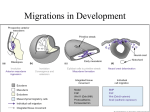
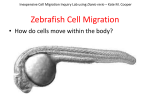
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Expression and function of cell adhesion molecules during neural
Chromatophore wikipedia , lookup
Cytokinesis wikipedia , lookup
Cell growth wikipedia , lookup
Tissue engineering wikipedia , lookup
Signal transduction wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Extracellular matrix wikipedia , lookup
Minerva Access is the Institutional Repository of The University of Melbourne Author/s: MCKEOWN, SONJA; ANDERSON, RICHARD; WALLACE, ADAM Title: Expression and function of cell adhesion molecules during neural crest migration Date: 2013-01-15 Citation: McKeown, SJ; Wallace, AS; Anderson, RB, Expression and function of cell adhesion molecules during neural crest migration, DEVELOPMENTAL BIOLOGY, 2013, 373 (2), pp. 244 - 257 (14) Persistent Link: http://hdl.handle.net/11343/43826 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Expression and function of cell adhesion molecules during neural crest migration Sonja J. McKeown1, Adam S. Wallace and Richard B. Anderson Department of Anatomy and Neuroscience, University of Melbourne, 3010, VIC, Australia. 1 Author for correspondence Sonja McKeown Department of Anatomy and Neuroscience University of Melbourne VIC 3010 Australia [email protected] Phone: +61383445637 Fax: +61393475219 Abstract Neural crest cells are highly migratory cells that give rise to many derivatives 19 including peripheral ganglia, craniofacial structures and melanocytes. Neural crest 20 cells migrate along defined pathways to their target sites, interacting with each other 21 and their environment as they migrate. Cell adhesion molecules are critical during 22 this process. In this review we discuss the expression and function of cell adhesion 23 molecules during the process of neural crest migration, in particular cadherins, 24 integrins, members of the immunoglobulin superfamily of cell adhesion molecules, 25 and the proteolytic enzymes that cleave these cell adhesion molecules. 26 expression and function of these cell adhesion molecules and proteases are compared 27 across neural crest emigrating from different axial levels, and across different species 28 of vertebrates. 29 30 31 32 33 34 Keywords Neural crest, cell adhesion, migration, mouse, chick, Xenopus 1 The 35 Introduction 36 37 Neural crest (NC) cells migrate extensively before giving rise to a variety of cell 38 types. NC cell-cell and cell-matrix interactions are an essential part of the migration 39 process. In fact, NC cell migration depends on optimal levels of adhesion, as too little 40 or too much adhesion result in migration defects (Anderson et al., 2006b). In this 41 review, we describe how the immunoglobulin superfamily of cell adhesion molecules 42 (IgCAMs), cadherins and integrins, and the proteolytic enzymes that cleave them, 43 regulate the migration of NC cells arising from different regions of the neural axis. 44 45 NC cells form at the junction of the neural plate and epidermal ectoderm during 46 neurulation. The formation of the NC requires multiple signaling pathways and 47 transcription factors to establish a pool of pre-migratory neural crest cells at the dorsal 48 most regions of the neural folds (Betancur et al., 2010). Once NC cells have formed, 49 they undergo an epithelial-mesenchymal transition (EMT) in order to migrate away 50 from the neural tube. They begin migration from the dorsal part of the neural tube 51 prior to, during or after the closure of the neural tube, depending on the axial level and 52 species. Cell adhesion molecules play roles in NC EMT, migration and also in their 53 aggregation at their final destination, but in this review we focus primarily on the 54 expression and function of cell adhesion molecules during NC migration. Changes in 55 cell-cell adhesion, cell-matrix adhesion and the cell cytoskeleton during EMT have 56 been reviewed recently (Clay and Halloran, 2011; Taneyhill, 2008; Thiery et al., 57 2009). 58 59 NC cells from different anteroposterior axial levels follow different migratory 60 pathways and give rise to different derivatives (Le Douarin and Kalcheim, 1999); Fig. 61 1). The main sub-divisions of the neural crest are cranial, vagal and trunk. In this 62 review, we discuss the role of cell adhesion molecules in regulating the migration of 63 each of these NC populations. The expression and function of the main cell adhesion 64 molecules are summarized in Table 1. 65 66 67 68 2 69 Cranial Neural Crest 70 71 Cranial NC cells give rise to much of the cartilage and bone of the skull and face, as 72 well as other connective tissues, and contribute to neurons and glia in cranial ganglia. 73 The cranial NC arise from the midbrain to somite 5 and migrate underneath the 74 ectoderm in a ventral direction to populate the frontonasal process, branchial arches, 75 and the trigeminal and epibranchial ganglia. While there is some intermingling of NC 76 cells arising from different anteroposterior levels (Kulesa and Fraser, 1998), NC cells 77 from particular axial levels follow pathways to populate corresponding facial 78 processes (Lumsden et al., 1991; Sadaghiani and Thiebaud, 1987; Trainor et al., 79 2002). Particular cranial regions express inhibitory molecules and repel NC cells, 80 narrowing the migratory pathways of NC from certain axial levels (Farlie et al., 1999; 81 Golding et al., 2004). The earliest migrating cells populate the facial processes and 82 give rise to mesenchymal derivatives, while later migrating NC cells remain in more 83 dorsal regions and contribute to cranial ganglia (Baker et al., 1997). 84 85 In contrast to other vertebrate species, the initial migration of cranial NC in Xenopus 86 is a collective migration, followed by a wave of single cell migration (Alfandari et al., 87 2003). This collective migration process involves contact inhibition of locomotion in 88 which protrusions of NC cells collapse when they contact another NC cell (Carmona- 89 Fontaine et al., 2008), and collective chemotaxis, mediated by the chemokine SDF1 90 (Theveneau et al., 2010). In other vertebrates, such as such as chick, time-lapse 91 imaging revealed that many NC cells migrate to the branchial arches in dynamic 92 chains (Kulesa and Fraser, 1998; 2000). Within these chains, the NC cells are in 93 nearly constant contact with each other over short and long distances via lamellipodia 94 or filopodia (Teddy and Kulesa, 2004). 95 exchange cytoplasmic material through thin cellular bridges (McKinney et al., 2011). 96 Cell-cell adhesion is involved in each of these modes of migration, and while N- 97 cadherin has been implicated in contact inhibition of locomotion in Xenopus, the 98 molecular mechanisms involved in chain migration have not yet been identified 99 (Alfandari et al., 2010; Teddy and Kulesa, 2004; Theveneau et al., 2010). 100 101 102 3 Moreover, some migrating cranial NC 103 Cadherins 104 105 The cadherin superfamily is divided into subfamilies, and of these, type I and type II 106 classical cadherins, and also protocadherins have been implicated in NC development 107 and migration. These cadherins are transmembrane molecules that extracellularly 108 undergo primarily homophilic binding to mediate cell-cell adhesion (Gumbiner, 109 2005). 110 cadherins promote stronger adhesiveness than type II cadherins (Chu et al., 2006). In 111 addition, some cadherins are also able to bind other molecules extracellularly 112 (Gumbiner, 2005). Intracellularly, classical cadherins interact with catenins, while 113 protocadherins can interact with Fyn kinases, each of which have signaling roles and 114 various effects on cell behavior (Gumbiner, 2005). The role of cadherins in mediating 115 cell adhesion is complex and can be regulated at many levels, both intracellularly and 116 extracellularly (Halbleib and Nelson, 2006). Cadherins are involved in NC EMT, 117 migration of cranial NC and the aggregation of cranial ganglia. The strength of the cell-cell adhesion differs between cadherins: type I 118 119 Cadherin 6B, a type II cadherin, is expressed by avian premigratory NC (Nakagawa 120 and Takeichi, 1995), and is transcriptionally downregulated by Snail2 prior to 121 emigration (Taneyhill et al., 2007). Cadherin-6B plays a role in the EMT undertaken 122 as NC begin to migrate (Coles et al., 2007; Taneyhill, 2008). 123 morpholino of cadherin-6B in chick embryos in ovo promoted premature and 124 increased migration of the cranial NC, while overexpression reduced the number of 125 migrating cranial NC and increased ectopic NC in the neural tube lumen in vivo, but 126 not in vitro (Coles et al., 2007). These findings are different from the effect of 127 manipulation of cadherin-6B in trunk NC (see below). Inhibition by 128 129 Expression of the type I N-cadherin by migrating avian cranial NC in vivo has not 130 been detected using antibody labeling (Bronner-Fraser et al., 1992b; Duband et al., 131 1988), however, it has been detected by qPCR (McLennan et al., 2012). A possible 132 explanation for this discrepancy is that processing for immunofluorescence may only 133 allow visualization of N-cadherin associated with adherens junctions, and N-cadherin 134 in migrating NC in vitro is mostly soluble and not associated with cell-cell contacts 135 (Monier-Gavelle and Duband, 1995). In contrast, during early cranial NC migration 136 in Xenopus embryos N-cadherin mRNA and protein can be detected (Theveneau et 4 137 al., 2010), and both overexpression and morpholino knockdown of N-cadherin in vivo 138 block cranial NC emigration (Theveneau et al., 2010). In vitro studies demonstrated a 139 role for N-cadherin during cranial NC collective migration in Xenopus. Function 140 blocking antibodies to N-cadherin prevented contact inhibition of locomotion and 141 collective polarization of cranial NC cells toward an SDF1 gradient (Theveneau et al., 142 2010). Interestingly, cranial NC cells treated with an N-cadherin morpholino were 143 highly motile in culture, and dispersed more rapidly than control cells (Theveneau et 144 al., 2010). This behaviour is similar to migratory cardiac NC from N-cadherin null 145 mutant mice, which have increased speed and persistence of movement in vitro, but 146 decreased directionality (Xu et al., 2001). A model of Xenopus cranial NC migration 147 has been proposed, in which N-cadherin-mediated cell adhesion causes contact- 148 dependent cell polarity, and when combined with a chemotactic signal, allows 149 collective chemotaxis of cranial NC (Theveneau et al., 2010). 150 intracellular trafficking of N-cadherin to the cell membrane is regulated in a subset of 151 cranial NC by the Wnt target gene, Ovo1, most likely through activity of Rab proteins 152 (Piloto and Schilling, 2010). However loss of functional N-cadherin in the zebrafish 153 pac mutant does not appear to perturb migration of cranial NC (Lele et al., 2002), and 154 the impact of loss of function of N-cadherin in migrating NC in vivo in Xenopus is not 155 yet known. In zebrafish, 156 157 Cadherin 11, a type II cadherin, is expressed by migrating cranial NC in mouse 158 (Kimura et al., 1995) and Xenopus (Hadeball et al., 1998; Vallin et al., 1998). Mice 159 homozygous for a mutant non-adhesive cadherin 11 have shorter frontal bones, but no 160 reported cranial NC migration defects (Manabe et al., 2000), whereas inhibition of 161 cadherin 11 in Xenopus in vivo revealed an important role in cranial NC migration 162 (Borchers et al., 2001; Kashef et al., 2009). Knockdown of cadherin 11 inhibits NC 163 migration into the second and more caudal branchial arches only, and mutant cells do 164 not form filopodia or lamellipodia (Kashef et al., 2009). In vivo rescue with mutated 165 versions of cadherin 11 showed that expression of the cytoplasmic tail is sufficient for 166 Xenopus cranial NC migration as long as it is linked to the plasma membrane. The E- 167 catenin binding site and a binding site for the guanine exchange factor Trio are critical 168 intracellular domains; as over-expression of human cadherin 11, Trio or constitutively 169 active small Rho GTPases rescued the loss of cadherin 11 in vivo, whereas over- 5 170 expression of mouse cadherin 6 was unable to rescue migration (Kashef et al., 2009). 171 Furthermore, cleavage of the extracellular domain of Xenopus cadherin 11 is 172 necessary for cranial NC migration (see below). Thus cadherin-11 appears to act 173 together with E-catenin and Trio at the plasma membrane to initiate protrusive activity 174 and drive cranial NC migration through the activation of small Rho GTPases. 175 Interestingly, Trio can have multiple cell surface protein partners (Backer et al., 2007; 176 Briancon-Marjollet et al., 2008; Yano et al., 2011), but whether it normally binds to 177 partners other than cadherin 11 in Xenopus cranial NC is not known. Xenopus cranial 178 NC also express a proto-cadherin named PCNS (Rangarajan et al., 2006). 179 Knockdown of PCNS by morpholino inhibits cranial NC migration in vivo and the 180 cells also fail to migrate and instead disaggregrate when cultured on fibronectin 181 (Rangarajan et al., 2006). The exact function of PCNS in cranial NC migration 182 remains to be determined, however it has been suggested that PCNS may influence 183 planar cell polarity signalling, which is required for contact inhibition of locomotion 184 in Xenopus cranial NC migration (Alfandari et al., 2010). 185 186 Avian cadherin 7, murine cadherin 6 and rat cadherin 19 are related type II cadherins 187 that are expressed by migrating cranial NC (Inoue et al., 1997; Nakagawa and 188 Takeichi, 1995; Takahashi and Osumi, 2005); however, a functional role has not yet 189 been established, and mice null for cadherin 6 are viable with no reported cranial NC 190 defects (Mah et al., 2000). Cadherin 7 expression has recently been shown to be 191 expressed at higher levels in trailing avian migratory cranial NC than in the leading 192 migratory NC in vivo (McLennan et al., 2012), suggesting cell-cell adhesion may be 193 more important for following rather than leading cells. Using transfected cell lines, 194 cadherin 7 has been shown to have higher adhesivity compared to cadherin 11, and 195 both type II cadherins have much lower adhesivity compared to N-cadherin (Chu et 196 al., 2006). 