* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Glycogen Phosphorylase
Photosynthetic reaction centre wikipedia , lookup
Catalytic triad wikipedia , lookup
Polyadenylation wikipedia , lookup
Enzyme inhibitor wikipedia , lookup
Proteolysis wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Metalloprotein wikipedia , lookup
Biosynthesis wikipedia , lookup
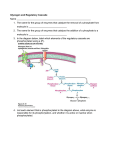
Glycogen Phosphorylase Carlos Mendoza Lee Sutton Kristen Harris • Glycogen Phosphorylase is an enzyme found in glycogen granules in the cytosol • Glycogen Phosphorylase releases sugar from glycogen by clipping off glucose from the chains of a glycogen molecule. It catalyzes a phosphorolysis reaction where it adds a phosphate to break off glucose: (glucose)n + Pi <<====>> glucose 1-phosphate + (glucose)n-1 Protein Characteristics 1 61 121 181 241 301 361 421 481 541 601 661 721 781 kqisvrglag qqhyyekdpk lgngglgrla karpeftlpv dfnlkdfnvg fksskfgcrd tcaytnhtvi veegavkrin wlvlcnpgla reykvhinpn pgyhmakmii teasgtgnmk yydripelrq yknprewtrm venvtelkkn riyylslefy acfldsmatl hfygrvehts gyiqavldrn pvrtnfdafp pealerwpvh mahlciagsh eiiaerigee slfdvqvkri klitaigdvv fmlngaltig iieqlssgff virniatsgk fnrhlhftlv mgrtlqntmv glaaygygir qgakwvdtqv laenisrvly dkvaiqlndt lletllprhl avngvarihs yisdldqlrk heykrqllnc nhdpvvgdrl tmdganvema spkqpdlfkd fssdrtiaqy kdrnvatprd nlalenacde yefgifnqki vlampydtpv pndnffegke hpslaipelm qiiyeinqrf eilkktifkd llsyvddeaf lhvitlynri rviflenyrv eeageenffi ivnmlmhhdr areiwgveps yyfalahtvr atyqlgldme cggwqmeead pgyrnnvvnt lrlkqeyfvv rvlvdlerld lnrvaaafpg fyelephkfq irdvakvkqe kkepnkfvvp slaekvipaa fgmrvedvdr fkvfadyeey rqrlpapdea dhlvgrwirt eleeieedag dwlrygnpwe mrlwsakapn aatlqdiirr wdkawevtvk dvdrlrrmsl nktngitprr nklkfaayle rtvmiggkaa dlseqistag ldqrgynaqe vkcqervsal ap 8GPB Amino acid chain • sizeof(Glycogen Phosphorylase) = 832 aa In humans found on chromosome 20 • Glycogen Phosphorylase can be in a dimer or tetramer form. However, it is thought to be mostly in the dimer form in vivo. Each monomer consists of two domains: • C terminal domain - domain responsible for catalysis of the reaction (blue) • N terminal domain - domain responsible for regulation of the enzyme (pink) Both domains consist of a beta sheet core surrounded by layers of alpha helical segments C TERMINAL DOMAIN • The active site is actually in the cleft between the C and N terminal domains • A cofactor PLP (pyridoxal phosphate) binds near the active site to facilitate the reaction N TERMINAL DOMAIN • Contains different allosteric effector sites that determine when the enzyme is active (R state) or inactive (T state.) • Can be phosphorylated at Serine 14 to induce active state (see previous slide) • Or can bind with Amp to induce active state Change from T to R state • Problem - T state is found as a monomer, but R state only crystallized as a tetramer. • Glycogen Phosphorylase forms tetramers when there is a lot of the enzyme present, which is the case when glucose is needed and there is a lot of the R state of the enzyme present. • This makes the two hard to compare visually. Upon phosphorylation • Amino acids 10-22 swing 120 degrees causing a change in both the N and C domain overall structures The phosphate group is in red, residues 10-22 surround it Additional change from T to R State • Residues 280-286 go from blocking active site in T state to pulled out away from it in the R state. AMINO ACIDS 280-286 IN YELLOW T (INACTIVE) R (ACTIVE) • MAIN POINT IS THAT BINDING AT EFFECTOR SITES FOR PHOPSHATE OR AMP LEAD TO CHANGES IN THE PROTEIN’S SHAPE THAT MAKES IT MORE ABLE TO ACCEPT GLYCOGEN AT IT’S ACTIVE SITE Phylogenetic trees unrooted Rooted(from unrooted tree) Tree characteristics •All sequences are closely conserved •Bacteria tend to be more closely related to other bacteria than to the others •The brain, liver, and muscle forms of Glycogen phosphorylase tend to group together •The fruit fly is grouped closer to the vertebrates than to the bacteria as expected •The brain, liver, and muscle forms in one organism are almost identical to the brain, liver, and muscle forms in different organisms •Glycogen phosphorylase showed up in a lot of bacteria, and then in vertebrates, but not much in the middle this might be because it hasn’t been studied much in other organisms Evolution of Glycogen phosphorylase •Over time glycogen phosphorylase has changed very little it is very similar in bacteria and vertebrates •There is more variation in bacteria glycogen phosphorylase than in vertebrates •This might be to the fact that bacteria has been around much longer and has had more time to change •Or it might be that Glycogen phosphorylase in vertebrates is unable to change with out causing major problems changes in bacteria are not as damaging Hydrophobicity •Exist in a hydrophilic environment •The percentage of solvent-accessible hydrophobic elements is much greater than in the “average” globular protein Hydrophobicity Grease PDB entry 8GPB Blast •Not much was returned besides glycogen phosphorylase •A few other enzymes that break down sugars were returned such as maltodextrin phosphorylase and Glucan phosphorylase Software Tools • Structure Visualization : Cn3D software, using • • • • images from NCBI. Phylogenetic Analysis : Used ClustalW on the Biology Workbench Hydrophobicity Analysis: Used Grease on the Biology Workbench BlastP used in biology Workbench to search for similar proteins Used Google for everything References Fletterick, Robert and Stephen Sprang. Glycogen phosphorylase Structures and Function.Department of Biochemistry and Biophysics, School of Medicine, University of California. Acc. Chem Res. 1982, 15, 361-369. Goodsell, David. Glycogen Phosphorylase. Molecule of the Month. Protein Data Bank. Internet. http://www.rcsb.org/pdb/molecules/pdb24_1.html. 5/13/03. Gillis, Jeremy. Glycogen Phosphorylase - Structure and Function Term Paper. Internet. http://www.chem.uwec.edu/webpapers_F98/gillis/Structure/struct_descrip.htm l. 5/13/03.