* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Glycogen Metabolism
Proteolysis wikipedia , lookup
Catalytic triad wikipedia , lookup
Biochemical cascade wikipedia , lookup
Lipid signaling wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Oligonucleotide synthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Metalloprotein wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Citric acid cycle wikipedia , lookup
Biosynthesis wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Blood sugar level wikipedia , lookup
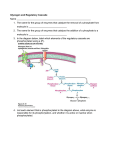
Pentose Phosphate Pathway Where the ribose comes from? • The pentose phosphate pathway is an alternate route for the oxidation of glucose. Used for nucleic acid synthesis Reactions of the pentose phosphate pathway occur in the cytosol in two phases • 1st phase Glucose 6-phosphate + 2 NADP++ H2O ribose 5-phosphate + CO2 + 2 NADPH + 2 H+ • 2nd phase The pentose phosphates are recycled back to glucose 6phosphate. Overall, 6 five-carbon sugars are converted to 5 six-carbon sugars. Overview • Function – NADPH production • Reducing power carrier – Synthetic pathways • Role as cellular antioxidants – Ribose synthesis • Nucleic acids and nucleotides 1st phase: NADPH producing reactions 1. 2. 3. Glucose-6-P dehydrogenase Lactonase 6-P-gluconate dehydrogenase 2nd phase: 1. Epimerase; 2. Isomerase 3. Transketolase 4. Transaldolase 5. Phosphohexose isomerase Ru–5-P: ribulose-5-P; X-5-P: xylulose-5-P; R-5-P: ribose -5-P Used for nucleic acid synthesis Regulation • Glucose-6-P dehydrogenase (G6PDH) – First step – Rate limiting – Feedback inhibited by NADPH – Induced by insulin Role of NADPH in the RBC • Production of superoxide – Hb-Fe2+-O2 -> Hb-Fe3+ + O2-. • Spontaneous reaction • O2-. + 2H+ > 2H2O2 • Both O2-. & H2O2 can damage cell membranes, and cause hemolysis Glycine – cycteine - glutamate G6PDH Deficiency and Hemolytic Anemia • One of the most common genetic diseases – 4 hundred variants of G6PDH deficiency – Mediterranean, Asian, African descent • 400 million people affected worldwide • 10-14% of African-American men with G6PD deficiency G6PDH Deficiency and Hemolytic Anemia • The chemicals known to increase oxidant stress – Primaquine and quinine (anti-malarial drug) – Sulfonamides (antibiotic) – Asprin – Quinadine – Naphthalene – Fava beans Exposure to these chemicals results in increased cellular production of superoxide and hydrogen peroxide Glycogen Metabolism Liver Cell Glucose is stored as glycogen predominantly in liver and muscle cells. Glycogen is a polymer of glucose residues linked by a(14) glycosidic bonds, mainly a(16) glycosidic bonds, at branch points. Glycogen catabolism (breakdown) Glycogen phosphorylase catalyzes phosphorolytic cleavage of the a(14) glycosidic linkages of glycogen, releasing glucose-1-phosphate as reaction product. glycogen(n residues) + Pi glycogen (n–1 residues) + glucose-1-phosphate Phosphorylase can cleave a(14) linkages only to within 4 residues of a branch point. This is called a "limit branch". Debranching enzyme has 2 enzyme actives: Transferase a-1,6-glucosidase The transferase transfers 3 glucose residues from a 4-residue limit branch to the end of another branch, reducing the limit branch to a single glucose residue. transferase The a-1,6-glucosidase catalyzes hydrolysis of the a(16) linkage, yielding free glucose. This is a minor fraction of glucose released from glycogen. Phosphoglucomutase catalyzes the reversible reaction: glucose-1-phosphate glucose-6-phosphate Glycogen Glucose-1-P Glucose Hexokinase or Glucokinase Glucose-6-Pase Glucose-6-P Glucose + Pi Glycolysis Pathway Pyruvate Glucose metabolism in liver. Glucose-6-phosphate may (mainly in liver) be dephosphorylated by glucose-6-phosphotase for release into the blood. glucose-6-phosphate + H2O glucose + Pi Most other tissues lack this enzyme. Glycogen synthesis UDP-glucose pyrophosphorylase Uridine diphosphate glucose (UDP-glucose) is the immediate precursor for glycogen synthesis. Glycogenin initiates glycogen synthesis. • Glycogenin is an enzyme that catalyzes attachment of a glucose molecule to one of its own tyrosine residues. • Glycogenin is a dimer, and evidence indicates that the 2 copies of the enzyme glucosylate one another. Tyr active site active site Tyr Glycogenin dimer Glycogenin catalyzes glucosylation (UDP-glucose as the donor) to yield an O-linked disaccharide with a(14) glycosidic linkage. This is repeated for second glucose added. Glycogen Synthase then catalyzes elongation of glycogen chains initiated by Glycogenin. A branching enzyme transfers a segment from the end of a glycogen chain to the C6 hydroxyl of a glucose residue of glycogen to yield a branch with an a(16) linkage. Regulation of glycogen metabolism • Regulating site for glycogen synthesis Glycogen synthase • Regulating site for glycogen catabolism Glycogen phosphorylase Glycogen Phosphorylase AMP activates Phosphorylase ATP & glucose-6-phosphate inhibit Phosphorylase Thus glycogen breakdown is inhibited when ATP and glucose6-phosphate are plentiful. Glycogen Synthase Activated by glucose-6-P (opposite of effect on Phosphorylase). Thus Glycogen Synthase is active when high blood glucose leads to elevated intracellular glucose-6-P. Regulation by hormones Glucagon and epinephrine: • Inhibit glycogen synthase • Activate glycogen phosphorylase • Increase glycogen catabolism and increase blood glucose Insulin: • Inhibit glycogen phosphorylase • Activate glycogen synthase • Increase glycogen synthesis and decrease blood glucose Hormone (epinephrine or glucagon) via G Protein (Ga-GTP) Adenylate cyclase (inactive) Adenylate cyclase (active) catalysis ATP cyclic AMP + PPi Activation Phosphodiesterase AMP Protein kinase A (inactive) Protein kinase A (active) ATP ADP Phosphorylase kinase (b-inactive) Phosphatase Phosphorylase kinase (P) (a-active) ATP Pi ADP Phosphorylase (b-allosteric) Phosphorylase (P) (a-active) Phosphatase Pi Regulation of Glycogen Phosphorylase by Hormones Regulation of Glycogen Synthase by Hormones Glycogen Function • In liver – The synthesis and breakdown of glycogen is regulated to maintain blood glucose levels. • In muscle - The synthesis and breakdown of glycogen is regulated to meet the energy requirements of the muscle cell.