197 198 N-cadherin is expressed by chick placodal cells but not cranial NC during the 199 aggregation of cranial sensory ganglia (Shiau and Bronner-Fraser, 2009). Blocking 200 N-cadherin translation in trigeminal placode cells with morpholinos in ovo led to 201 misshapen ganglia and decreased aggregation of the placode cells (Shiau and 202 Bronner-Fraser, 2009). The cell-surface expression of N-cadherin on placode cells 6 203 was regulated post-translationally by cranial NC-mediated Slit1 activation of Robo2 204 expressed by placode cells (Shiau and Bronner-Fraser, 2009). 205 206 Integrins 207 Integrins are heterodimeric transmembrane receptors composed of alpha and beta 208 subunits, which bind extracellularly to a variety of extracellular matrix molecules and 209 other ligands, depending on the combination of subunits (van der Flier and 210 Sonnenberg, 2001). Integrins are regulated on many levels; their affinity for ligands 211 can be reversibly altered by conformational changes, which can be mediated by 212 intracellular or extracellular factors (Hynes, 2002; Luo et al., 2007). Trafficking of 213 integrins and distribution of integrins at the cell membrane can also affect the level of 214 cell adhesion (Margadant et al., 2011; Ulrich and Heisenberg, 2009). Furthermore, 215 integrin-ligand interactions can result in intracellular changes including activation of 216 signal transduction pathways, regulating survival, proliferation and gene expression 217 (Hynes, 2002; Margadant et al., 2011). 218 219 Cranial NC express a variety of integrins, some of which are restricted to specific 220 axial levels, and some to particular species. The integrins that are expressed by 221 migrating cranial NC in multiple species are the E1 (Bronner-Fraser, 1985; Gawantka 222 et al., 1992; Krotoski and Bronner-Fraser, 1986; Pietri et al., 2004; Ransom et al., 223 1993) and D5 subunits (Alfandari et al., 2003; Bajanca et al., 2004; Joos et al., 1995; 224 Wang and Burke, 1995). Integrin D5E1 mediates migration of cranial NC from 225 multiple species on fibronectin in vitro (Alfandari et al., 2003; Kil et al., 1998; Lallier 226 and Bronner-Fraser, 1992). Inhibiting the function of E1 integrins in chick in ovo 227 resulted in reduced cranial NC migration and an ectopic buildup of NC cells in the 228 neural tube (Boucaut et al., 1984; Bronner-Fraser, 1985; 1986). Surprisingly, mouse 229 conditional knockout studies of E1 integrin using the Wnt1-Cre line showed migration 230 of cranial NC cells to the branchial arches (Turlo et al., 2012), however levels of 231 proliferation and apoptosis were not assessed. Moreover, in mice null for fibronectin, 232 cranial NC cells also still migrate to the branchial arches, but the number of cranial 233 NC cells is reduced due to increased apoptosis and decreased proliferation (Mittal et 7 234 al., 2010). Similarly, in integrin D5 null mice, apoptosis occurs in NC cells migrating 235 to the second branchial arch (Goh et al., 1997; Mittal et al., 2010). This suggests that 236 fibronectin and integrin D5 are critical for the survival of cranial NC, rather than 237 migration. Furthermore, the restricted regional effect of loss of D5 suggests that there 238 is compensation by other integrins rostral to the second arch. Indeed, integrin D4 is a 239 candidate whose expression is restricted to NC rostral to the second branchial arch 240 (Pinco et al., 2001). Exposure to D4 function-blocking antibodies in mouse cultures 241 inhibited cranial NC migration (Kil et al., 1998), however at least some cranial NC 242 migration is normal in D4 null mice, as the trigeminal ganglia are present (Yang et al., 243 1995). In contrast to the axially restricted expression of D4 in mouse, chick cranial 244 NC express D4 at all axial levels (Kil et al., 1998), while Xenopus cranial NC do not 245 appear to express D4 (Whittaker and Desimone, 1998). The expression of E3, D6 and 246 Dv by cranial NC also shows species differences (Alfandari et al., 2003; Joos et al., 247 1998; Lallier et al., 1996; Pietri et al., 2003; Ransom et al., 1993; Wang and Burke, 248 1995), but the significance of these differences is not yet clear. 249 250 While it may be surprising that loss of fibronectin or E1 integrin in mice does not 251 appear to prevent cranial NC migration, many other extracellular matrix molecules are 252 present within the cranial pathway. Chick cranial and trunk NC regulated surface 253 integrin expression, location and activity in different ways in response to different 254 concentrations and types of extracellular matrix substrates in vitro. 255 concentrations of laminin, cranial NC migrated faster than trunk NC (Strachan and 256 Condic, 2003), by modulating surface integrin expression in a manner involving 257 integrin receptor recycling (Strachan and Condic, 2004). In contrast, on fibronectin, 258 receptor recycling did not occur (Strachan and Condic, 2004). Cranial NC reduced 259 surface expression of D4 integrin on high concentrations of fibronectin, whereas trunk 260 NC did not (Strachan and Condic, 2003). Migration on fibronectin was regulated by 261 the activation of integrins, and interestingly cranial and trunk NC responded to 262 integrin activation in different ways depending on the concentration of the fibronectin 263 substrate (Strachan and Condic, 2008). Integrin activation of cranial NC on low 264 concentrations of fibronectin increased avidity (integrin clustering), and on high 265 concentrations of fibronectin increased affinity (Strachan and Condic, 2008). 8 On high In 266 contrast, in trunk NC, integrin activation on low concentrations of fibronectin 267 increased affinity, and on high concentrations of fibronectin increased avidity. In 268 each of these cases, integrin activation slowed migration on fibronectin, but not on 269 laminin (Strachan and Condic, 2008). These differences highlight the complexity of 270 integrin function and regulation, and demonstrate that the NC are capable not only of 271 regulation of integrin activity in response to the extracellular environment, but also 272 that NC at different axial levels have different responses and regulate integrin 273 function and activity differently. 274 275 Matrix Metalloproteases 276 277 Matrix metalloproteases (MMPs and members of the ADAM family) cleave a variety 278 of proteins, including extracellular matrix molecules, and cell surface molecules such 279 as cadherins, integrins and IgCAMs (Edwards et al., 2008; VanSaun and Matrisian, 280 2006). The proteolysis of cell adhesion molecules can disrupt cell-cell adhesion and 281 also release soluble extracellular or intracellular fragments that can have further 282 signaling effects (Edwards et al., 2008; VanSaun and Matrisian, 2006). In addition, 283 ADAMs contain a disintegrin loop which interacts with integrins to influence cell 284 adhesion and migration (Huang et al., 2005), and the cytoplasmic domain of ADAMs 285 can interact with a variety of proteins to affect catalytic activity, trafficking and cell 286 signaling events (Edwards et al., 2008). MMPs and ADAMs have been implicated in 287 regulating NC EMT and migration. ADAM13 and 19 are expressed in Xenopus 288 cranial NC prior to migration, and early knockdown using morpholinos to ADAM13 289 (in Xenopus tropicalis, but not Xenopus laevis), or ADAM19, reduces the expression 290 of cranial NC markers (Cousin et al., 2011; Neuner et al., 2009; Wei et al., 2010), and 291 inhibits NC migration (Cousin et al., 2011; Neuner et al., 2009). Moreover, knocking 292 down ADAM13 and 19 together inhibited NC migration to a greater extent than 293 knockdown of either gene alone (Cousin et al., 2011), suggesting that although they 294 have some distinct functions during NC development, there is also some 295 compensation between these genes (Cousin et al., 2011; Wei et al., 2010). 296 297 ADAM13 continues to be expressed by cranial NC during migration in Xenopus 298 (Alfandari et al., 1997) and is expressed in chick cranial NC pathways (Lin et al., 299 2007). ADAM33 is also expressed by migrating avian cranial NC (McLennan et al., 9 300 2012), and ADAM9 and ADAM11 by Xenopus cranial NC (Cai et al., 1998). 301 Multiple ADAMs (ADAM10, 12, 15, 17, 19 and 33) are expressed by cranial NC or 302 along the cranial NC migratory pathway in developing mouse embryos (Tomczuk, 303 2004). Blocking metalloprotease activity inhibited mouse cranial NC migration on 304 fibronectin (Tomczuk, 2004); however the role of specific ADAMs or MMPs during 305 mouse cranial NC migration is not yet known. 306 ADAM13 in Xenopus in vivo resulted in a large mass of NC cells near the neural tube 307 (Alfandari et al., 2001). Expression of a protease defective ADAM13, which acts as a 308 dominant negative, reduced migration of NC in the second and more caudal branchial 309 arch streams (Alfandari et al., 2001), a similar phenotype to the knockdown of 310 cadherin-11 (Kashef et al., 2009). Co-immunoprecipitation and Western blotting 311 demonstrated that ADAM13 or ADAM9 cleaves cadherin-11, and inhibiting ADAM 312 activity blocks cleavage of cadherin-11 in vivo (McCusker et al., 2009). Moreover, 313 increased expression of ADAM13 can rescue the inhibition of NC migration caused 314 by an over-expression of cadherin-11 (McCusker et al., 2009); demonstrating that 315 ADAM13 mediated cleavage of cadherin-11 is required for normal cranial NC 316 migration. Cleavage of the extracellular domain of cadherin-11 in Xenopus cranial 317 NC by ADAM13 reduces cell-cell adhesion directly, and the cleaved domain is then 318 able to bind to other full-length cadherin-11 molecules, further reducing cell-cell 319 adhesion and promoting migration (McCusker et al., 2009). 320 cadherin-11 by morpholino is not sufficient alone to rescue a loss of ADAM13 in vivo 321 (Cousin et al., 2012), demonstrating that ADAM13 has multiple functions in 322 mediating the migration of cranial NC. ADAM13 can also cleave fibronectin, and by 323 binding to fibronectin domains, promotes cell adhesion via E1 integrins (Gaultier et 324 al., 2002). Manual separation of the cranial NC from the ectoderm and mesoderm in 325 vivo partially rescues the loss of ADAM13 (Cousin et al., 2012), suggesting that 326 cleavage or modification of extracellular matrix is important in allowing NC 327 migration. Moreover, the loss of ADAM13 alone or both ADAM13 and ADAM19 328 together did not affect the migration of cranial NC in vitro. ADAM13 needs to be 329 expressed by at least some cranial NC in order to allow normal migration of the 330 population, and these data suggest that ADAM13 promotes migration partly by 331 modifying the extracellular environment through which the cranial NC migrate 332 (Cousin et al., 2012). 10 Over-expression of full-length However, reducing 333 334 ADAM13 lacking the cytoplasmic domain is unable to rescue the inhibition of NC 335 migration due to ADAM13 morpholino (Cousin et al., 2011). 336 domain regulates function by both interacting with the adaptor protein PACSIN2, 337 (Cousin et al., 2000) and by altering the subcellular localisation or activity of the 338 protease domain after cleavage (Cousin et al., 2011). Cleavage of the cytoplasmic 339 domain by J-secretase during NC development allows the translocation of the 340 cytoplasmic domain to the nucleus, altering the expression of multiple genes 341 including Calpain8-a, a member of a protease family linked to cell migration (Cousin 342 et al., 2011). Expression of both the extracellular domain of cadherin-11 combined 343 with the cytoplasmic domain of ADAM13 is sufficient to rescue a loss of migration 344 caused by combined ADAM13 and ADAM19 morpholino in vivo (Cousin et al., 345 2012). 346 multiple mechanisms; both extracellular cleavage of molecules, and regulation of 347 transcription via its cytoplasmic domain. The cytoplasmic Therefore, ADAM13 promotes Xenopus cranial NC migration through 348 349 Expression or function of ADAMs in particular subsets or stages of NC migration 350 may influence the ability of NC cells to migrate to particular regions. For example, 351 ADAM10 cleavage of a splice variant of the cell adhesion molecule CD44, CD44v6, 352 appears to facilitate migration of NC into the cornea in chick embryos (Huh et al., 353 2007). 354 355 MMP9 is expressed by avian NC cells during EMT and migration, and also by some 356 surrounding cells (Monsonego-Ornan et al., 2012). Inhibition of MMP9 activity 357 reduces the number of cells emigrating from the neural tube at cranial levels, while 358 overexpression, or MMP9 conditioned media, increases the number of migratory 359 cranial NC in vitro and in vivo (Monsonego-Ornan et al., 2012). Thus MMP9 has a 360 role in the onset of migration, possibly through degradation of the laminin basement 361 membrane. Later inhibition of MMP9 in neural tube explants reduced the distance 362 migrated by NC cells on the substrate, while MMP9 conditioned media significantly 363 increased the number of emigrating cranial NC (Monsonego-Ornan et al., 2012). 364 Migrating NC cells in these explants also demonstrated perturbations in the 365 organization of stress fibres, and rapid loss of N-cadherin expression, suggesting N- 11 366 cadherin may also be a substrate of MMP9. In contrast to the phenotype in chick, 367 MMP9 knockout mice do not display overt embryonic defects (Vu et al., 1998), 368 perhaps due to redundancy with other members of the MMP family. MMP2, which is 369 closely related to MMP9, is expressed by, or along, the cranial NC migratory pathway 370 in chick, mouse and Xenopus, with some differences in expression between species 371 (Cai et al., 2000; Robbins et al., 1999; Tomlinson et al., 2009). MMP2 in avian 372 cranial NC is expressed at higher levels in leading compared to trailing cells 373 (McLennan et al., 2012). In addition, MMP8 is expressed in mouse cranial NC 374 (Giambernardi et al., 2001), and MMP14 in a subset of Xenopus NC migrating from 375 rhombomeres 3 and 5 (Harrison et al., 2004). However the roles of these MMPs in 376 cranial NC migration have not been determined. 377 378 IgCAMs 379 380 Members of the IgCAM family interact extracellularly either homophilically or with 381 other proteins, and can exist as multiple isoforms due to alternative splicing. 