction. Anabolism is the energyrequiring process by which smaller molecules join to form larger ones. Anabolic reactions result in the synthesis of the molecules necessary for life. Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.6 1 Cytoplasm Glucose (six-carbon molecule) 1 Glycolysis 2 ATP 2 NADH 2 pyruvic acid (three-carbon molecules) 2 Outer mitochondrial membrane 4 6 O2 Electron-transport chains Inner mitochondrial membrane 6 H2O 2 CO2 3 34 ATP 2 NADH 2 2 acetyl-CoA (two-carbon molecules) 2 citric acid (six-carbon molecules) 4 2 four-carbon molecules 3 4 CO2 2 ATP 6 NADH 2 FADH2 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.6 1 Glycolysis in the cytoplasm converts glucose to two pyruvic acid molecules and produces two ATP and two NADH. The NADH can go to the electron-transport chain in the inner mitochondrial membrane. Cytoplasm Glucose (six-carbon molecule) 1 Glycolysis 2 ATP 2 NADH 2 pyruvic acid (three-carbon molecules) 2 Outer mitochondrial membrane 4 6 O2 Electron-transport chains Inner mitochondrial membrane 6 H2O 2 CO2 3 34 ATP 2 NADH 2 2 acetyl-CoA (two-carbon molecules) 2 citric acid (six-carbon molecules) 4 2 four-carbon molecules 3 4 CO2 2 ATP 6 NADH 2 FADH2 Fig. 17.5 Copyright © McGraw-Hill Education. Permission required for reproduction or display. 1 2 Glycolysis converts glucose to two pyruvic acid molecules. There is a net gain of two ATP and two NADH from glycolysis. Anaerobic respiration, which does not require O2, includes glycolysis and converts the two pyruvic acid molecules produced by glycolysis to two lactic acid molecules. This conversion requires energy, which is derived from the NADH generated in glycolysis. Glucose 2 ATP Glycolysis 1 2 NADH Anaerobic respiration 2 pyruvic acid 2 2 lactic acid Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.6 1 Glycolysis in the cytoplasm converts glucose to two pyruvic acid molecules and produces two ATP and two NADH. The NADH can go to the electron-transport chain in the inner mitochondrial membrane. Cytoplasm Glucose (six-carbon molecule) 1 Glycolysis 2 ATP 2 NADH 2 The two pyruvic acid molecules produced in glycolysis are converted to two acetyl-CoA molecules, producing two CO2 and two NADH. The NADH can go to the electron-transport chain. 2 pyruvic acid (three-carbon molecules) Outer mitochondrial membrane 4 6 O2 Electron-transport chains Inner mitochondrial membrane 6 H2O 2 CO2 3 34 ATP 2 NADH 2 2 acetyl-CoA (two-carbon molecules) 2 citric acid (six-carbon molecules) 4 2 four-carbon molecules 3 4 CO2 2 ATP 6 NADH 2 FADH2 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.6 1 Glycolysis in the cytoplasm converts glucose to two pyruvic acid molecules and produces two ATP and two NADH. The NADH can go to the electron-transport chain in the inner mitochondrial membrane. Cytoplasm Glucose (six-carbon molecule) 1 Glycolysis 2 ATP 2 NADH 2 The two pyruvic acid molecules produced in glycolysis are converted to two acetyl-CoA molecules, producing two CO2 and two NADH. The NADH can go to the electron-transport chain. 2 pyruvic acid (three-carbon molecules) Outer mitochondrial membrane 4 Inner mitochondrial membrane 6 H2O 2 CO2 3 The two acetyl-CoA molecules enter the citric acid cycle, which produces four CO2, six NADH, two FADH2, and two ATP. The NADH and FADH2 can go to the electron-transport chain. 6 O2 Electron-transport chains 34 ATP 2 NADH 2 2 acetyl-CoA (two-carbon molecules) 2 citric acid (six-carbon molecules) 4 2 four-carbon molecules 3 4 CO2 2 ATP 6 NADH 2 FADH2 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.6 1 Glycolysis in the cytoplasm converts glucose to two pyruvic acid molecules and produces two ATP and two NADH. The NADH can go to the electron-transport chain in the inner mitochondrial membrane. Cytoplasm Glucose (six-carbon molecule) 1 Glycolysis 2 ATP 2 NADH 2 The two pyruvic acid molecules produced in glycolysis are converted to two acetyl-CoA molecules, producing two CO2 and two NADH. The NADH can go to the electron-transport chain. 