* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Answers
Elias James Corey wikipedia , lookup
Marcus theory wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Hydroformylation wikipedia , lookup
Ene reaction wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
George S. Hammond wikipedia , lookup
Aldol reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Stille reaction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Asymmetric induction wikipedia , lookup
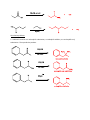
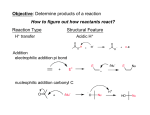
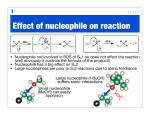
Nucleophilic Acyl Substitution—weak Nu Major concepts Nucleophilic acyl substitutions can occur with weak nucleophiles under basic conditions, but only if the carboxylic acid derivative is very reactive Nulceophilic acyl substitutions can occur with weak nucleophiles under acidic conditions Carboxylic acids can be esterified with alcohols under acidic conditions Vocabulary Fisher esterification Students should be able to: Draw a mechanism for nucleophilic acyl substitution under acidic conditions (Fisher esterification) Determine functional groups that undergo nucleophilic acyl substitution reactions Predict the products of nucleophilic acyl substitutions Predict the direction of equilibrium and draw energy diagrams for the mechanisms of nucleophilic acyl substitution taking into account Le Chatlier’s principle Daily Problems 1. Draw a mechanism for this Fisher esterification. What byproduct is produced? O CH3OH O H+ + OCH 3 OH H2O byproduct -H+ OH O OH CH3OH OH CH3OH O H O O HO OH HO HO OH H OHCH 3 OH2 2. Although the stability of the starting materials and products are about the same in the reaction above, the equilibrium lies far to the right. Explain how this is possible. How could this reaction be forced back to the left? (Hint: What is the byproduct of the reaction?) This equilibrium is possible through the addition of acid to the electrophile, making it a better electrophile. This means it does not need as strong of a nucleophile to react. The reaction could be forced back to the left is water was added. 3. These reactive carboxylic acid derivatives will react with these weak nucleophiles. Predict the product, and explain what the purpose of the triethylamine (TEA) and pyridine (pyr) is in each reaction. Triethylamine and pyridine are bases that “soak up” the byproduct (acid). 4. Predict the products of these nucleophilic acyl substitution reactions. Cumulative problems: 5. Label each reaction as a nucleophilic substitution, a nucleophilic addition, or a nucleophilic acyl substitution. Then predict the product.