382 IgCAMs interact with numerous intracellular binding partners, ranging from effectors 383 of signal-transduction pathways to cytoskeletal proteins. 384 cranial NC cells in the chick prior to and in the early stages of migration (Bronner- 385 Fraser et al., 1992b). NCAM mediated cell-cell adhesion is important for emigration 386 and early stages of cranial NC migration, but is not necessary once migration is 387 underway (Bronner-Fraser et al., 1992b). In Xenopus, NCAM is also expressed by 388 cranial NC prior to migration, and later during aggregation of cranial ganglia, but is 389 not expressed during NC migration (Balak et al., 1987). NCAM is detected on 390 391 Neuropilins and plexins can in some circumstances be involved in cell adhesion, 392 however during NC migration they are best understood for their role in acting as 393 receptors for the Ig domain containing Semaphorin molecules, which often act as 394 chemorepellants or chemoattractants in the guidance of neural crest at multiple axial 395 levels (reviewed in Schwarz and Ruhrberg, 2010). No other IgCAMs are known to 396 play a role in the migration of cranial NC. 397 398 399 12 400 Vagal Neural Crest 401 402 Vagal NC cells, which arise adjacent to somites 1-7, include the most caudal cranial 403 NC cells and the most rostral trunk NC cells. Vagal NC cells give rise to two distinct 404 populations of cells, cardiac NC and enteric NC cells. 405 406 Cardiac NC Cells 407 408 Cardiac NC cells, which arise adjacent to somites 1-3, migrate beneath the ectoderm 409 into branchial arches 3, 4, and 6, where they contribute to the great arteries, the 410 aorticopulmonary septum, the conotruncal cushions in the outflow tract and cardiac 411 ganglia of the developing heart (Kirby and Waldo, 1995; Waldo et al., 1998). Defects 412 in cardiac NC migration result in severe cardiac defects involving outflow tract 413 septation anomalies, such as persistent truncus arteriosus as well as various aortic arch 414 anomalies (Kirby and Hutson, 2010). A number of cell adhesion molecules, including 415 cadherins and integrins, as well as the proteolytic enzymes that cleave them, MMPs 416 and ADAMs, play a role in cardiac NC migration and formation of the developing 417 heart. 418 419 Cadherins 420 421 Cardiac NC cells express cadherin-6, cadherin-7 and caherin-11, as they migrate to 422 the heart. Avian cadherin-6B is expressed at the beginning of migration but is then 423 rapidly down-regulated, while avian cadherin-7 and murine cadherin-6 and murine 424 and Xenopus cadherin-11 are expressed throughout migration (Inoue et al., 1997; 425 Kimura et al., 1995; Nakagawa and Takeichi, 1995; Vallin et al., 1998). Over- 426 expression of these cadherins prevents NC emigration from the neural tube, while 427 down regulation of cadherin-6B results in premature NC emigration (Coles et al., 428 2007; McCusker et al., 2009; Nakagawa and Takeichi, 1998). 429 430 The main cadherin involved in cardiac NC migration appears to be N-cadherin. 431 Murine N-cadherin is weakly expressed during cardiac NC migration in vitro (Xu et 432 al., 2001) and at regions of cell-cell contact by cardiac NC arriving at their destination 433 in vivo (Luo et al., 2006). N-cadherin is dramatically up-regulated as the cells 13 434 condense in the outflow tract, and is required for the remodeling of the cardiac 435 outflow tract (Luo et al., 2006). In the absence of N-cadherin, mouse cardiac NC 436 cells migrate faster in vitro, but exhibit a loss of directionality resulting in an overall 437 decrease in the distance migrated (Xu et al., 2001). Despite this, cardiac NC cells 438 from N-cadherin deficient mice do reach the cardiac outflow tract, but are unable to 439 undergo the normal morphogenetic changes, resulting in persistent truncus arteriosus, 440 and in a thinning of the ventricular myocardium (Luo et al., 2006). 441 442 Connexin 43 (Cx43D1) also plays a role in cardiac NC migration by interacting with 443 N-cadherin (Xu et al., 2001) and mediating interactions with signaling pathways that 444 regulate polarized cell movement (Xu et al., 2006). In N-cadherin deficient mice, 445 cardiac NC cells display a reduced level of gap junction communication that is similar 446 to Cx43D1 deficient NC cells (Xu et al., 2001). Cx43D1 deficient mice die at birth 447 due to severe pulmonary outflow tract obstruction (Reaume et al., 1995). Loss of 448 Cx43D1 inhibits cardiac NC cell migration, resulting in fewer cells reaching the 449 outflow tract, while over-expression of Cx43D1 enhanced migration, resulting in 450 more cardiac NC cells reaching the outflow tract (Huang et al., 1998). Like N- 451 cadherin deficient cells, Cx43D1 deficient cardiac NC cells also show reduced 452 directionality (Xu et al., 2001). Interactions between N-cadherin and Cx43D1 may be 453 modulated through p120-catenin signaling (Xu et al., 2001). Although Cx43D1 forms 454 gap junctions, cell-cell communication via gap junctions does not appear to modulate 455 cardiac NC motility (Xu et al., 2006). Instead, Cx43D1 co-localises with many actin 456 binding proteins and is hypothesized to play an important role in regulating the actin 457 cytoskeleton during migration (Xu et al., 2006). 458 459 Integrins 460 Cardiac NC cells in Aves express multiple integrins, including the Dand D subunits 461 (Kil et al., 1998; Wang and Burke, 1995). The D5 and E1 subunits have also been 462 detected in mice (Goh et al., 1997; Xu et al., 2006), and murine cardiac NC synthesise 463 fibronectin as they migrate (Mittal et al., 2010). In D5 integrin null or conditional 464 fibronectin null mutant mice, migration of cardiac NC cells to the heart still occurs 465 (Goh et al., 1997; Mittal et al., 2010), but there is a reduction in the number of cardiac 14 466 NC and increased apoptosis, suggesting that, like more rostral cranial NC, D5 integrin 467 and fibronectin are required for the proliferation of cardiac NC progenitors, and 468 proliferation and survival of cardiac NC during migration (Mittal et al., 2010). 469 470 1 integrins are up-regulated as avian cardiac NC cells emigrate from the neural tube 471 in vivo (Duband et al., 1986). Exposure to 1 integrin function blocking antibodies 472 results in a reduction in the overall speed and directionality of mouse cardiac NC cells 473 in vitro (Xu et al., 2006). Although there are no detectable defects in cardiac NC 474 migration in conditional Wnt1-Cre E1 integrin null mice, cardiac NC did not undergo 475 vascular remodeling, resulting in embryonic lethality (Turlo et al., 2012). Similar 476 cardiac defects and early death are also observed in mice where E1 integrins are 477 eliminated prior to NC emigration from the neural tube (Pietri et al., 2004). 478 479 MMPs 480 481 In order for cardiac NC cells to migrate through the extracellular matrix, they require 482 the proteolytic activity of MMPs and/or ADAMs. Migrating avian cardiac NC cells 483 express MMP2, MMP14 and tissue inhibitor of metalloproteinase (TIMP)-2 484 (Cantemir et al., 2004). Similarly to MMP2 at cranial levels, TIMP-2 is expressed at 485 higher levels in leading compared to trailing cells (Cantemir et al., 2004). Use of anti- 486 sense TIMP-2 oligonucleotides in chick resulted in perturbed cardiac NC formation 487 and decreased NC cell migration away from the neural tube in ovo, while over- 488 expression of TIMP-2 increased cardiac neural cell motility in vitro (Cantemir et al., 489 2004). TIMP-2 is required for activation of proMMP-2 by MMP14 (Cantemir et al., 490 2004). MMP-2 is expressed by avian cardiac NC in vitro (Cantemir et al., 2004), and 491 within the substrate through which cardiac NC cells migrate in vivo (Cai et al., 2000). 492 Pharmacological inhibition of MMP-2 in Aves decreases the distance cardiac NC 493 cells migrate in vitro and in vivo, decreases motility and increases cell area and cell 494 perimeter (Cai and Brauer, 2002). However, the migration of cardiac NC cells is not 495 affected in MMP-2 or TIMP-2 null mutant mice (Caterina et al., 2000), suggesting 496 that MMP-2 and TIMP-2 are either not involved in cardiac NC migration in mice in 497 vivo, or that there is functional redundancy with other matrix metalloproteases. 498 15 499 Cardiac NC cells also express ADAM19 in mice (Komatsu et al., 2007), ADAM13 in 500 chick (Lin et al., 2007) and ADAM9, ADAM11 and ADAM13 in Xenopus (Alfandari 501 et al., 2001; Cai et al., 1998). The migration of cardiac NC cells is unaffected in 502 ADAM19-deficient mice (Komatsu et al., 2007). 503 cardiac NC cells are unable to fuse the left and right ridges of the endocardial cushion, 504 resulting in defects in the outflow tract and formation of the ventricular septum 505 (Komatsu et al., 2007). However, ADAM19-deficient 506 507 Enteric NC Cells 508 509 Enteric NC-derived cells arise from two levels of the neural tube. The largest 510 contribution of cells originates from the vagal level (adjacent to somites 1-7) (Le 511 Douarin and Teillet, 1973; Yntema and Hammond, 1954), which overlaps with the 512 region that gives rise to cardiac NC. A second, smaller, population of cells originates 513 from the sacral level (caudal to somite 28) and contributes some neurons to the colon 514 in chick and mouse (Anderson et al., 2006a; Burns and Le Douarin, 1998; Kapur, 515 2000; Wang et al., 2011). Defects in vagal NC migration result in variable lengths of 516 the distal gastrointestinal tract lacking enteric neurons; a condition in humans called 517 Hirschsprung disease (HSCR) or congenital aganglionosis. Most of the expression 518 and functional studies of cell adhesion in enteric NC have been carried out using 519 mice, with some data also available from Aves. 520 521 Cadherins 522 523 Enteric NC cells in mice express N-cadherin, Cadherin-11, Cadherin 9 and Cadherin- 524 6 as well as Protocadherins D1, D4, D10 and 15 (Breau et al., 2006; Heanue and 525 Pachnis, 2006; Vohra et al., 2006). Mice null for protocadherin 15 do not display any 526 defects in the structure of the enteric nervous system (Vohra et al., 2006). In contrast 527 to the early down regulation of Cadherin 6B observed in avian vagal NC cells 528 (Nakagawa and Takeichi, 1995), murine Cadherin-6 expression is maintained in 529 enteric NC cells as they colonise the entire length of the gut (Breau et al., 2006). 530 Mouse and avian enteric NC express N-cadherin at low levels during migration, 531 which is up-regulated during aggregation into ganglia (Hackett-Jones et al., 2011). 16 532 Conditional loss of N-cadherin in NC in Ht-PA-Cre mice results in a transient delay in 533 colonization of the gut, with mutant enteric NC exhibiting perturbed directionality 534 compared to control enteric NC and loss of the normal chain-like pattern of migration 535 (Broders-Bondon et al., 2012). However, the delayed migration did not result in 536 intestinal aganglionosis in the mutant mice. Double conditional mouse knockouts of 537 N-cadherin and E1 integrin resulted in a more severe phenotype and intestinal 538 aganglionosis (see below). 539 540 Integrins 541 542 Enteric murine and rat NC cells express several integrins, including the 4, 5, 6, 543 v, 1, E3 and E5 subunits (Bixby et al., 2002; Breau et al., 2009; Breau et al., 2006; 544 Iwashita et al., 2003). The migration of enteric NC cells is impaired in 1 integrin 545 conditional null mutant mice, resulting in absence of enteric ganglia from the distal 546 colon (Breau et al., 2006; Pietri et al., 2003). The absence of 1 integrin also results 547 in an abnormal organization of the enteric ganglia network (Breau et al., 2006; Pietri 548 et al., 2003), with larger aggregations and more space between ganglia. Time-lapse 549 imaging analysis revealed that enteric NC cells lacking 1 integrin migrated more 550 slowly and with less persistence than control cells, most notably around the time of 551 invasion of the caecum and hindgut (Breau et al., 2009). Fibronectin and tenascin-C 552 are expressed at high levels in the caecum and proximal hindgut relative to the 553 midgut. In vitro assays demonstrated that tenascin-C inhibited both adhesion and 554 migration of enteric NC, with a stronger effect on E1 integrin null cells. In contrast, 555 fibronectin stimulated adhesion and migration of control enteric NC, but not integrin 556 E1-null cells. Thus E1 integrins appear to be necessary for fibronectin-mediated 557 migration in the caecum and proximal hindgut, and to overcome the tenascin-C 558 mediated inhibition of migration in these regions (Breau et al., 2009). 559 560 Double conditional mouse knockout of E1 integrin and N-cadherin caused a more 561 severe intestinal aganglionosis than the single E1 integrin mutation, suggesting there 562 is co-operation between these two adhesion molecules during enteric NC migration 563 (Broders-Bondon et al., 2012). However, the abnormal ganglia network observed in 564 E1 integrin mutants was partially rescued in these double mutants. Therefore, the 17 565 organization of the network of enteric ganglia requires a balance of cell-adhesion and 566 extracellular matrix adhesion, modulated by the activity of N-cadherin and E1 567 integrins. 568 569 Integrin E1 is also involved in the migration of chick enteric NC along endothelial 570 cells (Nagy et al., 2009). In chick and zebrafish, there is contact between enteric NC 571 and blood vessels along the gut (Nagy et al., 2009). Pharmacological inhibition of 572 endothelial cell development in cultured avian gut resulted in cessation of enteric NC 573 migration and distal aganglionosis. However, there may be a species difference in 574 this pattern of migration: although enteric NC are reported to be adjacent to blood 575 vessels in chick, in mice they are not co-localised (Young et al., 2004). 576 577 The integrin 2-like gene and integrin DE1 are also expressed by murine enteric NC 578 cells (Iwashita et al., 2003; Vohra et al., 2006), but their role in migration is not yet 579 known. 