2 pyruvic acid (three-carbon molecules) Outer mitochondrial membrane 4 Inner mitochondrial membrane 6 H2O 2 CO2 3 4 The two acetyl-CoA molecules enter the citric acid cycle, which produces four CO2, six NADH, two FADH2, and two ATP. The NADH and FADH2 can go to the electron-transport chain. The electron-transport chain uses NADH and FADH2 to produce 34 ATP. This process requires O2, which combines with H+ to form H2O. 6 O2 Electron-transport chains 34 ATP 2 NADH 2 2 acetyl-CoA (two-carbon molecules) 2 citric acid (six-carbon molecules) 2 four-carbon molecules 3 4 CO2 2 ATP 6 NADH 2 FADH2 Fig. 17.7 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Mitochondrion Cytosol Outer membrane H+ H+ H+ Outer compartment H+ 2e– I H+ 2e– III H+ H+ H+ H+ II Inner compartment H+ H+ 2 H+ Inner membrane H+ H+ H+ Carrier protein ATP H+ ATP synthase Pi IV 2e– NADH Pi + ADP 2e– 2e– H+ 1 FADH2 H+ H+ ATP 2e– NAD+ H2O 2H+ 1 2 1 ADP NADH or FADH2 transfer their electrons to the electron-transport chain. 2 As the electrons move through the electron-transport chain, some of their energy is used to pump H+ into the outer compartment, resulting in a higher concentration of H+ in the outer than in the inner compartment. 3 O2 4 H+ 3 The H+ diffuse back into the inner compartment through special channels (ATP synthase) that couple the H+ movement with the production of ATP. The electrons, H+, and O2 combine to form H2O. 4 ATP is transported out of the inner compartment by a carrier protein that exchanges ATP for ADP. A different carrier protein moves phosphate into the inner compartment. Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.6 1 Glycolysis in the cytoplasm converts glucose to two pyruvic acid molecules and produces two ATP and two NADH. The NADH can go to the electron-transport chain in the inner mitochondrial membrane. Cytoplasm Glucose (six-carbon molecule) 1 Glycolysis 2 ATP 2 NADH 2 The two pyruvic acid molecules produced in glycolysis are converted to two acetyl-CoA molecules, producing two CO2 and two NADH. The NADH can go to the electron-transport chain. 2 pyruvic acid (three-carbon molecules) Outer mitochondrial membrane 4 Inner mitochondrial membrane 6 H2O 2 CO2 3 4 The two acetyl-CoA molecules enter the citric acid cycle, which produces four CO2, six NADH, two FADH2, and two ATP. The NADH and FADH2 can go to the electron-transport chain. The electron-transport chain uses NADH and FADH2 to produce 34 ATP. This process requires O2, which combines with H+ to form H2O. 6 O2 Electron-transport chains 34 ATP 2 NADH 2 2 acetyl-CoA (two-carbon molecules) 2 citric acid (six-carbon molecules) 2 four-carbon molecules 3 4 CO2 2 ATP 6 NADH 2 FADH2 Table 17.2 Fig. 10.22 Fig. 17.9 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Nutrients Absorbed Nutrients are absorbed from the digestive tract and carried by the blood to the liver. Nutrients Processed The liver converts nutrients into energy-storage molecules, such as glycogen, fatty acids, and triglycerides. Amino acids are also used to synthesize proteins, such as plasma proteins. Fatty acids and triglycerides produced by the liver are released into the blood. Nutrients not processed by the liver are also carried by the blood to tissues. Amino acids Proteins Triglycerides Nonessential amino acids Glucose Glycogen Glycerol -keto acids Ammonia Acetyl-CoA Fatty acids Urea Energy Nutrients Stored and Used Nutrients are stored in adipose