* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download W19 Aldehydes ketones I
Woodward–Hoffmann rules wikipedia , lookup
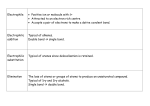
Asymmetric hydrogenation wikipedia , lookup
Marcus theory wikipedia , lookup
Elias James Corey wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Homoaromaticity wikipedia , lookup
Metal carbonyl wikipedia , lookup
George S. Hammond wikipedia , lookup
Stille reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Ene reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Aldol reaction wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Hydroformylation wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Organic chemistry II Zdeněk Friedl Chapter 19 Aldehydes and ketones I Solomons & Fryhle: Organic Chemistry 8th Ed., Wiley 2004 Nucleophilic addition to carbonyl C=O group • • • • • • • structure of C=O carbonyl group physical properties of aldehydes and ketones reaction scheme of aldehydes and ketones nucleophilic addition AN to C=O group: cyanohydrins, hemiacetals, acetals, thioacetals nucleophilic addition-elimination AN(E) to C=O group: imines, oximes, hydrazones, enamines nucleophilic addition of phosphorus ylides oxidation of aldehydes and ketones 2 Aldehydes and ketones 3 Physical properties of aldehydes and ketones I 4 Physical properties of aldehydes and ketones II 5 The structure of C=O carbonyl group 6 Electron structure of carbonyl C=O group an electrostatic potential map of acetaldehyde and acetone Acetone 7 The nomenclature of addition, addition-elimination and addition-substitution reactions of C=O compounds AN AN(E) AN(S) O OH R C + H NuH R CH Nu O R C H + NuH2 O + NuH R C O R1 R CH Nu + H2O O + R1-OH R C Nu 8 AN, AN(E) The reaction scheme of aldehydes a ketones H+ δ− δ+ H O C δ− δ+ O C H H δ− δ+ O C α CH R H B R1 α CH R H B δ− Nu (AN) 9 AN The nucleophilic addition to C=O bond I 10 AN The nucleophilic addition to C=O bond II an addition of STRONG nucleophile 11 AN The nucleophilic addition to C=O bond III the reaction is not stereoselective 12 AN Addition of hydrogen cyanide: CYANOHYDRINS 13 Amygdalin [(6-O-β-D-Glucopyranosyl-β-D-glucopyranosyl)oxy]-2-phenylacetonitrile HO O O O O CN Cyanogenic glycoside which occurs in bitter almonds and seeds of apricots and peaches 14 AN The nucleophilic addition to C=O bond IV ACID-catalyzed nucleophilic addition 15 AN Addition of alcohols: HEMIACETALS ACID-catalyzed nucleophilic addition 16 AN Addition of alcohols : ACETALS 17 AN Addition of thiols: THIOACETALS 18 AN(E) Addition of derivatives of ammonia 19 AN(E) Addition of 1° and 2° amines: IMINES and ENAMINES ACID-catalyzed addition followed by elimination of water 20 AN(E) Addition of 1° amines: IMINES ACID-catalyzed addition followed by elimination of water 21 AN(E) Addition of hydroxylamine: OXIMES I ACID-catalyzed addition followed by elimination of water 22 AN(E) Addition of hydroxylamine: OXIMES II 23 AN(E) HYDRAZONES WOLFF-KISHNER reduction NH2-NH2 + C=O NH2-N=C KOH / DEG 180 °C CH2 + N2 24 AN(E) ENAMINES I mostly used 2° amines 25 AN(E) Addition of 2° amines: ENAMINES II the first step: ADDITION 26 AN(E) Addition of 2° amines: ENAMINES III the second step: acid-catalyzed ELIMINATION 27 AE(E) ENAMINES IV enamines as nucleophilic substrates 28 AE(E) ENAMINES V enamines as nucleophilic substrates : ACYLATION 29 AE(E) ENAMINES VI enamines as nucleophilic substrates : ALKYLATION 30 AE(E) ENAMINES VII enamines as nucleophilic substrates : ALKYLATION 31 AN Addition of phosphorus YLIDES I the WITTIG reaction 32 AN Addition of phosphorus YLIDES II preparation of phosphorus ylides: (R-X = CH3, 1° and 2° alkyl halides) 33 AN Addition of phosphorus YLIDES III a mechanism of WITTIG reaction 34 Oxidation of aldehydes and ketones I 35 Oxidation of aldehydes and ketones II the BAYER-VILLIGER oxidation (H>Ph>3°>2°>1°>CH3) 36 AN , AN(E) Reactions of carbonyl compounds I 37 AN Reactions of carbonyl compounds II 38 AN Reactions of carbonyl compounds III 39 Reactions of carbonyl compounds IV 40