580 581 MMPs 582 583 Enteric NC cells migrate in chains (Young et al., 2004). However, in order to 584 advance, the cells must transiently weaken adhesion to other enteric NC cells. MMP2 585 (Anderson et al., 2006b), ADAM17 and ADAMTS1 (Iwashita et al., 2003) are all 586 expressed by mouse enteric NC. The expression of MMPs by enteric NC-derived 587 cells permits individual migrating cells to break away from chains (Anderson et al., 588 2006b). Broad spectum inhibitors to gelatinases MMP 2 and 9 retarded enteric NC 589 migration in explants of mouse gut, as did a specific MMP2 inhibitor (Anderson, 590 2010). Furthermore, the topography of the network was different in that fewer, 591 thicker chains were present following inhibition of MMP2 and 9. 592 593 IgCAMs 594 595 NCAM is expressed by all enteric NC cells in mice (Heanue and Pachnis, 2006; 596 Vohra et al., 2006) and Aves, and expression of NCAM increases following neuronal 597 differentiation (Hackett-Jones et al., 2011). PSA modification of NCAM regulates 18 598 nerve fibre fasciculation and is involved in the clustering of individual enteric NC 599 cells into enteric ganglia. Excessive PSA modification induced by exposure to BMP4 600 reduced the distance migrated by mouse NC cells in gut organ culture (Fu et al., 601 2006). 602 603 Like NCAM, L1CAM is also expressed by most enteric NC cells in mice (Anderson 604 et al., 2006b) and in Aves by all enteric NC at the wavefront (Hackett-Jones et al., 605 2011; Nagy et al., 2012). Perturbation studies, using function blocking antibodies and 606 recombinant protein, showed that disrupting L1 activity delays enteric NC cell 607 migration and increases the presence of solitary NC cells close to the migratory 608 wavefront within explants of mouse gut (Anderson et al., 2006b). The migration of 609 enteric NC cells within the developing gut is also delayed in L1cam deficient mice, 610 although no change in the number of solitary cells was reported (Anderson et al., 611 2006b). Subsequently L1cam has been shown to act as a modifier gene for three 612 HSCR associated genes, Sox10, Et3 and Ednrb, in mice, as interactions between these 613 genes result in a significant increase in the penetrance and/or severity of intestinal 614 aganglionosis in mutant mice (Wallace et al., 2010; Wallace et al., 2011). L1CAM 615 can be cleaved by ADAM10 and ADAM17, which may regulate L1-dependent cell 616 adhesion (Maretzky et al., 2005), and while both L1CAM and ADAM17 are 617 expressed by enteric NC in mice, it is not yet known if this occurs during enteric NC 618 migration. 619 620 Trunk Neural Crest 621 622 Trunk NC cells give rise to neurons and glia in dorsal root ganglia and sympathetic 623 ganglia, Schwann cells, boundary cap cells, cells of the adrenal medulla and 624 melanocytes (Le Douarin and Kalcheim, 1999; Serbedzija et al., 1994; Weston, 1963). 625 Time lapse imaging of migrating trunk NC cells in chick embryos has revealed that 626 they maintain close, extensive and dynamic contact with other NC cells and many 627 migrate in chains through the somites (Kasemeier-Kulesa et al., 2005). 628 629 630 631 19 632 Cadherins 633 634 Both N-cadherin and cadherin 6B have been shown to be involved in avian trunk NC 635 EMT, with quite different roles. Cadherin 6B is expressed in premigratory NC and 636 during EMT, but is absent in NC migrating away from the neural tube similarly to 637 other axial levels (Nakagawa and Takeichi, 1995; Park and Gumbiner, 2010). 638 Inhibition or knockdown of cadherin 6B in avian trunk NC caused a decrease in 639 migrating NC cells, while overexpression increased the number of cells ectopically 640 localised in the neural tube lumen (Park and Gumbiner, 2010). This effect of cadherin 641 6B in the de-epithelialization of the trunk NC is mediated by non-canonical BMP 642 signaling via the BMP type II receptor, involving LIM kinase 1 which phosphorylates 643 cofilin to regulate actin dynamics (Park and Gumbiner, 2012). While overexpression 644 of cadherin 6B at both cranial and trunk levels promoted the ectopic localization of 645 NC cells in the neural tube lumen, inhibition or knockdown had opposing effects on 646 NC EMT. This may be due to differences in methodology, or may reflect inherent 647 differences between cranial and trunk NC, similarly to the requirement for G1-S cell 648 cycle transition, which is necessary for trunk but not cranial NC EMT (Burstyn- 649 Cohen et al., 2004; Theveneau et al., 2007). 650 651 N-cadherin mRNA is expressed in the chick dorsal neural tube prior to and at the time 652 of neural crest migration (Shoval et al., 2007). Overexpression many hours prior to 653 NC emigration in ovo of full-length N-cadherin, but not dominant-negative forms, 654 causes inhibition of trunk EMT (Shoval et al., 2007). N-cadherin can be cleaved by 655 ADAM10, and a second cleavage step intracellularly yields a soluble cytoplasmic tail 656 termed CTF2, which increases cytosolic E-catenin and translocates to the nucleus to 657 regulate gene expression (Reiss et al., 2005). Blocking the action of ADAM10 in 658 vitro inhibits chick trunk NC EMT, which can be rescued by transfection of CTF2 659 (Shoval et al., 2007). Electroporation of the CTF2 fragment in ovo increased EMT, 660 and E-catenin and cyclin D1 expression (Shoval et al., 2007). These experiments 661 suggest that during trunk NC EMT ADAM10 and N-cadherin function in cell 662 signaling and regulation of gene transcription through the action of CTF2/E-catenin. 663 Consistent with this, E-catenin is localized to the nucleus at the onset of migration in 664 avian trunk NC in vitro, but during migration, it is associated with N-cadherin at 20 However, loss of E-catenin in 665 intercellular contacts (de Melker et al., 2004). 666 premigratory neural crest in Wnt1-cre mice does not inhibit NC EMT, but NC-derived 667 melanocytes and dorsal root ganglia fail to develop, suggesting involvement of E- 668 catenin signaling in differentiation of specific lineages rather than EMT or migration 669 (Hari et al., 2002). Interestingly, overstimulation of E-catenin in avian trunk NC in 670 vitro promoted translocation to the nucleus and inhibited EMT and NC migration, by 671 decreasing integrin-mediated cell-matrix adhesion and reduced proliferation (de 672 Melker et al., 2004), showing cross-talk between the actions of different cell adhesion 673 and signaling pathways. 674 675 In contrast to enteric NC, N-cadherin protein does not appear to be expressed during 676 avian trunk NC migration in vivo; expression of N-cadherin is later observed after 677 cells coalesce to form dorsal root and sympathetic ganglia (Akitaya and Bronner- 678 Fraser, 1992; Duband et al., 1988). However, during migration in vitro, avian trunk 679 NC cells retain N-cadherin expression, although it is constitutively phosphorylated 680 and is not associated with the cytoskeleton, such that N-cadherin mediated cell-cell 681 contacts are not stable (Monier-Gavelle and Duband, 1995). 682 location of N-cadherin and stability of N-cadherin mediated cell-cell interactions were 683 modified by the activity or inhibition of protein kinases and phosphatases, but the 684 phosphorylation of N-cadherin was not affected by these protein kinases and 685 phosphatases (Monier-Gavelle and Duband, 1995). Moreover, the localisation of N- 686 cadherin in migrating avian NC in vitro is regulated by E1 and E3 integrin signalling 687 (Monier-Gavelle and Duband, 1997). 688 adhesion increased N-cadherin mediated intercellular adhesion in vitro (de Melker et 689 al., 2004). 690 mRNA. Recent in vivo studies showed that NC cells over-expressing full-length N- 691 cadherin migrated more slowly, as did NC cells expressing a dominant-negative, 692 catenin-binding domain (Kasemeier-Kulesa et al., 2006). These N-cadherin over- 693 expressing NC cells also displayed altered morphology: increased levels of N- 694 cadherin increased the number of filopodia, while those expressing dominant-negative 695 N-cadherin had fewer filopodia. However, the N-cadherin over-expressing NC cells 696 were still able to migrate ventrally to the sympathetic ganglia (Kasemeier-Kulesa et 697 al., 2006). The intracellular Decreasing integrin-mediated cell-matrix It is not yet known if migrating trunk NC in vivo express N-cadherin In contrast, N-cadherin expressing sarcoma cells failed to migrate 21 698 following transplantation into the avian trunk NC pathway in ovo, in contrast to the 699 parental cell line and cadherin 7 expressing cells (Dufour et al., 1999). 700 differing observations suggests that in trunk NC, high levels of N-cadherin expression 701 may be regulated such that they do not completely inhibit migration. Furthermore, 702 the similar effect of the full-length and dominant-negative N-cadherin on slowing but 703 not abolishing migration further support the notion that increased or decreased levels 704 of cell-cell adhesion slow neural crest migration, and shows similarities to the slowed 705 migration of mouse enteric NC with a conditional loss of N-cadherin (see above). These 706 707 Cadherin 6 is expressed in mouse NC cells prior to and during migration (Inoue et al., 708 1997). In Aves, cadherin 7 is expressed by the majority of trunk NC migrating in the 709 ventral pathway to the sympathetic and dorsal root ganglia, and in the dorsolateral 710 pathway (Nakagawa and Takeichi, 1995; 1998). 711 expression of cadherin 7 in a large subpopulation slows the migration of these cells, 712 keeping them together and helping to mediate cell sorting into discrete locations and 713 cell types (Nakagawa and Takeichi, 1995), however recent observations of cranial NC 714 suggest cadherin 7 is expressed at higher levels in following rather than leading or 715 pioneer cells (McLennan et al., 2012). Over-expression of cadherin 7 inhibited the 716 emigration of some NC cells and caused ectopic localisation in the neural tube, in the 717 same manner as over-expression of N-cadherin or cadherin 6B (Nakagawa and 718 Takeichi, 1998; Park and Gumbiner, 2010), which is likely a general effect of 719 increased cell-cell adhesion. Expression of cadherin 7 by sarcoma cells did not inhibit 720 migration of these cells following transplantation to the avian trunk NC pathway 721 (Dufour et al., 1999). The cell-cell interactions between these cadherin 7-expressing 722 sarcoma cells in vitro were transient and more rapidly turned over than N-cadherin 723 mediated interactions (Dufour et al., 1999). Furthermore, the cadherin 7 and N- 724 cadherin mediated adhesion had different effects on integrin-dependent signaling and 725 consequent behavior of the cells on fibronectin (Dufour et al., 1999). Cadherin 11 is 726 expressed the early migrating trunk avian NC and in the mesenchyme through which 727 the NC migrate (Chalpe et al., 2010). The location of these cadherin 11-positive cells 728 suggests that the most ventrally migrating trunk NC express cadherin 11, and that it is 729 only weakly or not expressed in later migrating NC. As trunk NC aggregate into 730 dorsal root ganglia, they only very weakly express cadherin 11, while the surrounding 731 mesenchyme is strongly positive (Chalpe et al., 2010). Both cadherin 7 and cadherin 22 It has been hypothesised that 732 11 are upregulated in avian trunk NC in vitro in response to Wnt3a, at both mRNA 733 level and protein at cell-cell adhesions (Chalpe et al., 2010), suggesting these 734 adhesion molecules are regulated by Wnt signalling. 735 Xenopus trunk neural crest migrating in the dorsal fin (Vallin et al., 1998), and 736 cadherin 19 is expressed in a subpopulation of trunk NC migrating in the ventral 737 pathway in rat (Takahashi and Osumi, 2005). Overall, many questions remain to be 738 answered on the role of type II cadherins during the migration of trunk NC. Cadherin 11 is expressed by 739 740 In contrast to the expression of cadherins by NC, truncated cadherin (T-cadherin), an 741 atypical cadherin which lacks cytoplasmic sequences, is expressed in the caudal half 742 of each somite, which trunk NC avoid (Ranscht and Bronner-Fraser, 1991). NC 743 avoidance of the caudal half-somite has been shown to be mediated by semaphorin- 744 neuropilin signaling (Gammill and Roffers-Agarwal, 2010), and a role for T-cadherin 745 in NC migration through the rostral somite has not yet been demonstrated. 746 747 Later expression of N-cadherin is required for the aggregation of NC cells in the chick 748 sympathetic ganglia. Inhibition of N-cadherin in trunk NC by function blocking 749 antibodies or dominant-negative constructs resulted in abnormal elongation and 750 increased size of the sympathetic ganglia, while over-expression of N-cadherin 751 decreased the size of the sympathetic ganglia but not length (Kasemeier-Kulesa et al., 752 2006). These findings demonstrate the role of N-cadherin in mediating aggregation of 753 the ganglia and consequently ganglia size following migration. 754 755 Integrins 756 757 The importance of integrin signalling in NC migration has been demonstrated by 758 manipulations of a scaffolding protein within the integrin signalling pathway, Nedd9 759 (Aquino et al., 2009). Loss or gain of function of Nedd9 decreased or increased 760 respectively the extent of chick trunk NC migration. The expression and function of 761 integrins in the migration of trunk NC has primarily been studied in cultures of avian 762 trunk NC. These studies have shown that many integrins are expressed by trunk NC 763 in vitro: D1E1, D3E1, D4E1, D5E1, D6E1, D7E1, D8E1, DvE1, DvE3, DvE5, and a E8 764 (Delannet et al., 1994; Desban and Duband, 1997; Kil and Bronner-Fraser, 1996; 23 765 Testaz et al., 1999), however the pattern of expression of most integrins in vivo 766 remains to be determined. The expression of integrins E1, D1, D4, D6 and D7 have, 767 however, been examined in vivo in Aves (Bronner-Fraser et al., 1992a; Duband et al., 768 1992; Kil and Bronner-Fraser, 1996; Kil et al., 1998; Krotoski et al., 1986; Stepp et 769 al., 1994), and some differences have been observed between expression of integrins 770 in vitro and in vivo. Integrin D1 is expressed weakly in vivo (Duband et al., 1992), but 771 strong expression is reported in vitro (Desban and Duband, 1997). Other integrins 772 show dynamic changes of expression in vivo. 773 subpopulation of migrating trunk NC (Kil and Bronner-Fraser, 1996), while integrin 774 D6 is absent from many cells during migration in vivo (Bronner-Fraser et al., 1992a), 775 and is expressed in vitro by a subpopulation of NC, in which it is mostly cytoplasmic 776 (Bronner-Fraser et al., 1992a; Desban and Duband, 1997). During gangliogenesis, 777 integrin D7 is expressed in the dorsal root ganglia, while D6 is expressed in the 778 sympathetic ganglia (Kil and Bronner-Fraser, 1996). Many integrins appear to be 779 widely distributed over the cell surface of cultured NC; however integrins DvE1, D1 780 and E3 also showed localisation to focal contacts (Delannet et al., 1994; Desban and 781 Duband, 1997; Testaz et al., 1999). The integrins expressed by trunk NC mediate 782 migration on multiple substrates, including fibronectin, laminin and vitronectin, all of 783 which are present along the pathways on which NC migrate in vivo and can support 784 migration in vitro (Duband and Thiery, 1987; Krotoski and Bronner-Fraser, 1990; 785 Newgreen and Thiery, 1980; Rovasio et al., 1983). Integrin D7 is observed on a 786 787 The function of different integrins in mediating migration of avian trunk NC has been 788 mostly addressed by culturing trunk NC on different substrates and using function- 789 blocking antibodies or antisense oligonucleotides to perturb particular integrins. 