tissue as triglycerides and in muscle as glycogen. Nutrients also are a source of energy for tissues. Amino acids are used to synthesize proteins. Amino acids Glucose Glucose Fatty acids Glucose Fatty acids Glucose Glycerol Proteins Energy Glycogen Muscle Triglycerides Adipose tissues Most tissues (including muscle and adipose) Energy Nervous tissue Fig. 16.26 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Chylomicron Phospholipid (4%) Triglyceride (90%) Cholesterol (5%) Protein (1%) Low-density lipoprotein (LDL) Phospholipid (20%) Triglyceride (10%) Cholesterol (45%) Protein (25%) High-density lipoprotein (HDL) Phospholipid (30%) Triglyceride (5%) Cholesterol (20%) Protein (45%) Fig. 10.23 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.8 Food Lipid Carbohydrate Protein Monosaccharides (e.g., glucose) Fatty acids Glycerol Glycolysis Ketones Pyruvic acid Acetyl-CoA Amino acids -keto acid NADH Citric acid cycle Ammonia ATP CO2 Urea O2 NADH FADH2 Electrontransport chain H2O ATP Fig. 17.10 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Stored Nutrients Used Stored energy molecules are used as sources of energy: Glycogen is converted to glucose, and triglycerides are converted to fatty acids. Molecules released from tissues are carried by the blood to the liver. Adipose tissue Muscle Proteins Amino acids Glycogen Glucose Triglycerides Glycerol Energy Nutrients Processed The liver processes molecules to produce additional energy sources: Glycogen and amino acids are converted to glucose and fatty acids to ketones. Glucose and ketones are released into the blood and are transported to tissues. Lactic acid Fatty acids Nervous tissue Energy Energy Fatty acids Ketone bodies Glucose Energy Glycerol Fatty acids Glycogen Glucose -keto acid Amino acids Most tissues (including muscle) Ammonia Energy Urea Acetyl-CoA Energy Ketone bodies Copyright © McGraw-Hill Education. Permission required for reproduction or display. Fig. 17.8 Food Lipid Carbohydrate Protein Monosaccharides (e.g., glucose) Fatty acids Glycerol Glycolysis Ketones Pyruvic acid Acetyl-CoA Amino acids -keto acid NADH Citric acid cycle Ammonia ATP CO2 Urea O2 NADH FADH2 Electrontransport chain H2O ATP Fig. 10.23 Fig. 10.22 Table 10.3 Fig. 10.19 Fig. 10.21 Fig. 17.11 Copyright © McGraw-Hill Education. Permission required for reproduction or display. Radiation from sun and water Evaporation Convection from cool breeze Radiation from sand Conduction from hot sand ©M.M. Sweet/Flickr/Getty Images RF Fig. 17.12 Copyright © McGraw-Hill Education. Permission required for reproduction or display. 3 4 Actions Receptors in the skin and hypothalamus detect increases in body temperature. The control center in the hypothalamus activates heat loss mechanisms. Effectors Respond: Increased sweating increases evaporative heat loss. Dilation of skin blood vessels increases heat loss from the skin. Behavioral modifications, such as taking off a jacket or seeking a cooler environment, increase heat loss. Homeostasis Disturbed: Body temperature increases. Start here Homeostasis Disturbed: Body temperature decreases. Actions Receptors in the skin and hypothalamus detect decreases in body temperature. The control center in the hypothalamus activates heat-conserving and heat-generating mechanisms. 5 6 Homeostasis Restored: Body temperature decreases. Body temperature (normal range) 1 Body temperature (normal range) 2 Reactions Homeostasis Restored: Body temperature increases. Reactions Effectors Respond: Constriction of skin blood vessels decreases heat loss from the skin. Shivering increases heat production. Behavioral modifications, such as putting on a jacket or seeking a warmer environment, decrease heat loss.