* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download CHAPTER 21 PHENOLS AND ARYL HALIDES
Bottromycin wikipedia , lookup
Elias James Corey wikipedia , lookup
George S. Hammond wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
Homoaromaticity wikipedia , lookup
Discodermolide wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Aza-Cope rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Stille reaction wikipedia , lookup
Aromaticity wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
Aromatization wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
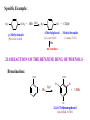
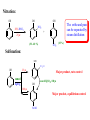
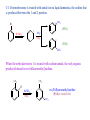
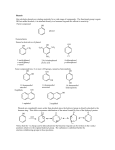
CHAPTER 21 PHENOLS AND ARYL HALIDES NUCLEOPHILIC AROMATIC SUBSTITUTION 21.1 STRUCTURE AND NOMENCLATURE OF PHENOLS Phenol: Compounds that have a hydroxyl group directly attached to a benzene ring For example: OH H3C OH Phenol 4-Methylphenol (苯酚) (4-甲基苯酚) Naphthols or phenanthrols: Compounds that have a hydroxyl group attached to a polycyclic benzenoid ring. For example: HO OH 8 1 7 2 6 3 5 10 1 8 OH 2 7 4 1-Naphthol (1-ÝÁ·Ó£© 9 6 2-Naphthol (2-ÝÁ·Ó£© 5 4 3 9-Phenanthrol (9-·Æ·Ó£© 21.1A NOMENCLATURE OF PHENOLS (1) In many compounds phenol is the base name. For example: Br NO2 Cl OH OH 4-Chlerophenol (对-氯苯酚) 2-Nitrophenol (临-硝基苯酚) OH 3-Bromophenol (间-溴苯酚) (2) The methylphenols are commonly called cresols. For example: CH3 CH3 OH H3C OH OH 2-Methylphenol (临-甲酚) 4-Methylphenol (对-甲酚) 3-Methylphenol (间-甲酚) (3) The benzenediols also have common names. OH OH OH HO OH OH 1,2-Benzenediol (¶ù²è ·Ó,ÁÙ±½¶þ·Ó) 1,3-Benzenediol (À×Ëö·Ó,¼ä±½¶þ·Ó£© 1,4-Benzenediol (¶Ô±½¶þ·Ó) 21.2 NATURALLY OCCURRING PHENOLS Phenols and related compounds occur widely in nature. For example: CH2CH=CH2 CO2CH3 CH3 OH NH3+ CH2CHCO2- HO Tyrosine(ÀÒ°±Ëá) OCH3 OH OH CH(CH3)2 Methyl salicylate Eugenol (×Ó¶¡ Ïã ·Û) (Ë®ÑîËá¼×õ¥£© Thymo (÷êÏ㠲ݷÓ) oil of wintergreen oil of cloves (¶¡ Ïã ÓÍ£©thyme (÷êÏã ²ÝÊô) £¨ ¶¬ÇàÓÍ£© CH3 OH OH O OH H H O CONH2 H HO Estradiol (´Æ¶þ´¼) OH Y HO CH3 Z H OH N(CH3)2 Y = Cl, Z = H; Aureomycin (½ðùËØ) Y = H, Z = OH; terramycin (ÍÁùËØ£© 21.3 PHYSICAL PROPERTIES OF PHENOLS (1) Having higher boiling points: phenols are able to form strong intermolecular hydrogen bonds . For example: phenol (bp,182℃) has a boiling point more than 70℃ higher than toluene(bp,110.6℃),even though the two molecular have almost the same molecular weight. (2) Modest solubility in water: the ability to form strong hydrogen bonds to molecules of water 21.4 SYNTHESIS OF PHENOLS 21.4A LABORATORY SYNTHESIS General Reaction: Ar-NH2 HONO Ar-N2+ H3O+ heat Ar-OH Specific Examples: NH2 OH (1) NaNO2, H2SO4 0-5¡æ (2) H3O+, heat R R = Br 3-Bromophenol 66% R = NO2 3-Nitrophenol 80% R NH2 OH Br Br (1) NaNO2, H2SO4 0-5¡æ (2) H3O+, heat CH3 2-Bromo-4-methylphenol 80-92% CH3 21.4B INDUSTRIAL SYNTHESIS 1. Hydrolysis of Chlorobenzene Cl NaOH 350¡æ (high pressure) ONa