790 These experiments have revealed that different integrins regulate specific aspects of 791 cell behaviour: some integrins play a role in mediating migration of the NC on 792 particular substrates, while others are involved in adhesion or spreading of the NC and 793 some do not appear to mediate any of these functions. The integrins implicated in 794 migration include: integrin DvE3 on fibronectin (Testaz et al., 1999), vitronectin 795 (Delannet et al., 1994) and laminin-1 (Desban and Duband, 1997), DvE5 on 796 vitronectin (Delannet et al., 1994), and the E1 integrin subunit can pair with D4, D8 or 797 D3 to mediate migration on fibronectin (Lallier and Bronner-Fraser, 1993; Testaz et 24 798 al., 1999), or with D1 on laminin-1 (Desban and Duband, 1997; Desban et al., 2006; 799 Lallier and Bronner-Fraser, 1993). 800 801 Knocking out D1 or E3 integrins in vivo in mice does not cause an abnormal neural 802 crest migration phenotype (Gardner et al., 1996; Hodivala-Dilke et al., 1999), which 803 may reflect functional redundancy (see below). The roles of D4, D5 and E1 subunits 804 have been complicated by the early embryonic lethality of these knockouts (Fassler 805 and Meyer, 1995; Goh et al., 1997; Yang et al., 1995). Some cells lacking E1 integrin 806 can migrate to NC derivatives in chimaeric E1 null mice or following antisense 807 blocking of E1 in chick (Fassler and Meyer, 1995; Tucker, 2004). However it is 808 possible that these E1 integrin null cells were able to migrate due to the presence of 809 wild-type E1 integrin expressing cells. Ht-PA-Cre conditional E1 null mice show 810 normal trunk NC migration, but these cells still retain a low level of E1 integrin 811 expression during NC migration (Pietri et al., 2004). Wnt1-Cre conditional E1 null 812 mice also showed migration of trunk NC; however the extent of migration and 813 number of migratory cells was not examined (Turlo et al., 2012). Integrins D4 and D5 814 are expressed by mouse trunk NC (Goh et al., 1997; Kil et al., 1998), and in D4 null 815 mice, trunk NC migration and early gangliogenesis appear normal (Yang et al., 1995), 816 however D5 null mice die during trunk NC migration (Goh et al., 1997). To study the 817 importance of D4 or D5 integrins, NC null for either integrin were examined in 818 culture, and also grafted into chick NC pathways. Integrin D5 null NC cells migrated 819 poorly on fibronectin, but well on vitronectin, whereas D4 null cells showed normal 820 migration on fibronectin (Haack and Hynes, 2001). This is in contrast to in vitro 821 studies, which show an inhibition of migration using function blocking antibodies to 822 D4 in chick and mouse (Kil et al., 1998; Testaz and Duband, 2001), and no effect on 823 migration using function blocking antibodies to D5 (Testaz et al., 1999). When 824 grafted into chick embryos, many D4-null glial progenitor cells underwent apoptosis 825 during migration, especially those migrating dorsally, however some were able to 826 migrate and contribute to the peripheral nervous system (Haack and Hynes, 2001). 827 Blocking D4E1 integrin in cultured chick NC also results in a large increase in 828 apoptosis (Testaz and Duband, 2001). Integrin D5 null cells migrate well in chick NC 829 pathways, although the cells did not survive in the glial lineage (Haack and Hynes, 25 830 2001). Although there are some discrepancies in the behavior of null cells compared 831 to function-blocking antibody studies, overall the data suggest that a lack of integrin 832 D5 alone does not impair trunk NC migration and integrin D4 may play a role in 833 mediating NC survival. 834 835 In summary, trunk NC express a wide variety of integrins during migration, and 836 multiple substrates are present along NC pathways in vivo. There is likely to be 837 significant functional redundancy as the loss of single integrin subunits does not 838 reduce migration in vivo, and blocking of multiple integrins is required to completely 839 prevent NC migration on fibronectin: either blocking of both E1 and DvE3, or D3, D4 840 and Dv combined (Testaz et al., 1999). Combined knockdown studies of multiple 841 integrins in vivo, particularly E1 and Dv, may be necessary to gain a greater 842 understanding of the role of integrins during trunk NC migration. 843 complicating the analysis of integrin function is cross-talk that occurs between 844 different integrins, by which integrins can regulate the functioning and expression of 845 other integrins (Retta et al., 2001; Testaz and Duband, 2001). In addition, in vitro 846 work using avian NC demonstrated regulation of integrin activity in response to 847 different extracellular environments (see Cranial Neural Crest section above), with 848 responses differing between cranial and trunk NC (Strachan and Condic, 2003; 2004; 849 2008). Lastly, expression of particular integrin subunits, D4 and D5, may be required 850 for cell survival rather than, or in addition to, mediating migration, highlighting the 851 importance of integrin-mediated signaling during NC development. Further 852 853 MMPs 854 855 ADAM10 is expressed in the dorsal neural tube in the avian trunk, and in the 856 ectoderm, dermatome and myotome, and in trunk NC in culture (Hall and Erickson, 857 2003). ADAM10 expression in the dorsal neural tube cleaves N-cadherin and plays a 858 role in regulating NC EMT as described above (Shoval et al., 2007); however roles 859 for ADAM family members in regulating trunk NC migration have not been 860 demonstrated. 861 26 862 Multiple MMPs are expressed by migrating trunk NC, including MMP8 in mouse NC 863 (Giambernardi et al., 2001), MMP2 weakly in avian NC (Duong and Erickson, 2004) 864 or Xenopus mesenchyme (Tomlinson et al., 2009), MMP14 in migrating Xenopus NC 865 (Harrison et al., 2004), and MMP9 in avian premigratory and migratory NC 866 (Monsonego-Ornan et al., 2012). 867 differences between species; MMPs may be required for NC EMT in chick but not in 868 Xenopus. Chemical inhibition of MMP activity or knocking down MMP2 in chick by 869 morpholino prior to NC EMT reduced EMT and migration in vitro and in ovo, while 870 knockdown in NC cells just emerging from the neural tube did not inhibit their 871 migration (Duong and Erickson, 2004). Similarly, inhibition of MMP9 or use of 872 MMP9 morpholinos reduced the number of migrating trunk NC, while overexpression 873 or presence of MMP9 conditioned media increased the number of migratory cells and 874 promoted premature EMT of trunk NC in chick in culture and in ovo (Monsonego- 875 Ornan et al., 2012). This effect may be due to degradation of laminin in the basement 876 membrane (Monsonego-Ornan et al., 2012). 877 inhibition of either MMP2 or MMP14 or both in Xenopus perturbed the migration of 878 melanophores, but no effect on EMT was observed, nor was the migration of other 879 NC cells affected (Tomlinson et al., 2009). The function of these MMPs shows some In contrast, in vivo knockdown or 880 881 IgCAMs 882 883 NCAM is expressed in the neural tube, and remains in avian trunk NC cells as they 884 undergo EMT and begin to migrate, but is gradually down-regulated during migration 885 (Akitaya and Bronner-Fraser, 1992), before being later upregulated in the ganglia 886 (Duband et al., 1985; Lallier and Bronner-Fraser, 1988; Thiery et al., 1982). Whether 887 NCAM expression during the early phase of trunk NC migration is important is not 888 yet understood. In contrast to the downregulation of NCAM during NC migration, 889 Melanoma Cell Adhesion Molecule (MCAM, also known as gicerin, HEMCAM and 890 CD146) is expressed in migrating avian trunk NC (Alais et al., 2001). MCAM can 891 regulate adhesion by controlling expression of integrins (Alais et al., 2001), and is 892 upregulated during melanoma metastasis (Lehmann et al., 1987; Luca et al., 1993; Xie 893 et al., 1997), but its function in NC migration is unknown. 894 895 27 896 Conclusions 897 898 In summary, the process of cell migration requires interactions between the migrating 899 cells and the surrounding environment, consisting of neighboring cells and 900 extracellular matrix molecules. An optimal level of cell adhesion is required for 901 migration, as too little adhesion does not generate sufficient traction for motility, 902 while too much adhesion will result in the cells remaining stationary. The families of 903 cell adhesion molecules involved in NC migration, cadherins, integrins and the 904 immunoglobulin superfamily, regulate cell-cell and/or cell-matrix adhesion, and are 905 also important components of various cell signaling pathways that affect many 906 aspects of cell behavior including proliferation, cytoskeleton dynamics and gene 907 transcription. 908 intracellularly and extracellularly. 909 regulate the activity, membrane localization and trafficking, and hence cell adhesion 910 function of the adhesion molecules. Furthermore, activity of one family of adhesion 911 molecules can influence the function of other adhesion molecules, such as integrins 912 and cadherins. Extracellularly, cleavage by matrix metalloproteases can affect the 913 function of cell adhesion molecules, not only by reducing the level of adhesion, but 914 also by releasing the adhesion molecule for intracellular signaling functions, such as 915 the cleavage of N-cadherin by ADAM10 during trunk NC EMT. In addition, NC 916 cells can respond to differences in the extracellular environment to which they are 917 exposed by altering their activity, expression and/or localization of adhesion 918 molecules, such as integrins. 919 adhesion molecules during NC migration is complex and multifaceted. The function of the adhesion molecules can be regulated both Various intracellular signaling pathways can Consequently, the regulation and function of cell 920 921 Understanding how NC cells use cell adhesion molecules to migrate has important 922 implications for understanding the etiology of some neurocristopathies, and the 923 mechanisms neural crest-derived cancerous cells may use to migrate during 924 metastasis. 925 numerous cell adhesion molecules during neural crest migration has been elucidated. 926 There are, however, many gaps in our knowledge and understanding of the role of 927 these cell adhesion molecules during the process of migration, particularly in vivo. Over the past thirty years, the expression pattern and function of 928 28 929 Along the embryonic axis there are differences in the extracellular substrates and 930 pathways available to NC, differences in derivatives formed and even differences in 931 mechanisms of EMT. 932 mechanisms used by the NC to undergo EMT and migrate are likely to reflect this. 933 Some cell adhesion molecules appear to have similar functions at all axial levels, such 934 as MMP9, which, when inhibited in chick embryos causes a reduction in NC 935 migration at both cranial and trunk levels. Other cell adhesion molecules appear to 936 play a role at only one axial level, for example L1CAM, expressed by the enteric NC 937 in mice and chick. Loss or inhibition of L1CAM slows the colonization of the gut by 938 enteric NC in mice. In addition, NC from a single axial level migrate to multiple 939 target sites and give rise to different types of derivatives. Several cell adhesion 940 molecules, such as some integrins and cadherins, appear to be restricted to 941 subpopulations of NC within an axial level, but the function of these differences is not 942 yet clear. Recent data suggest that the level of expression of some integrins and 943 cadherins differs depending on whether the cell is a leading or following cell, that this 944 expression is plastic and likely reflects different mechanisms of migration between 945 leading and following cells (McLennan et al., 2012). There are both differences and 946 similarities in the cell adhesion molecules employed by the NC in different species. 947 Some of these variations may reflect different mechanisms by which NC cells use to 948 migrate in different species. Other variations may reflect different members of a gene 949 family playing a similar role in multiple species. Therefore, the cell adhesion molecules and molecular 950 951 Many of the studies carried out on NC cells have been conducted in vitro, particularly 952 with the integrins. This was done out of necessity, and these studies have been very 953 informative. However, there are limitations in the application of these results to NC 954 migration in vivo. Another complication to understanding the role of cell adhesion 955 molecules in vivo is functional redundancy or compensation between members of a 956 family, and in the case of integrins, cross talk between integrins expressed by a single 957 cell. These factors can make it very difficult to tease out the role of particular 958 molecules. Applying some of the newer technologies to these questions may provide 959 important insights. For example, the use of knockdown technologies, fluorescent 960 tagging of particular cell adhesion molecules or their intracellular partners, in vivo 961 imaging and other technologies have great potential for advances in this field, to give 29 962 us a much greater understanding of how these cell adhesion molecules function during 963 NC cell migration. 