* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Novel In Vitro Method for Screening Inhibitors of Protein Translation
Protein moonlighting wikipedia , lookup
Genetic code wikipedia , lookup
RNA polymerase II holoenzyme wikipedia , lookup
Gene regulatory network wikipedia , lookup
Protein (nutrient) wikipedia , lookup
Transcriptional regulation wikipedia , lookup
Secreted frizzled-related protein 1 wikipedia , lookup
Western blot wikipedia , lookup
Deoxyribozyme wikipedia , lookup
RNA interference wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Protein adsorption wikipedia , lookup
Gene expression profiling wikipedia , lookup
Silencer (genetics) wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Non-coding RNA wikipedia , lookup
Expression vector wikipedia , lookup
Polyadenylation wikipedia , lookup
De novo protein synthesis theory of memory formation wikipedia , lookup
Gene expression wikipedia , lookup
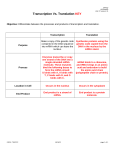
Novel In Vitro Method for Screening Inhibitors of Protein Translation Darius Kavaliauskas1, Krishna Vattem2, Juozas Šiurkus1, Janaki Narahari2, Atul Deshpande2, Brian Webb2 1 Thermo Fisher Scientific • Vilnius, Lithuania 2Thermo Fisher Scientific • Rockford, IL Abstract Inhibitors of protein synthesis represent an emerging class of anti-viral and anti-cancer therapeutics. In vitro translation (IVT) or cell-free expression offers a convenient and powerful assay to screen for translational inhibitors that regulate both cellular as well as viral mRNA expression. We have developed a simple method to identify inhibitors of cap-dependent and cap-independent protein translation using expression of luciferase mRNA (5’-cap-TuboLuc luciferase mRNA and IRES-Red Firefly luciferase mRNA) in the Thermo Scientific 1-Step Human Coupled IVT system. To facilitate detection of both cap-dependent and capindependent translation in the same well, we developed a 2-step luciferase assay reagent for TurboLuc and Firefly luciferases. Rapid assay read-out (<90 min), ability for miniaturization, and insensitivity to compound toxicity make the in vitro translation format an attractive alternative to cell-based screens for HTS identification of novel inhibitors of protein synthesis. Figure 1. Novel Method for Screening for Translational Inhibitors Using an IVT-based format. Inhibitors of protein synthesis represent a current and recurring interest of Pharma and academic researchers for anti-viral and anti-cancer therapeutics. In vitro translation offers a unique and powerful assay to screen for translational inhibitors because it provides rapid protein expression and is amenable to miniaturization for HTS. Compounds specific for both cap-dependent and cap-independent (IRES) translation mechanisms can be screened & identified using IVT reactions containing two different mRNAs, one capped mRNA encoding for TurboLuc luciferase and another uncapped (IRES) mRNA encoding for Red Firefly luciferase. The levels of both luciferases are measured following a 90 min IVT reaction using a newly developed 2-Step TurboLuc/ RFF dual assay reagent. Theoretical cap-dependent inhibitor screening results are illustrated. Figure 3. Evaluation of different mRNA configurations for screening inhibitors specific for cap-dependent and cap-independent translation in IVT. Panel A demonstrates expression of capped-TurboLuc luciferase (Tluc) mRNA in IVT. This format can be used to obtain identify inhibitors of cap-dependent translation. Inhibition of translation is observed with either cap specific m7GDP (0.1 mM) or any of the general inhibitors of translation such as Cyclohexamide (CHX, 0.1 mM ) and Puromycin (0.1 mM). In Panel B, where capped-Tluc and IRES- Red Firefly (RFF) mRNA’s were simultaneously expressed in IVT, TLuc expression was again inhibited by m7GDP, whereas RFF expression was unaffected, suggesting specificity of m7GDP inhibition towards capped mRNA’s. IRES-RFF mRNA expression in these experiments could potentially serve as internal control making sure the IVT expression system is functional. In Panel C, instead of having two separate mRNA’s as in Panel B, a single dual-luciferase mRNA containing both Tluc and RFF on the same construct was