OH HCl 2. Alkali Fusion of Sodium Benzenesulfonate SO3Na ONa NaOH 350¡æ (high pressure) OH HCl Sodium benzenesulfonate (±½»ÇËáÄÆ £© 3. From Cumene Hydroperoxide Friedel-Crafts alkylation: + CH2=CHCH3 H3PO4 250¡æ pressure Cumene (Òì±û»ù±½) Oxidization: + O2 95-135¡æ O O H Cumene hydroperoxide (¹ý Ñõ»¯Òì±û»ù±½) Hydrolytic rearrangement: O O H O H+, H2O 50-90¡æ OH + Acetone (±ûͪ £© Corresponding Mechanism: Friedel-Crafts alkylation: H H+ - H+ Oxidization: H R O O O2 + O O O O H + C• Hydrolytic rearrangement: O O H H+ H2O O O+ O O H CH3H O - H2O H phenyl anion migration to oxygen H + O OH2 C O - H+ O + HO CH3 25.1 REACTIONS OF PHENOLS AS ACIDS 21.5A STRENGTH OF PHENOLS AS ACIDS Phenols are much stronger acids than alcohols. For example: Cyclohexanol OH Phenol (»· ¼º´¼£© OH pKa = 18 (±½·Ó£© pKa = 9.89 The reason : (1) The carbon atom that bears the hydroxyl group in phenol is sp2-hybridized, whereas, in cyclohexane , it is sp3 –hybridized. (2) Resonance structures for phenol: H O H O H H O O H O 21.5B DISTINGUISHING AND SEPARATING PHENOLS FROM ALCOHOLS AND CARBOXYLIC ACIDS Phenols dissolve in aqueous sodium hydroxide : Phenols are more acidic than water. OH + NaOH Stronger acid Stronger pKa = 10 base (slightly soluble) H2O O-Na+ + H2O Weaker base (soluble) Weaker acid pKa = 16 Whereas most alcohols with six carbon atoms or more do not dissolve in aqueous sodium hydroxide . we can distinguish And separate phenols from most alcohols by this way. 21.6 OTHER REACTION PF THE OH- GROUP OF PHENOL Phenols react with carboxylic acid anhydrides and acid chlorides to form esters. For example: OH O RCCl O O CR base + Cl 21.6A PHENOLS IN THE WILLIAMSON SYNTHESIS Phenols can be convert to ethers through the williamson synthesis. General reaction: ArOH NaOH ArO-Na+ R-X X = Cl, Br, I, OSO2R' or ,OSO2OR. ArOH + NaX Specific Examples: O-Na+ OH CH2CH3_ I NaOH CH3 OCH2CH3 CH3 + NaI CH3 O-Na+ OH + NaOH H2O OCH3 CH3OSO3OCH3 + NaOSO2OCH3 21.7 CLEAVAGE OF ALKYL ARYL ETHERS When alkyl aryl ethers react with strong acids such as HI and HBr, the reaction produces an alkyl halide and a phenol. For example: Specific Example: H3C OCH3 + HBr H2O OH + CH3 Br H3C 4-Methylphenol p-Methylanisole (4-¼×»ù±½·Ó£© (¶Ô-¼×»ù±½¼×ÃÑ) Methyl bromide (¼×»ùäå »¯Îï £© HBr no reaction 21.8 REACTION OF THE BENZENE RING OF PHENOLS Bromination: OH OH Br + 3Br2 Br H2O + 3 HBr Br 2,4,6-Tribromophenol (2,4,6-Èýäå ±½·Ó£© Nitration: OH OH OH The ortho and para can be separated by steam distillation NO2 20% HNO3 + 25¡æ (30 - 40 %) NO2 (15 %) Sulfonation: OH SO3H OH 25¡æ Major product, rate control concd OH H2SO4 concd H2SO4, 100¡æ 100¡æ Major product, equilibrium control SO3H Kolbe Reaction: O OH OCCH3 COOH + Salicylic acid (Ë®ÑîËᣩ COOH O CH3C_ O 2 H+ Acetic anhydride (ÒÒËáôû£© O + CH3 COH Axetylsalicylic acid (ÒÒõ£Ë®ÑîËᣩ 21.9 THE CLAISEN REARRANGEMENT Claisen rearrangement: heating allyl phenyl ether to 200℃ effects an intramolecular reaction. 