964 965 966 Acknowledgements 967 We thank Prof Heather Young for insightful comments and feedback on the 968 manuscript. SJM was supported by an NHMRC CJ Martin Fellowship (400433), and 969 RBA by an NHMRC CDA Fellowship (454773). 970 971 30 972 973 974 References 975 during initiation and cessation of neural crest cell migration. Dev Dyn 194, 12-20. 976 977 978 979 980 981 982 983 984 985 986 987 988 989 990 991 992 993 994 995 996 997 998 999 1000 1001 1002 1003 1004 1005 1006 1007 1008 1009 1010 1011 1012 1013 1014 1015 1016 1017 1018 1019 Akitaya, T. and Bronner-Fraser, M., 1992. Expression of cell adhesion molecules Alais, S., Allioli, N., Pujades, C., Duband, J. L., Vainio, O., Imhof, B. A. and Dunon, D., 2001. HEMCAM/CD146 downregulates cell surface expression of beta1 integrins. J Cell Sci 114, 1847-59. Alfandari, D., Cousin, H., Gaultier, A., Hoffstrom, B. G. and DeSimone, D. W., 2003. Integrin alpha5beta1 supports the migration of Xenopus cranial neural crest on fibronectin. Dev Biol 260, 449-64. Alfandari, D., Cousin, H., Gaultier, A., Smith, K., White, J. M., Darribere, T. and DeSimone, D. W., 2001. Xenopus ADAM 13 is a metalloprotease required for cranial neural crest-cell migration. Curr Biol 11, 918-30. Alfandari, D., Cousin, H. and Marsden, M., 2010. Mechanism of Xenopus cranial neural crest cell migration. Cell Adh Migr 4, 553-60. Alfandari, D., Wolfsberg, T. G., White, J. M. and DeSimone, D. W., 1997. ADAM 13: a novel ADAM expressed in somitic mesoderm and neural crest cells during Xenopus laevis development. Dev Biol 182, 314-30. Anderson, R. B., 2010. Matrix metalloproteinase-2 is involved in the migration and network formation of enteric neural crest-derived cells. Int J Dev Biol 54, 639. Anderson, R. B., Stewart, A. L. and Young, H. M., 2006a. Phenotypes of neural-crestderived cells in vagal and sacral pathways. Cell Tissue Res 323, 11-25. Anderson, R. B., Turner, K. N., Nikonenko, A. G., Hemperly, J., Schachner, M. and Young, H. M., 2006b. The cell adhesion molecule L1 is required for chain migration of neural crest cells in the developing mouse gut. Gastroenterology 130, 1221-32. Aquino, J. B., Lallemend, F., Marmigere, F., Adameyko, II, Golemis, E. A. and Ernfors, P., 2009. The retinoic acid inducible Cas-family signaling protein Nedd9 regulates neural crest cell migration by modulating adhesion and actin dynamics. Neuroscience 162, 1106-19. Backer, S., Hidalgo-Sanchez, M., Offner, N., Portales-Casamar, E., Debant, A., Fort, P., Gauthier-Rouviere, C. and Bloch-Gallego, E., 2007. Trio controls the mature organization of neuronal clusters in the hindbrain. J Neurosci 27, 10323-32. Bajanca, F., Luz, M., Duxson, M. J. and Thorsteinsdottir, S., 2004. Integrins in the mouse myotome: developmental changes and differences between the epaxial and hypaxial lineage. Dev Dyn 231, 402-15. Baker, C. V., Bronner-Fraser, M., Le Douarin, N. M. and Teillet, M. A., 1997. Earlyand late-migrating cranial neural crest cell populations have equivalent developmental potential in vivo. Development 124, 3077-87. Balak, K., Jacobson, M., Sunshine, J. and Rutishauser, U., 1987. Neural cell adhesion molecule expression in Xenopus embryos. Dev Biol 119, 540-50. Betancur, P., Bronner-Fraser, M. and Sauka-Spengler, T., 2010. Assembling neural crest regulatory circuits into a gene regulatory network. Annu Rev Cell Dev Biol 26, 581-603. Bixby, S., Kruger, G. M., Mosher, J. T., Joseph, N. M. and Morrison, S. J., 2002. Cell-intrinsic differences between stem cells from different regions of the 31 1020 1021 1022 1023 1024 1025 1026 1027 1028 1029 1030 1031 1032 1033 1034 1035 1036 1037 1038 1039 1040 1041 1042 1043 1044 1045 1046 1047 1048 1049 1050 1051 1052 1053 1054 1055 1056 1057 1058 1059 1060 1061 1062 1063 1064 1065 1066 1067 peripheral nervous system regulate the generation of neural diversity. Neuron 35, 643-56. Borchers, A., David, R. and Wedlich, D., 2001. Xenopus cadherin-11 restrains cranial neural crest migration and influences neural crest specification. Development 128, 3049-60. Boucaut, J. C., Darribere, T., Poole, T. J., Aoyama, H., Yamada, K. M. and Thiery, J. P., 1984. Biologically active synthetic peptides as probes of embryonic development: a competitive peptide inhibitor of fibronectin function inhibits gastrulation in amphibian embryos and neural crest cell migration in avian embryos. J Cell Biol 99, 1822-30. Breau, M. A., Dahmani, A., Broders-Bondon, F., Thiery, J. P. and Dufour, S., 2009. Beta1 integrins are required for the invasion of the caecum and proximal hindgut by enteric neural crest cells. Development 136, 2791-801. Breau, M. A., Pietri, T., Eder, O., Blanche, M., Brakebusch, C., Fassler, R., Thiery, J. P. and Dufour, S., 2006. Lack of beta1 integrins in enteric neural crest cells leads to a Hirschsprung-like phenotype. Development 133, 1725-34. Briancon-Marjollet, A., Ghogha, A., Nawabi, H., Triki, I., Auziol, C., Fromont, S., Piche, C., Enslen, H., Chebli, K., Cloutier, J. F., Castellani, V., Debant, A. and Lamarche-Vane, N., 2008. Trio mediates netrin-1-induced Rac1 activation in axon outgrowth and guidance. Mol Cell Biol 28, 2314-23. Broders-Bondon, F., Paul-Gilloteaux, P., Carlier, C., Radice, G. L. and Dufour, S., 2012. N-cadherin and beta1-integrins cooperate during the development of the enteric nervous system. Dev Biol 364, 178-91. Bronner-Fraser, M., 1985. Alterations in neural crest migration by a monoclonal antibody that affects cell adhesion. J Cell Biol 101, 610-7. Bronner-Fraser, M., 1986. An antibody to a receptor for fibronectin and laminin perturbs cranial neural crest development in vivo. Dev Biol 117, 528-36. Bronner-Fraser, M., Artinger, M., Muschler, J. and Horwitz, A. F., 1992a. Developmentally regulated expression of alpha 6 integrin in avian embryos. Development 115, 197-211. Bronner-Fraser, M., Wolf, J. J. and Murray, B. A., 1992b. Effects of antibodies against N-cadherin and N-CAM on the cranial neural crest and neural tube. Dev Biol 153, 291-301. Burns, A. J. and Le Douarin, N. M., 1998. The sacral neural crest contributes neurons and glia to the post- umbilical gut: spatiotemporal analysis of the development of the enteric nervous system. Development 125, 4335-47. Burstyn-Cohen, T., Stanleigh, J., Sela-Donenfeld, D. and Kalcheim, C., 2004. Canonical Wnt activity regulates trunk neural crest delamination linking BMP/noggin signaling with G1/S transition. Development 131, 5327-39. Cai, D. H. and Brauer, P. R., 2002. Synthetic matrix metalloproteinase inhibitor decreases early cardiac neural crest migration in chicken embryos. Dev Dyn 224, 441-9. Cai, D. H., Vollberg, T. M., Sr., Hahn-Dantona, E., Quigley, J. P. and Brauer, P. R., 2000. MMP-2 expression during early avian cardiac and neural crest morphogenesis. Anat Rec 259, 168-79. Cai, H., Kratzschmar, J., Alfandari, D., Hunnicutt, G. and Blobel, C. P., 1998. Neural crest-specific and general expression of distinct metalloprotease-disintegrins in early Xenopus laevis development. Dev Biol 204, 508-24. 32 1068 1069 1070 1071 1072 1073 1074 1075 1076 1077 1078 1079 1080 1081 1082 1083 1084 1085 1086 1087 1088 1089 1090 1091 1092 1093 1094 1095 1096 1097 1098 1099 1100 1101 1102 1103 1104 1105 1106 1107 1108 1109 1110 1111 1112 1113 1114 1115 1116 1117 Cantemir, V., Cai, D. H., Reedy, M. V. and Brauer, P. R., 2004. Tissue inhibitor of metalloproteinase-2 (TIMP-2) expression during cardiac neural crest cell migration and its role in proMMP-2 activation. Dev Dyn 231, 709-19. Carmona-Fontaine, C., Matthews, H. K., Kuriyama, S., Moreno, M., Dunn, G. A., Parsons, M., Stern, C. D. and Mayor, R., 2008. Contact inhibition of locomotion in vivo controls neural crest directional migration. Nature 456, 957-61. Caterina, J. J., Yamada, S., Caterina, N. C., Longenecker, G., Holmback, K., Shi, J., Yermovsky, A. E., Engler, J. A. and Birkedal-Hansen, H., 2000. Inactivating mutation of the mouse tissue inhibitor of metalloproteinases-2(Timp-2) gene alters proMMP-2 activation. J Biol Chem 275, 26416-22. Chalpe, A. J., Prasad, M., Henke, A. J. and Paulson, A. F., 2010. Regulation of cadherin expression in the chicken neural crest by the Wnt/beta-catenin signaling pathway. Cell Adh Migr 4, 431-8. Chu, Y. S., Eder, O., Thomas, W. A., Simcha, I., Pincet, F., Ben-Ze'ev, A., Perez, E., Thiery, J. P. and Dufour, S., 2006. Prototypical type I E-cadherin and type II cadherin-7 mediate very distinct adhesiveness through their extracellular domains. J Biol Chem 281, 2901-10. Clay, M. R. and Halloran, M. C., 2011. Regulation of cell adhesions and motility during initiation of neural crest migration. Curr Opin Neurobiol 21, 17-22. Coles, E. G., Taneyhill, L. A. and Bronner-Fraser, M., 2007. A critical role for Cadherin6B in regulating avian neural crest emigration. Dev Biol 312, 533-44. Cousin, H., Abbruzzese, G., Kerdavid, E., Gaultier, A. and Alfandari, D., 2011. Translocation of the cytoplasmic domain of ADAM13 to the nucleus is essential for Calpain8-a expression and cranial neural crest cell migration. Dev Cell 20, 256-63. Cousin, H., Abbruzzese, G., McCusker, C. and Alfandari, D., 2012. ADAM13 function is required in the 3 dimensional context of the embryo during cranial neural crest cell migration in Xenopus laevis. Dev Biol Cousin, H., Gaultier, A., Bleux, C., Darribere, T. and Alfandari, D., 2000. PACSIN2 is a regulator of the metalloprotease/disintegrin ADAM13. Dev Biol 227, 197210. de Melker, A. A., Desban, N. and Duband, J. L., 2004. Cellular localization and signaling activity of beta-catenin in migrating neural crest cells. Dev Dyn 230, 708-26. Delannet, M., Martin, F., Bossy, B., Cheresh, D. A., Reichardt, L. F. and Duband, J. L., 1994. Specific roles of the alpha V beta 1, alpha V beta 3 and alpha V beta 5 integrins in avian neural crest cell adhesion and migration on vitronectin. Development 120, 2687-702. Desban, N. and Duband, J. L., 1997. Avian neural crest cell migration on laminin: interaction of the alpha1beta1 integrin with distinct laminin-1 domains mediates different adhesive responses. J Cell Sci 110 ( Pt 21), 2729-44. Desban, N., Lissitzky, J. C., Rousselle, P. and Duband, J. L., 2006. alpha1beta1integrin engagement to distinct laminin-1 domains orchestrates spreading, migration and survival of neural crest cells through independent signaling pathways. J Cell Sci 119, 3206-18. Duband, J. L., Belkin, A. M., Syfrig, J., Thiery, J. P. and Koteliansky, V. E., 1992. Expression of alpha 1 integrin, a laminin-collagen receptor, during myogenesis and neurogenesis in the avian embryo. Development 116, 585600. 33 1118 1119 1120 1121 1122 1123 1124 1125 1126 1127 1128 1129 1130 1131 1132 1133 1134 1135 1136 1137 1138 1139 1140 1141 1142 1143 1144 1145 1146 1147 1148 1149 1150 1151 1152 1153 1154 1155 1156 1157 1158 1159 1160 1161 1162 1163 1164 1165 1166 1167 Duband, J. L., Rocher, S., Chen, W. T., Yamada, K. M. and Thiery, J. P., 1986. Cell adhesion and migration in the early vertebrate embryo: location and possible role of the putative fibronectin receptor complex. J Cell Biol 102, 160-78. Duband, J. L. and Thiery, J. P., 1987. Distribution of laminin and collagens during avian neural crest development. Development 101, 461-78. Duband, J. L., Tucker, G. C., Poole, T. J., Vincent, M., Aoyama, H. and Thiery, J. P., 1985. How do the migratory and adhesive properties of the neural crest govern ganglia formation in the avian peripheral nervous system? J Cell Biochem 27, 189-203. Duband, J. L., Volberg, T., Sabanay, I., Thiery, J. P. and Geiger, B., 1988. Spatial and temporal distribution of the adherens-junction-associated adhesion molecule A-CAM during avian embryogenesis. Development 103, 325-44. Dufour, S., Beauvais-Jouneau, A., Delouvee, A. and Thiery, J. P., 1999. Differential function of N-cadherin and cadherin-7 in the control of embryonic cell motility. J Cell Biol 146, 501-16. Duong, T. D. and Erickson, C. A., 2004. MMP-2 plays an essential role in producing epithelial-mesenchymal transformations in the avian embryo. Dev Dyn 229, 42-53. Edwards, D. R., Handsley, M. M. and Pennington, C. J., 2008. The ADAM metalloproteinases. Mol Aspects Med 29, 258-89. Farlie, P. G., Kerr, R., Thomas, P., Symes, T., Minichiello, J., Hearn, C. J. and Newgreen, D., 1999. A paraxial exclusion zone creates patterned cranial neural crest cell outgrowth adjacent to rhombomeres 3 and 5. Dev Biol 213, 70-84. Fassler, R. and Meyer, M., 1995. Consequences of lack of beta 1 integrin gene expression in mice. Genes Dev 9, 1896-908. Fu, M., Vohra, B. P., Wind, D. and Heuckeroth, R. O., 2006. BMP signaling regulates murine enteric nervous system precursor migration, neurite fasciculation, and patterning via altered Ncam1 polysialic acid addition. Dev Biol 299, 137-50. Gammill, L. S. and Roffers-Agarwal, J., 2010. Division of labor during trunk neural crest development. Dev Biol 344, 555-65. Gardner, H., Kreidberg, J., Koteliansky, V. and Jaenisch, R., 1996. Deletion of integrin alpha 1 by homologous recombination permits normal murine development but gives rise to a specific deficit in cell adhesion. Dev Biol 175, 301-13. Gaultier, A., Cousin, H., Darribere, T. and Alfandari, D., 2002. ADAM13 disintegrin and cysteine-rich domains bind to the second heparin-binding domain of fibronectin. J Biol Chem 277, 23336-44. Gawantka, V., Ellinger-Ziegelbauer, H. and Hausen, P., 1992. Beta 1-integrin is a maternal protein that is inserted into all newly formed plasma membranes during early Xenopus embryogenesis. Development 115, 595-605. Giambernardi, T. A., Sakaguchi, A. Y., Gluhak, J., Pavlin, D., Troyer, D. A., Das, G., Rodeck, U. and Klebe, R. J., 2001. Neutrophil collagenase (MMP-8) is expressed during early development in neural crest cells as well as in adult melanoma cells. Matrix Biol 20, 577-87. Goh, K. L., Yang, J. T. and Hynes, R. O., 1997. Mesodermal defects and cranial neural crest apoptosis in alpha5 integrin-null embryos. Development 124, 4309-19. Golding, J. P., Sobieszczuk, D., Dixon, M., Coles, E., Christiansen, J., Wilkinson, D. and Gassmann, M., 2004. Roles of erbB4, rhombomere-specific, and 34 1168 1169 1170 1171 1172 1173 1174 1175 1176 1177 1178 1179 1180 1181 1182 1183 1184 1185 1186 1187 1188 1189 1190 1191 1192 1193 1194 1195 1196 1197 1198 1199 1200 1201 1202 1203 1204 1205 1206 1207 1208 1209 1210 1211 1212 1213 1214 1215 rhombomere-independent cues in maintaining neural crest-free zones