* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download nucleophilic addition
Discodermolide wikipedia , lookup
Metal carbonyl wikipedia , lookup
Kinetic resolution wikipedia , lookup
Elias James Corey wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Aldol reaction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Hydroformylation wikipedia , lookup
Asymmetric induction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
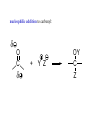
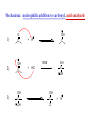
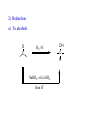
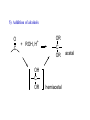
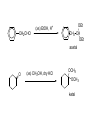
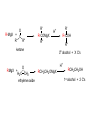
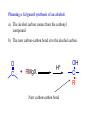
REACTIONS OF ALDEHYDES AND KETONES BY HARJEET GUJRAL ASSOCIATE PROFESSOR GOVT. COLLEGE ROPAR Reactions of aldehydes and ketones: oxidation reduction nucleophilic addition 1) Aldehydes are easily oxidized, ketones are not. 2) Aldehydes are more reactive in nucleophilic additions than ketones. alkane alcohol reduction reduction aldehyde ketone oxidation carboxylic acid addition product nucleophilic addition nucleophilic addition to carbonyl: O C + Y Z OY C Z Mechanism: nucleophilic addition to carbonyl 1) O C 2) O C Z Z O C Z Y OY C Z RDS + + Mechanism: nucleophilic addition to carbonyl, acid catalyzed 1) O C 2) OH C 3) OH C ZH + OH C H OH C ZH RDS + HZ OH C Z + H Aldehydes & ketones, reactions: 1) Oxidation 2) Reduction 3) Addition of cyanide 4) Addition of derivatives of ammonia 5) Addition of alcohols 6) Cannizzaro reaction 7) Addition of Grignard reagents 8) (Alpha-halogenation of ketones) 9) (Addition of carbanions) 1) Oxidation a) Aldehydes (very easily oxidized!) CH3CH2CH2CH=O + KMnO4, etc. CH3CH2CH2COOH carboxylic acid CH3CH2CH2CH=O + Ag+ CH3CH2CH2COO- + Ag Silver mirror Tollen’s test for easily oxidized compounds like aldehydes. (AgNO3, NH4OH(aq)) Ketones only oxidize under vigorous conditions via the enol. O + KMnO4 NR Cyclohexanone O + KMnO4, heat HOOCCH2CH2CH2CH2COOH adipic acid OH enol b) Methyl ketones: R O C CH3 + OI- R O C O- + CHI3 iodoform Yellow ppt test for methyl ketones O CH3CH2CH2CCH3 2-pentanone + (xs) NaOI CH3CH2CH2CO2- + CHI3 2) Reduction: a) To alcohols O C H2, Ni NaBH4 or LiAlH4 then H+ OH C H H2, Pt H O OH cyclopentanol cyclopentanone O C CH3 acetophenone 1. NaBH4 2. H+ OH CHCH3 1-phenylethanol O H2, Pt C H CH2OH benzaldehyde CH3 CH3CHCH=O isobutyraldehyde benzyl alcohol LiAlH4 H+ CH3 CH3CHCH2OH isobutyl alcohol hydride reduction mechanism: nucleophilic addition; nucleophile = hydride 1) 2) O C O C H RDS H: Al + + Al O C H + Al H C O Al Then + H+ alcohol Reduction b) To hydrocarbons O C NH2NH2, OH- O C Zn(Hg), HCl CH2 Wolff-Kishner Clemmensen CH2 O Cl + O AlCl3 Zn(Hg), HCl n-pentylbenzene cannot be made by Friedel-Crafts alkylation due to rearrangement of carbocation 3) Addition of cyanide O C 1. CN- OH C CN cyanohydrin 2. H+ + O + NaCN; then H OH CN mechanism for addition of cyanide nucleophilic addition 1) 2) O C O C C N RDS + C N + Na+ O C C N ONa C C N then + H+ Cyanohydrins have two functional groups plus one additional carbon. Nitriles can be hydrolyzed to carboxylic acids in acid or base: OH CH2CH C N H2O, OH- OH CH2CH C N H2O, H+ heat heat OH CH2CH COO- C C COOH H H 4) Addition of derivatives of ammonia O + H2N G (H+) + H2O N G O H2N NH2 H2N OH hydrazine hydroxylamine NH2 H2N N H semicarbazide O2N H2N HN H2N HN NO2 phenylhydrazine 2,4-dinitrophenylhydrazine acid catalyzed nucleophilic addition mechanism followed by dehydration 1) 2) 3) O C OH C OH C NH2 G + OH C H+ OH C NH2 G + H2N G C N + G RDS H2O + H+ CH2 CHO + H2NOH CH2 CH NOH hydroxylamine an oxime phenylacetaldehyde H+ O O + H2NHNCNH2 O NHNCNH2 semicarbazide cyclohexanone CH3CH2CH2CH2CHO pentanal a semicarbazone + NH2 NH phenylhydrazine CH3CH2CH2CH2CH N NH a phenylhydrazone 5) Addition of alcohols O C + ROH, H+ OH C OR OR C OR hemiacetal acetal Mechanism = nucleophilic addition, acid catalyzed 1) O C + OH C H RDS 2) 3) OH2 C OH C HOR + ROH OH C OR OH C HOR + H (xs) EtOH, H+ CH2CHO OEt CH2 CH OEt acetal O (xs) CH3OH, dry HCl OCH3 OCH3 ketal 6) Cannizzaro reaction. (self oxidation/reduction) a reaction of aldehydes without α-hydrogens COO- CH2OH CHO conc. NaOH + Br Br conc. NaOH H2C=O CH3OH + HCOO- Br Formaldehyde is the most easily oxidized aldehyde. When mixed with another aldehyde that doesn’t have any alphahydrogens and conc. NaOH, all of the formaldehyde is oxidized and all of the other aldehyde is reduced. Crossed Cannizzaro: CH=O CH2OH + H2C=O conc. NaOH + HCOO- OCH3 OH vanillin OCH3 OH 7) Addition of Grignard reagents. O C + RMgX O MgBr + H2O C R O MgBr C R OH + Mg(OH)Br C R larger alcohol mechanism = nucleophilic addition 1) O C 2) O C R RDS + RMgBr + MgBr O C R + MgBr OMgBr C R #3 synthesis of alcohols. Used to build larger molecules from smaller organic compounds. RMgX + O C H H formaldehyde RMgX + O C R' H other aldehydes RCH2OMgX R'CHOMgX R H+ RCH2OH 1o alcohol + 1 C H+ R'CHOH R 2o alcohol + X C's O R-MgX + C R' R" R' R-COMgX R" ketone O RMgX + H2C CH2 ethylene oxide R' H+ R-COH R" 3o alcohol + X C's H+ RCH2CH2OMgX RCH2CH2OH 1o alcohol + 2 C's Planning a Grignard synthesis of an alcohol: a) The alcohol carbon comes from the carbonyl compound. b) The new carbon-carbon bond is to the alcohol carbon. O C + + RMgX H New carbon-carbon bond OH C R “The Grignard Song” (sung to the tune of “America the Beautiful”) Harry Wasserman The carbonyl is polarized, the carbon end is plus. A nucleophile will thus attack the carbon nucleus. The Grignard yields an alcohol of types there are but three. It makes a bond that corresponds from “C” to shining “C.”