expressed. Again, m7GDP specifically affected the expression of Tluc but not the IRES. 7-Methylguanosine 5′-diphosphate (m7GDP) is a eukaryotic mRNA cap inhibitor that binds to cap binding protein eIF4E and prevents its function. Cyclohexamide and Puromycin inhibit general protein synthesis by either blocking elongation step or inducing premature termination of protein synthesis, respectively. Ribavirin is a RNA nucleoside analogue known to block viral RNA replication in vivo, and its mechanism is not clearly understood. A. B. Single capped mRNA Dual capped and uncaped mRNAs C. Capped bi-cistronic mRNA + inhibitor mRNA or Linear or Circular DNA template Figure 4. Demonstration of both selective and general inhibition of translation using dual mRNA luciferase assay in both HeLa and CHO IVT. Transcription/Translation 90 minutes, 30°C A. In vitro protein expression is a A. Inconvenient vitro proteinsystem expression is a rapid, convenient system to study protein rapid, to study + inhibitor mRNA or Linear or Circular translation in a cell-free format protein translation in a cell-free DNA template format• Protein produced in <60 min • Can screen toxic • Protein produced incompounds <60 min • Amenable to HTS • Can screen toxic compounds • Amenable to HTS mRNAs for cap-dependent expressing Turbo-luc and cap independent expressing Red Fire Fly luciferase were expressed in both HeLa and CHO IVT reactions where various inhibitors where added and luciferase activity was assayed and quantified as percent control. Either IVT lysate (HeLa or CHO) can be used for screening depending on needs/preferences. Transcription/Translation 90 minutes, 30°C B. Simultaneously monitor cap-dependent and cap-independent (IRES) translation in IVT reaction • Capped TurboLuc (Tluc) luciferase mRNA • IRES Red Firefly (RFF) luciferase mRNA IVT reaction + compound library Detect luciferase activity using 2-Step TurboLuc/RFF Dual assay HeLa or CHO IVT reaction 30°C, 1.5 h Figure 5. Selective inhibition of cap-dependent translation (m7GDP) and non-selective inhibition of translation with puromycin detected with dual mRNA IVT assay. Specific titration of m7GDP and puromycin in HeLa IVT system using both cap-dependent-Turboluc and cap-independent-RFF mRNAs. Inhibition of cap-dependent translation (Tluc) is seen with addition of m7GDP but not cap-independent translation (RFF). Puromycin inhibits both cap-dependent and cap-independent translation. Figure 2. Optimization of mRNA capping for IVT. mRNA capped co-transcriptionally with ARCA by using T7 TranscriptAid High Yield Transcription kit (Thermo Scientific) or by using NEB and CellScript enzymatic capping kits after transcription with GTP and S-adenosylmethionine. IVT reactions were run in HeLa cell lysate and luminescence detected with TurboLuc™ Luciferase One-Step Glow Assay Kit (Thermo Scientific). The yellow circle represents the mRNA cap structure. Results showed that ARCA capping of mRNA is more efficient than enzymatic capping, thus ARCA capped mRNA was used in all further experiments. ARCA 5’ TLuc AAAAAA 3’ Relative luminescence (a.u.) Relative luminescence (a.u.) Tluc RFF 100 80 60 40 20 IC50(Tluc) =10.5 μM 0 1E-5 1E-4 1E-3 0.01 m7GDP, mM 0.1 1 Tluc RFF 100 80 60 40 20 IC50 (Tluc)=0.087 μM IC50 (RFF)=0.024 μM 0 1E-6 1E-5 1E-4 1E-3 0.01 0.1 1 10 Puromycin, M Summary For Research Use Only, Not for Use in Diagnostic Procedures. ©Thermo Fisher Scientific Inc. All Rights Reserved. All trademarks are property of Thermo Fisher Scientific and its subsidiaries unless otherwise specified. • In vitro translation systems based on HeLa and CHO lysate were successfully used to demonstrate a rapid in vitro assay for screening protein synthesis inhibitors in a miniaturized, fast and HTS compatible format. • Simultaneous monitoring of cap-dependent and IRES-dependent translation using two distinct luciferase mRNA templates differentiates cap-dependent vs. cap-independent translation mechanisms. A single assay reagent can further be used to measure both the luciferases sequentially in the same microplate well. • The Anti-Reverse Cap Analog (ARCA) from Thermo Scientific produces at least 70% more translatable capped transcripts than capping kits from other suppliers.