in the embryonic head. Dev Biol 266, 361-72. Gumbiner, B. M., 2005. Regulation of cadherin-mediated adhesion in morphogenesis. Nat Rev Mol Cell Biol 6, 622-34. Haack, H. and Hynes, R. O., 2001. Integrin receptors are required for cell survival and proliferation during development of the peripheral glial lineage. Dev Biol 233, 38-55. Hackett-Jones, E. J., Landman, K. A., Newgreen, D. F. and Zhang, D., 2011. On the role of differential adhesion in gangliogenesis in the enteric nervous system. J Theor Biol 287, 148-59. Hadeball, B., Borchers, A. and Wedlich, D., 1998. Xenopus cadherin-11 (Xcadherin11) expression requires the Wg/Wnt signal. Mech Dev 72, 101-13. Halbleib, J. M. and Nelson, W. J., 2006. Cadherins in development: cell adhesion, sorting, and tissue morphogenesis. Genes Dev 20, 3199-214. Hall, R. J. and Erickson, C. A., 2003. ADAM 10: an active metalloprotease expressed during avian epithelial morphogenesis. Dev Biol 256, 146-59. Hari, L., Brault, V., Kleber, M., Lee, H. Y., Ille, F., Leimeroth, R., Paratore, C., Suter, U., Kemler, R. and Sommer, L., 2002. Lineage-specific requirements of betacatenin in neural crest development. J Cell Biol 159, 867-80. Harrison, M., Abu-Elmagd, M., Grocott, T., Yates, C., Gavrilovic, J. and Wheeler, G. N., 2004. Matrix metalloproteinase genes in Xenopus development. Dev Dyn 231, 214-20. Heanue, T. A. and Pachnis, V., 2006. Expression profiling the developing mammalian enteric nervous system identifies marker and candidate Hirschsprung disease genes. Proc Natl Acad Sci U S A 103, 6919-24. Hodivala-Dilke, K. M., McHugh, K. P., Tsakiris, D. A., Rayburn, H., Crowley, D., Ullman-Cullere, M., Ross, F. P., Coller, B. S., Teitelbaum, S. and Hynes, R. O., 1999. Beta3-integrin-deficient mice are a model for Glanzmann thrombasthenia showing placental defects and reduced survival. J Clin Invest 103, 229-38. Huang, G. Y., Cooper, E. S., Waldo, K., Kirby, M. L., Gilula, N. B. and Lo, C. W., 1998. Gap junction-mediated cell-cell communication modulates mouse neural crest migration. J Cell Biol 143, 1725-34. Huang, J., Bridges, L. C. and White, J. M., 2005. Selective modulation of integrinmediated cell migration by distinct ADAM family members. Mol Biol Cell 16, 4982-91. Huh, M. I., Lee, Y. M., Seo, S. K., Kang, B. S., Chang, Y., Lee, Y. S., Fini, M. E., Kang, S. S. and Jung, J. C., 2007. Roles of MMP/TIMP in regulating matrix swelling and cell migration during chick corneal development. J Cell Biochem 101, 1222-37. Hynes, R. O., 2002. Integrins: bidirectional, allosteric signaling machines. Cell 110, 673-87. Inoue, T., Chisaka, O., Matsunami, H. and Takeichi, M., 1997. Cadherin-6 expression transiently delineates specific rhombomeres, other neural tube subdivisions, and neural crest subpopulations in mouse embryos. Dev Biol 183, 183-94. Iwashita, T., Kruger, G. M., Pardal, R., Kiel, M. J. and Morrison, S. J., 2003. Hirschsprung disease is linked to defects in neural crest stem cell function. Science 301, 972-6. 35 1216 1217 1218 1219 1220 1221 1222 1223 1224 1225 1226 1227 1228 1229 1230 1231 1232 1233 1234 1235 1236 1237 1238 1239 1240 1241 1242 1243 1244 1245 1246 1247 1248 1249 1250 1251 1252 1253 1254 1255 1256 1257 1258 1259 1260 1261 1262 1263 Joos, T. O., Reintsch, W. E., Brinker, A., Klein, C. and Hausen, P., 1998. Cloning of the Xenopus integrin alpha(v) subunit and analysis of its distribution during early development. Int J Dev Biol 42, 171-9. Joos, T. O., Whittaker, C. A., Meng, F., DeSimone, D. W., Gnau, V. and Hausen, P., 1995. Integrin alpha 5 during early development of Xenopus laevis. Mech Dev 50, 187-99. Kapur, R. P., 2000. Colonization of the murine hindgut by sacral crest-derived neural precursors: experimental support for an evolutionarily conserved model. Dev Biol 227, 146-155. Kasemeier-Kulesa, J. C., Bradley, R., Pasquale, E. B., Lefcort, F. and Kulesa, P. M., 2006. Eph/ephrins and N-cadherin coordinate to control the pattern of sympathetic ganglia. Development 133, 4839-47. Kasemeier-Kulesa, J. C., Kulesa, P. M. and Lefcort, F., 2005. Imaging neural crest cell dynamics during formation of dorsal root ganglia and sympathetic ganglia. Development 132, 235-45. Kashef, J., Kohler, A., Kuriyama, S., Alfandari, D., Mayor, R. and Wedlich, D., 2009. Cadherin-11 regulates protrusive activity in Xenopus cranial neural crest cells upstream of Trio and the small GTPases. Genes Dev 23, 1393-8. Kil, S. H. and Bronner-Fraser, M., 1996. Expression of the avian alpha 7-integrin in developing nervous system and myotome. Int J Dev Neurosci 14, 181-90. Kil, S. H., Krull, C. E., Cann, G., Clegg, D. and Bronner-Fraser, M., 1998. The alpha4 subunit of integrin is important for neural crest cell migration. Dev Biol 202, 29-42. Kimura, Y., Matsunami, H., Inoue, T., Shimamura, K., Uchida, N., Ueno, T., Miyazaki, T. and Takeichi, M., 1995. Cadherin-11 expressed in association with mesenchymal morphogenesis in the head, somite, and limb bud of early mouse embryos. Dev Biol 169, 347-58. Kirby, M. L. and Hutson, M. R., 2010. Factors controlling cardiac neural crest cell migration. Cell Adh Migr 4, 609-21. Kirby, M. L. and Waldo, K. L., 1995. Neural crest and cardiovascular patterning. Circ Res 77, 211-5. Komatsu, K., Wakatsuki, S., Yamada, S., Yamamura, K., Miyazaki, J. and SeharaFujisawa, A., 2007. Meltrin beta expressed in cardiac neural crest cells is required for ventricular septum formation of the heart. Dev Biol 303, 82-92. Krotoski, D. and Bronner-Fraser, M., 1990. Distribution of integrins and their ligands in the trunk of Xenopus laevis during neural crest cell migration. J Exp Zool 253, 139-50. Krotoski, D. M. and Bronner-Fraser, M., 1986. Mapping of neural crest pathways in Xenopus laevis. Prog Clin Biol Res 217B, 229-33. Krotoski, D. M., Domingo, C. and Bronner-Fraser, M., 1986. Distribution of a putative cell surface receptor for fibronectin and laminin in the avian embryo. J Cell Biol 103, 1061-71. Kulesa, P. M. and Fraser, S. E., 1998. Neural crest cell dynamics revealed by timelapse video microscopy of whole embryo chick explant cultures. Dev Biol 204, 327-44. Kulesa, P. M. and Fraser, S. E., 2000. In ovo time-lapse analysis of chick hindbrain neural crest cell migration shows cell interactions during migration to the branchial arches. Development 127, 1161-72. 36 1264 1265 1266 1267 1268 1269 1270 1271 1272 1273 1274 1275 1276 1277 1278 1279 1280 1281 1282 1283 1284 1285 1286 1287 1288 1289 1290 1291 1292 1293 1294 1295 1296 1297 1298 1299 1300 1301 1302 1303 1304 1305 1306 1307 1308 1309 1310 1311 Lallier, T. and Bronner-Fraser, M., 1992. Alpha 1 beta 1 integrin on neural crest cells recognizes some laminin substrata in a Ca(2+)-independent manner. J Cell Biol 119, 1335-45. Lallier, T. and Bronner-Fraser, M., 1993. Inhibition of neural crest cell attachment by integrin antisense oligonucleotides. Science 259, 692-5. Lallier, T. E. and Bronner-Fraser, M., 1988. A spatial and temporal analysis of dorsal root and sympathetic ganglion formation in the avian embryo. Dev Biol 127, 99-112. Lallier, T. E., Whittaker, C. A. and DeSimone, D. W., 1996. Integrin alpha 6 expression is required for early nervous system development in Xenopus laevis. Development 122, 2539-54. Le Douarin, N. M. and Kalcheim, C. 1999. The Neural Crest,2nd Edition. Cambridge University Press. Le Douarin, N. M. and Teillet, M. A., 1973. The migration of neural crest cells to the wall of the digestive tract in avian embryo. J Embryol Exp Morphol 30, 31-48. Lehmann, J. M., Holzmann, B., Breitbart, E. W., Schmiegelow, P., Riethmuller, G. and Johnson, J. P., 1987. Discrimination between benign and malignant cells of melanocytic lineage by two novel antigens, a glycoprotein with a molecular weight of 113,000 and a protein with a molecular weight of 76,000. Cancer Res 47, 841-5. Lele, Z., Folchert, A., Concha, M., Rauch, G. J., Geisler, R., Rosa, F., Wilson, S. W., Hammerschmidt, M. and Bally-Cuif, L., 2002. parachute/n-cadherin is required for morphogenesis and maintained integrity of the zebrafish neural tube. Development 129, 3281-94. Lin, J., Redies, C. and Luo, J., 2007. Regionalized expression of ADAM13 during chicken embryonic development. Dev Dyn 236, 862-70. Luca, M., Hunt, B., Bucana, C. D., Johnson, J. P., Fidler, I. J. and Bar-Eli, M., 1993. Direct correlation between MUC18 expression and metastatic potential of human melanoma cells. Melanoma Res 3, 35-41. Lumsden, A., Sprawson, N. and Graham, A., 1991. Segmental origin and migration of neural crest cells in the hindbrain region of the chick embryo. Development 113, 1281-91. Luo, B. H., Carman, C. V. and Springer, T. A., 2007. Structural basis of integrin regulation and signaling. Annu Rev Immunol 25, 619-47. Luo, Y., High, F. A., Epstein, J. A. and Radice, G. L., 2006. N-cadherin is required for neural crest remodeling of the cardiac outflow tract. Dev Biol 299, 517-28. Mah, S. P., Saueressig, H., Goulding, M., Kintner, C. and Dressler, G. R., 2000. Kidney development in cadherin-6 mutants: delayed mesenchyme-to-epithelial conversion and loss of nephrons. Dev Biol 223, 38-53. Manabe, T., Togashi, H., Uchida, N., Suzuki, S. C., Hayakawa, Y., Yamamoto, M., Yoda, H., Miyakawa, T., Takeichi, M. and Chisaka, O., 2000. Loss of cadherin-11 adhesion receptor enhances plastic changes in hippocampal synapses and modifies behavioral responses. Mol Cell Neurosci 15, 534-46. Maretzky, T., Schulte, M., Ludwig, A., Rose-John, S., Blobel, C., Hartmann, D., Altevogt, P., Saftig, P. and Reiss, K., 2005. L1 is sequentially processed by two differently activated metalloproteases and presenilin/gamma-secretase and regulates neural cell adhesion, cell migration, and neurite outgrowth. Mol Cell Biol 25, 9040-53. 37 1312 1313 1314 1315 1316 1317 1318 1319 1320 1321 1322 1323 1324 1325 1326 1327 1328 1329 1330 1331 1332 1333 1334 1335 1336 1337 1338 1339 1340 1341 1342 1343 1344 1345 1346 1347 1348 1349 1350 1351 1352 1353 1354 1355 1356 1357 1358 1359 1360 1361 Margadant, C., Monsuur, H. N., Norman, J. C. and Sonnenberg, A., 2011. Mechanisms of integrin activation and trafficking. Curr Opin Cell Biol 23, 607-14. McCusker, C., Cousin, H., Neuner, R. and Alfandari, D., 2009. Extracellular cleavage of cadherin-11 by ADAM metalloproteases is essential for Xenopus cranial neural crest cell migration. Mol Biol Cell 20, 78-89. McKinney, M. C., Stark, D. A., Teddy, J. and Kulesa, P. M., 2011. Neural crest cell communication involves an exchange of cytoplasmic material through cellular bridges revealed by photoconversion of KikGR. Dev Dyn 240, 1391-401. McLennan, R., Dyson, L., Prather, K. W., Morrison, J. A., Baker, R. E., Maini, P. K. and Kulesa, P. M., 2012. Multiscale mechanisms of cell migration during development: theory and experiment. Development Mittal, A., Pulina, M., Hou, S. Y. and Astrof, S., 2010. Fibronectin and integrin alpha 5 play essential roles in the development of the cardiac neural crest. Mech Dev 127, 472-84. Monier-Gavelle, F. and Duband, J. L., 1995. Control of N-cadherin-mediated intercellular adhesion in migrating neural crest cells in vitro. J Cell Sci 108 ( Pt 12), 3839-53. Monier-Gavelle, F. and Duband, J. L., 1997. Cross talk between adhesion molecules: control of N-cadherin activity by intracellular signals elicited by beta1 and beta3 integrins in migrating neural crest cells. J Cell Biol 137, 1663-81. Monsonego-Ornan, E., Kosonovsky, J., Bar, A., Roth, L., Fraggi-Rankis, V., Simsa, S., Kohl, A. and Sela-Donenfeld, D., 2012. Matrix metalloproteinase 9/gelatinase B is required for neural crest cell migration. Dev Biol 364, 16277. Nagy, N., Burns, A. J. and Goldstein, A. M., 2012. Immunophenotypic characterization of enteric neural crest cells in the developing avian colorectum. Dev Dyn 241, 842-51. Nagy, N., Mwizerwa, O., Yaniv, K., Carmel, L., Pieretti-Vanmarcke, R., Weinstein, B. M. and Goldstein, A. M., 2009. Endothelial cells promote migration and proliferation of enteric neural crest cells via beta1 integrin signaling. Dev Biol 330, 263-72. Nakagawa, S. and Takeichi, M., 1995. Neural crest cell-cell adhesion controlled by sequential and subpopulation-specific expression of novel cadherins. Development 121, 1321-32. Nakagawa, S. and Takeichi, M., 1998. Neural crest emigration from the neural tube depends on regulated cadherin expression. Development 125, 2963-71. Neuner, R., Cousin, H., McCusker, C., Coyne, M. and Alfandari, D., 2009. Xenopus ADAM19 is involved in neural, neural crest and muscle development. Mech Dev 126, 240-55. Newgreen, D. and Thiery, J. P., 1980. Fibronectin in early avian embryos: synthesis and distribution along the migration pathways of neural crest cells. Cell Tissue Res 211, 269-91. Park, K. S. and Gumbiner, B. M., 2010. Cadherin 6B induces BMP signaling and deepithelialization during the epithelial mesenchymal transition of the neural crest. Development 137, 2691-701. Park, K. S. and Gumbiner, B. M., 2012. Cadherin-6B stimulates an epithelial mesenchymal transition and the delamination of cells from the neural ectoderm via LIMK/cofilin mediated non-canonical BMP receptor signaling. Dev Biol 366, 232-43. 