14 OCH2CH=CH2 OH 14 200¡æ Allyl phenol ether (Ï©±û»ù·ÓÃÑ£© CH2CH=CH2 o-Allylphenol (¶ÔÏ©±û»ù±½·Ó£© Mechanism: O CH O H2C H2C CH2 14CH 14 CH 2 2 OH CH H tautomerization -H+, +H+ CH 14CH 2 A Claisen rearrangement also takes place when allyl vinyl ethers are heated. O heat Allyl vinyl ether (Ï©±û»ùÏ©¶¡ »ùÃÑ£© O Aromatic transition state (·¼»· ¹ý ¶É̬ £© O 4-Pentenal 21.10 QUINONES Hydroquinones produces ρ-Benzoquinone by mide oxidizing agents OH O -2e+2e- OH Hydroquinone (¶Ô±½¶þ·Ó) + 2H+ O p-Benzoquinone (¶Ô-±½õ«) ρ-Benzoquinone is easily reduced by mild reducing agents to hydroquinones 21.11 ARYL HALIDES AND NUCLEOPHILIC AROMATIC SUBSTITUTION Aryl halides and vinylic halides are relatively unreactive toward nucleophilic substitution under conditions that give facile nucleophilic substitution with alkyl halides. Reason: (1) Phenyl cations are very unstable. (2) Halogen bonds of aryl (and vinylic) halides are shorter and stronger than those of alkyl, allylic, and benzylic halides because of the hybridized state and the resonance. But aryl halides can be remarkably reactive toward nucleophiles if they bear certain substituents or when we allow them to react under the proper conditions. 21.11A NUCLEOPHILIC AROMATIC SUBSTITUTION BY ADD ELIMINATION: THE SNAr MECHANISM Nucleophilic substitution can occur when strong electron-withdrawing groups are ortho or para to the halogen atom. Cl OH NO2 - + OH aq. NaHCO3 NO2 H+ 130¡æ OH Cl NO2 + OH- NO2 H+ aq. NaHCO3 100¡æ NO2 NO2 OH Cl O2N NO2 + OH NO2 - aq. NaHCO3 H+ O2N NO2 35¡æ NO2 The temperature is related to the number of ortho or para nitro groups But the meta-nitro group does not produce a similar activating effect. Mechanism: Cl Cl elimination fast addition slow + OH- NO2 NO2 CF3 O- OH OH + Cl- OH + ClNO2 NO2 CF3 Cl m-(Trifluoromethyl)aniline NaNH2 NH3 (¶Ô-Èý·ú ¼×»ù±½°±£© NH2 The delocalized carbanion is stabilized by electron-withdrawing groups in the positions ortho and para to the halogen atom. HO O N Cl HO O O Cl HO O O N N Cl HO O O Cl N O 21.11B NUCLEOPHILIC AROMATIC SUBSTITUTION THROUGH AN ELIMINATION-ADDITION MECHANISM: BENZYNE Chlorobenzene can be converted to phenol by heating it with aqueous sodium hydroxide in a pressurized reactor . Cl ONa NaOH 350¡æ (high pressure) OH HCl Bromobenzene reacts with the very powerful base, in liquid ammonia Br NH + K+ NH -33¡æ NH3 + KBr Aniline (±½°· £© C-14 bromobenzene is treated with amide ion in liquid ammonia, the aniline that is produced between the 1 and 2 position. 14 Br 14 14 K+NH2- NH2 (50%) NH2NH3 14 (50%) NH2 When the ortho derivative 1 is treated with sodium amide, the only organic product obtained is m-(trifluoromethyl)aniline. CF3 CF3 Cl m-(Trifluoromethyl)aniline NaNH2 NH3 (¶Ô-Èý·ú ¼×»ù±½°±£© NH2 Mechanism: CF3 CF3 Cl H 1 NaNH2 X NH3 NH3 (-HCl) Less stable carbanion NH CF3 CF3 4 CF3 2 NH2 NH3 + NH2 NH2 3 More stable cabanion Carbanion 3 is more stable than 4 because the carbon atom bearing the negative charge is closer to the highly electronegative trifluoromethyl group.