38 1362 1363 1364 1365 1366 1367 1368 1369 1370 1371 1372 1373 1374 1375 1376 1377 1378 1379 1380 1381 1382 1383 1384 1385 1386 1387 1388 1389 1390 1391 1392 1393 1394 1395 1396 1397 1398 1399 1400 1401 1402 1403 1404 1405 1406 1407 1408 1409 1410 1411 Pietri, T., Eder, O., Breau, M. A., Topilko, P., Blanche, M., Brakebusch, C., Fassler, R., Thiery, J. P. and Dufour, S., 2004. Conditional beta1-integrin gene deletion in neural crest cells causes severe developmental alterations of the peripheral nervous system. Development 131, 3871-83. Pietri, T., Thiery, J. P. and Dufour, S., 2003. Differential expression of beta3 integrin gene in chick and mouse cranial neural crest cells. Dev Dyn 227, 309-13. Piloto, S. and Schilling, T. F., 2010. Ovo1 links Wnt signaling with N-cadherin localization during neural crest migration. Development 137, 1981-90. Pinco, K. A., Liu, S. and Yang, J. T., 2001. alpha4 integrin is expressed in a subset of cranial neural crest cells and in epicardial progenitor cells during early mouse development. Mech Dev 100, 99-103. Rangarajan, J., Luo, T. and Sargent, T. D., 2006. PCNS: a novel protocadherin required for cranial neural crest migration and somite morphogenesis in Xenopus. Dev Biol 295, 206-18. Ranscht, B. and Bronner-Fraser, M., 1991. T-cadherin expression alternates with migrating neural crest cells in the trunk of the avian embryo. Development 111, 15-22. Ransom, D. G., Hens, M. D. and DeSimone, D. W., 1993. Integrin expression in early amphibian embryos: cDNA cloning and characterization of Xenopus beta 1, beta 2, beta 3, and beta 6 subunits. Dev Biol 160, 265-75. Reaume, A. G., de Sousa, P. A., Kulkarni, S., Langille, B. L., Zhu, D., Davies, T. C., Juneja, S. C., Kidder, G. M. and Rossant, J., 1995. Cardiac malformation in neonatal mice lacking connexin43. Science 267, 1831-4. Reiss, K., Maretzky, T., Ludwig, A., Tousseyn, T., de Strooper, B., Hartmann, D. and Saftig, P., 2005. ADAM10 cleavage of N-cadherin and regulation of cell-cell adhesion and beta-catenin nuclear signalling. EMBO J 24, 742-52. Retta, S. F., Cassara, G., D'Amato, M., Alessandro, R., Pellegrino, M., Degani, S., De Leo, G., Silengo, L. and Tarone, G., 2001. Cross talk between beta(1) and alpha(V) integrins: beta(1) affects beta(3) mRNA stability. Mol Biol Cell 12, 3126-38. Robbins, J. R., McGuire, P. G., Wehrle-Haller, B. and Rogers, S. L., 1999. Diminished matrix metalloproteinase 2 (MMP-2) in ectomesenchyme-derived tissues of the Patch mutant mouse: regulation of MMP-2 by PDGF and effects on mesenchymal cell migration. Dev Biol 212, 255-63. Rovasio, R. A., Delouvee, A., Yamada, K. M., Timpl, R. and Thiery, J. P., 1983. Neural crest cell migration: requirements for exogenous fibronectin and high cell density. J Cell Biol 96, 462-73. Sadaghiani, B. and Thiebaud, C. H., 1987. Neural crest development in the Xenopus laevis embryo, studied by interspecific transplantation and scanning electron microscopy. Dev Biol 124, 91-110. Schwarz, Q. and Ruhrberg, C., 2010. Neuropilin, you gotta let me know: should I stay or should I go? Cell Adh Migr 4, 61-6. Serbedzija, G. N., Bronner-Fraser, M. and Fraser, S. E., 1994. Developmental potential of trunk neural crest cells in the mouse. Development 120, 1709-18. Shiau, C. E. and Bronner-Fraser, M., 2009. N-cadherin acts in concert with Slit1Robo2 signaling in regulating aggregation of placode-derived cranial sensory neurons. Development 136, 4155-64. Shoval, I., Ludwig, A. and Kalcheim, C., 2007. Antagonistic roles of full-length Ncadherin and its soluble BMP cleavage product in neural crest delamination. Development 134, 491-501. 39 1412 1413 1414 1415 1416 1417 1418 1419 1420 1421 1422 1423 1424 1425 1426 1427 1428 1429 1430 1431 1432 1433 1434 1435 1436 1437 1438 1439 1440 1441 1442 1443 1444 1445 1446 1447 1448 1449 1450 1451 1452 1453 1454 1455 1456 1457 1458 1459 1460 1461 Stepp, M. A., Urry, L. A. and Hynes, R. O., 1994. Expression of alpha 4 integrin mRNA and protein and fibronectin in the early chicken embryo. Cell Adhes Commun 2, 359-75. Strachan, L. R. and Condic, M. L., 2003. Neural crest motility and integrin regulation are distinct in cranial and trunk populations. Dev Biol 259, 288-302. Strachan, L. R. and Condic, M. L., 2004. Cranial neural crest recycle surface integrins in a substratum-dependent manner to promote rapid motility. J Cell Biol 167, 545-54. Strachan, L. R. and Condic, M. L., 2008. Neural crest motility on fibronectin is regulated by integrin activation. Exp Cell Res 314, 441-52. Takahashi, M. and Osumi, N., 2005. Identification of a novel type II classical cadherin: rat cadherin19 is expressed in the cranial ganglia and Schwann cell precursors during development. Dev Dyn 232, 200-8. Taneyhill, L. A., 2008. To adhere or not to adhere: the role of Cadherins in neural crest development. Cell Adh Migr 2, 223-30. Taneyhill, L. A., Coles, E. G. and Bronner-Fraser, M., 2007. Snail2 directly represses cadherin6B during epithelial-to-mesenchymal transitions of the neural crest. Development 134, 1481-90. Teddy, J. M. and Kulesa, P. M., 2004. In vivo evidence for short- and long-range cell communication in cranial neural crest cells. Development 131, 6141-51. Testaz, S., Delannet, M. and Duband, J., 1999. Adhesion and migration of avian neural crest cells on fibronectin require the cooperating activities of multiple integrins of the (beta)1 and (beta)3 families. J Cell Sci 112 ( Pt 24), 4715-28. Testaz, S. and Duband, J. L., 2001. Central role of the alpha4beta1 integrin in the coordination of avian truncal neural crest cell adhesion, migration, and survival. Dev Dyn 222, 127-40. Theveneau, E., Duband, J. L. and Altabef, M., 2007. Ets-1 confers cranial features on neural crest delamination. PLoS One 2, e1142. Theveneau, E., Marchant, L., Kuriyama, S., Gull, M., Moepps, B., Parsons, M. and Mayor, R., 2010. Collective chemotaxis requires contact-dependent cell polarity. Dev Cell 19, 39-53. Thiery, J. P., Acloque, H., Huang, R. Y. and Nieto, M. A., 2009. Epithelialmesenchymal transitions in development and disease. Cell 139, 871-90. Thiery, J. P., Duband, J. L., Rutishauser, U. and Edelman, G. M., 1982. Cell adhesion molecules in early chicken embryogenesis. Proc Natl Acad Sci U S A 79, 6737-41. Tomczuk, M. (2004) ADAMs in early mouse development, vol. PhD. Charlottesville: University of Virginia. Tomlinson, M. L., Guan, P., Morris, R. J., Fidock, M. D., Rejzek, M., GarciaMorales, C., Field, R. A. and Wheeler, G. N., 2009. A chemical genomic approach identifies matrix metalloproteinases as playing an essential and specific role in Xenopus melanophore migration. Chem Biol 16, 93-104. Trainor, P. A., Sobieszczuk, D., Wilkinson, D. and Krumlauf, R., 2002. Signalling between the hindbrain and paraxial tissues dictates neural crest migration pathways. Development 129, 433-42. Tucker, R. P., 2004. Antisense knockdown of the beta1 integrin subunit in the chicken embryo results in abnormal neural crest cell development. Int J Biochem Cell Biol 36, 1135-9. Turlo, K. A., Noel, O. D., Vora, R., Larussa, M., Fassler, R., Hall-Glenn, F. and Iruela-Arispe, M. L., 2012. An essential requirement for beta1 integrin in the 40 1462 1463 1464 1465 1466 1467 1468 1469 1470 1471 1472 1473 1474 1475 1476 1477 1478 1479 1480 1481 1482 1483 1484 1485 1486 1487 1488 1489 1490 1491 1492 1493 1494 1495 1496 1497 1498 1499 1500 1501 1502 1503 1504 1505 1506 1507 1508 1509 1510 assembly of extracellular matrix proteins within the vascular wall. Dev Biol 365, 23-35. Ulrich, F. and Heisenberg, C. P., 2009. Trafficking and cell migration. Traffic 10, 811-8. Vallin, J., Girault, J. M., Thiery, J. P. and Broders, F., 1998. Xenopus cadherin-11 is expressed in different populations of migrating neural crest cells. Mech Dev 75, 171-4. van der Flier, A. and Sonnenberg, A., 2001. Function and interactions of integrins. Cell Tissue Res 305, 285-98. VanSaun, M. N. and Matrisian, L. M., 2006. Matrix metalloproteinases and cellular motility in development and disease. Birth Defects Res C Embryo Today 78, 69-79. Vohra, B. P., Tsuji, K., Nagashimada, M., Uesaka, T., Wind, D., Fu, M., Armon, J., Enomoto, H. and Heuckeroth, R. O., 2006. Differential gene expression and functional analysis implicate novel mechanisms in enteric nervous system precursor migration and neuritogenesis. Dev Biol 298, 259-71. Vu, T. H., Shipley, J. M., Bergers, G., Berger, J. E., Helms, J. A., Hanahan, D., Shapiro, S. D., Senior, R. M. and Werb, Z., 1998. MMP-9/gelatinase B is a key regulator of growth plate angiogenesis and apoptosis of hypertrophic chondrocytes. Cell 93, 411-22. Waldo, K., Miyagawa-Tomita, S., Kumiski, D. and Kirby, M. L., 1998. Cardiac neural crest cells provide new insight into septation of the cardiac outflow tract: aortic sac to ventricular septal closure. Dev Biol 196, 129-44. Wallace, A. S., Schmidt, C., Schachner, M., Wegner, M. and Anderson, R. B., 2010. L1cam acts as a modifier gene during enteric nervous system development. Neurobiol Dis 40, 622-33. Wallace, A. S., Tan, M. X., Schachner, M. and Anderson, R. B., 2011. L1cam acts as a modifier gene for members of the endothelin signaling pathway during enteric nervous system development. Neurogastroenterol Motil 23:e510-22, Wang, D. and Burke, R. D., 1995. Rhombencephalic cranial neural crest cells show temporal regulation and axial differences in the expression of alpha-integrins. Mol Biol Cell Supplement 6, 323a. Wang, X., Chan, A. K., Sham, M. H., Burns, A. J. and Chan, W. Y., 2011. Analysis of the sacral neural crest cell contribution to the hindgut enteric nervous system in the mouse embryo. Gastroenterology 141, 992-1002 e1-6. Wei, S., Xu, G., Bridges, L. C., Williams, P., White, J. M. and DeSimone, D. W., 2010. ADAM13 induces cranial neural crest by cleaving class B Ephrins and regulating Wnt signaling. Dev Cell 19, 345-52. Weston, J. A., 1963. A radioautographic analysis of the migration and localization of trunk neural crest cells in the chick. Dev Biol 6, 279-310. Whittaker, C. A. and Desimone, D. W., 1998. Molecular cloning and developmental expression of the Xenopus homolog of integrin alpha 4. Ann N Y Acad Sci 857, 56-73. Xie, S., Luca, M., Huang, S., Gutman, M., Reich, R., Johnson, J. P. and Bar-Eli, M., 1997. Expression of MCAM/MUC18 by human melanoma cells leads to increased tumor growth and metastasis. Cancer Res 57, 2295-303. Xu, X., Francis, R., Wei, C. J., Linask, K. L. and Lo, C. W., 2006. Connexin 43mediated modulation of polarized cell movement and the directional migration of cardiac neural crest cells. Development 133, 3629-39. 41 1511 1512 1513 1514 1515 1516 1517 1518 1519 1520 1521 1522 1523 1524 1525 1526 1527 1528 1529 1530 1531 1532 1533 1534 1535 1536 1537 1538 Xu, X., Li, W. E., Huang, G. Y., Meyer, R., Chen, T., Luo, Y., Thomas, M. P., Radice, G. L. and Lo, C. W., 2001. Modulation of mouse neural crest cell motility by N-cadherin and connexin 43 gap junctions. J Cell Biol 154, 21730. Yang, J. T., Rayburn, H. and Hynes, R. O., 1995. Cell adhesion events mediated by alpha 4 integrins are essential in placental and cardiac development. Development 121, 549-60. Yano, T., Yamazaki, Y., Adachi, M., Okawa, K., Fort, P., Uji, M. and Tsukita, S., 2011. Tara up-regulates E-cadherin transcription by binding to the Trio RhoGEF and inhibiting Rac signaling. J Cell Biol 193, 319-32. Yntema, C. L. and Hammond, W. S., 1954. The origin of intrinsic ganglia of trunk viscera from vagal neural crest in the chick embryo. J Comp Neurol 101, 515 541. Young, H. M., Bergner, A. J., Anderson, R. B., Enomoto, H., Milbrandt, J., Newgreen, D. F. and Whitington, P. M., 2004. Dynamics of neural crestderived cell migration in the embryonic mouse gut. Dev Biol 270, 455-73. Highlights x x x x Migrating neural crest cells express a variety of cell adhesion molecules Expression of adhesion molecules can vary between axial levels and species Cleaving of cell adhesion molecules by matrix metalloproteases can affect migration Function of adhesion molecules can be similar or different between axial levels 42 Vagal enteri c Vagal cardia c 6 Cadherin 74 Ncadherin 4,5 Cadheri n 6B (EMT) Cadherin 6 39,40,41,42 43 Cadherin 6 Cadherin 1 6B (EMT) 4 Cadherin Cadherin 11 2 74 Ncadherin (weak) 33 NNcadherin cadherin Rodent Axial level of NC Crani Cadherinal 61 Cadherin11 2 Cadherin19 (rat) 3 Cadherins Avian Cadherin 11 34 Cadheri n-11 8 PCNS 9 146 4 44 5 45 6 42 v 45 v 1 16 3 15 56 15 4 13 5 14 14 4 13 5 14 6 (subset R6-8) 5 35 1 36 4 10 5 11 1 12 Ncadherin 7 Roden t Xenopus Rodent ADAM1 7 42 ADAMT S1 42 5 17 ADAM1 0 21 6 (subset) ADAM1 18 2 21 V ADAM1 (weak) 5 21 19 ADAM1 7 21 1 20 ADAM1 9 21 ADAM3 3 21 MMP2 22 MMP8 23 ADAM1 9 37 Integrins Avian Xenopu s ADAM11 27 MMP2 25,38 MMP14 TIMP-2 38 28 ADAM13 27 38 ADAM9 24 MMP2 30 MMP14 31 29 ADAM19 28 ADAM13 MMP2 MMP9 26 ADAM13 25 25 ADAM11 6 27 ADAM33 ADAM9 24 Xenopus ADAM13 MMPs Avian NCAM2 40,48 NCAM 39 L1CAM L1CA M 43 49 NCAM 32 NCAM IgCAMs Rodent Avian Xen opus Table 1: Expression of the major classes cell adhesion molecules in NC by axial level and species. Underlining or bolding indicates a function for the molecule during NC migration has been demonstrated in vitro or in vivo respectively. 10 4 1 52 Trunk Cadherin 6 Cadheri 1 n 6B Cadherin (EMT) 4, 50 3 19 (rat) Cadherin 7 (subset) 4 Cadherin 11 51 Ncadherin (in vitro) Pcdh 15 41 41 Pcdh 41 Pcdh 40 Pcdh 41 Cadherin 11 39 Cadherin 9 39,41 39 6 11 1 12 11,35 4 (weak ) 13 5 42 3 5 39 2like 41 E1 39,42 1 54,55 8 55 v 5 59 8 54,55 54,55,59 1 60 3 54,55,59 58 3 4 55,56 5 55 6 54,57 7 (subset) 53,54 1 (weak) MMP2 (weak) 62 MMP9 26 61 MMP8 23 ADAM 10 47 MMP2 (in NC or pathway) MMP14 30 31 MMP2 (in pathway) Igsf4a 41 41 NCAM (downreg) 64 63 MCAM (Inoue et al., 1997), 2 (Kimura et al., 1995), 3 (Takahashi and Osumi, 2005), 4 (Nakagawa and Takeichi, 1995), 5 (Taneyhill et al., 2007) 6 (McLennan et al., 2012), 7 (Theveneau et al., 2010), 8 (Hadeball et al., 1998; Vallin et al., 1998), 9 (Rangarajan et al., 2006), 10 (Pinco et al., 2001), 11 (Bajanca et al., 2004), 12 (Pietri et al., 2004), 13 (Kil et al., 1998), 14 (Wang and Burke, 1995), 15 (Pietri et al., 2003), 16 (Bronner-Fraser, 1985; Duband et al., 1986), 17 (Joos et al., 1995), 18 (Lallier et al., 1996), 19 (Joos et al., 1998), 20 (Krotoski and Bronner-Fraser, 1990; Ransom et al., 1993), 21 (Tomczuk, 2004), 22 (Robbins et al., 1999), 23 (Giambernardi et al., 2001), 24 (Lin et al., 2007), 25 (Cai et al., 2000), 26 (MonsonegoOrnan et al., 2012), 27 (Cai et al., 1998), 28 (Alfandari et al., 1997), 29 (Neuner et al., 2009), 30 (Tomlinson et al., 2009), 31 (Harrison et al., 2004), 32 (Bronner-Fraser et al., 1992b), 33 (Luo et al., 2006; Xu et al., 2001), 34 (Vallin et al., 1998), 35 (Goh et al., 1997), 36 (Xu et al., 2006), 37 (Komatsu et al., 2007), 38 (Cantemir et al., 2004), 39 (Breau et al., 2006), 40 (Heanue and Pachnis, 2006), 41 (Vohra et al., 2006), 42 (Iwashita et al., 2003), 43 (Hackett-Jones et al., 2011; Nagy et al., 2012), 44 (Bixby et al., 2002), 45 (Breau et al., 2009), 46 (Nagy et al., 2009), 47 (Anderson, 2010), 48 (Anderson et al., 2006b), 49 (Hackett-Jones et al., 2011), 50 (Park and Gumbiner, 2010), 51 (Chalpe et al., 2010), 52 (Monier-Gavelle and Duband, 1995), 53 (Duband et al., 1992), 54 (Desban and Duband, 1997), 55 (Testaz et al., 1999), 56 (Kil et al., 1998; Stepp et al., 1994), 57 (Bronner-Fraser et al., 1992a), 58 (Kil and Bronner-Fraser, 1996), 59 (Delannet et al., 1994), 60 (Duband et al., 1986; Krotoski et al., 1986), 61 (Hall and Erickson, 2003), 62 (Duong and Erickson, 2004), 63 (Alais et al., 2001), 64 (Akitaya and Bronner-Fraser, 1992) 1