* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Identification of a C-terminal Poly(A)-binding Protein (PABP)
Protein (nutrient) wikipedia , lookup
Protein phosphorylation wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Magnesium transporter wikipedia , lookup
Protein moonlighting wikipedia , lookup
Signal transduction wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
List of types of proteins wikipedia , lookup
Gene expression wikipedia , lookup
Messenger RNA wikipedia , lookup
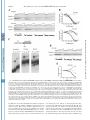
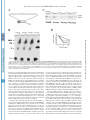
THE JOURNAL OF BIOLOGICAL CHEMISTRY © 2003 by The American Society for Biochemistry and Molecular Biology, Inc. Vol. 278, No. 47, Issue of November 21, pp. 46357–46368, 2003 Printed in U.S.A. Identification of a C-terminal Poly(A)-binding Protein (PABP)-PABP Interaction Domain ROLE IN COOPERATIVE BINDING TO POLY(A) AND EFFICIENT CAP DISTAL TRANSLATIONAL REPRESSION* Received for publication, July 15, 2003, and in revised form, August 25, 2003 Published, JBC Papers in Press, September 2, 2003, DOI 10.1074/jbc.M307624200 Eduardo O. Melo‡§, Rafael Dhalia‡¶, Cezar Martins de Sa‡, Nancy Standart储, and Osvaldo P. de Melo Neto¶** From the ‡Departamento de Biologia Celular, Universidade de Brasilia, Brasilia DF 70910-900, Brazil, §Embrapa Recursos Genéticos e Biotecnologia, Parque Rural, Final W5, Asa Norte, Brası́lia DF 70770-900, Brazil, ¶Centro de Pesquisas Aggeu Magalhães, Fundação Oswaldo Cruz, Campus da Universidade Federal de Pernambuco, Avenida Moraes Rego s/n, Recife PE 50670-420, Brazil, and the 储Department of Biochemistry, University of Cambridge, Tennis Court Road, Cambridge CB2 1GA, United Kingdom * This work was supported by the Conselho Nacional de Desenvolvimento Cientifico e Tecnológico, Coordemação de Aperfeiçoamento de Pessoal de Nı́vel Superior, and the British Council. The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. ** To whom correspondence should be addressed. Tel.: 55-81-33012568; Fax: 55-81-3453-2449; E-mail: [email protected]. This paper is available on line at http://www.jbc.org The poly(A)-binding protein (PABP)1 bound to the 3⬘ poly(A) tail of eukaryotic mRNAs is now recognized as an important if not an essential component of the apparatus required for mRNA translation. Evidence from yeast, plant, and animal models has shown a role for the PABP-poly(A) complex in translation initiation and points to a synergism between the 3⬘ and 5⬘ ends of the mRNAs (through the cap- and poly(A)associated proteins) in promoting translation (reviewed in Refs. 1– 4). The demonstration of an interaction between the translation initiation factor eIF4G and PABP provides an elegant explanation as to how this synergism occurs (5). Simultaneous binding of eIF4G to both PABP and eIF4E (the capbinding protein) leads to a circularization of the mRNA, as seen by atomic force microscopy, which may enhance its ability to undergo re-initiation (6). The PABP-poly(A) complex has also been implicated in the control of mRNA stability in yeast (reviewed in Refs. 7 and 8) and in mammals (9, 10). PABP consists of a highly conserved N terminus containing four tandem RNA recognition motifs (RRM) followed by a more variable C terminus (for review, see Ref. 4). The first two RRMs are sufficient for specific poly(A)-binding (11–14). In addition, RRM2 promotes the interaction between PABP and eIF4G (15–17). RRM4 is responsible for most of the nonspecific RNA binding of PABP (12, 13); in yeast the first half of RRM4 is sufficient to confer viability to cells depleted of the normal PABP gene (11). In Xenopus oocytes, RRMs can stimulate translation when tethered to reporter mRNA (18). As to the C terminus, although it does not bind RNA, it enables PABP to multimerize on poly(A) (12). Recently it was shown to include a domain responsible for binding to the PABP-interacting proteins Paip1 and Paip2 as well as to the release factor eRF3 (19). This 74-residue domain (called PABC) is conserved in all PABPs described so far (from protists to vertebrates) and is also present in the hyperplastic discs protein (HYD) family of ubiquitin ligases (20). The structures of both human PABC and its HYD homologue confirmed it as a novel phylogenetically conserved domain responsible for protein-protein interactions (19, 20). The C terminus can contribute to mRNA stabilization 1 The abbreviations used are: PABP, poly(A)-binding protein; RRM, RNA recognition motif; CAT, chloramphenicol acetyltransferase; nt, nucleotide(s); UTR, untranslated region; IRE, iron regulatory element; IRP, iron regulatory protein; GST, glutathione S-transferase; BB, binding buffer; BSA, bovine serum albumin; Ni-NTA, nickel-nitrilotriacetic acid; DMEM, Dulbecco’s modified Eagle’s medium. 46357 Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 The poly(A)-binding protein (PABP), bound to the 3ⴕ poly(A) tail of eukaryotic mRNAs, plays critical roles in mRNA translation and stability. PABP autoregulates its synthesis by binding to a conserved A-rich sequence present in the 5ⴕ-untranslated region of PABP mRNA and repressing its translation. PABP is composed of two parts: the highly conserved N terminus, containing 4 RNA recognition motifs (RRMs) responsible for poly(A) and eIF4G binding; and the more variable C terminus, which includes the recently described PABC domain, and promotes intermolecular interaction between PABP molecules as well as cooperative binding to poly(A). Here we show that, in vitro, GST-PABP represses the translation of reporter mRNAs containing 20 or more A residues in their 5ⴕ-untranslated regions and remains effective as a repressor when an A61 tract is placed at different distances from the cap, up to 126 nucleotides. Deletion of the PABP C terminus, but not the PABC domain alone, significantly reduces its ability to inhibit translation when bound to sequences distal to the cap, but not to proximal ones. Moreover, cooperative binding by multiple PABP molecules to poly(A) requires the C terminus, but not the PABC domain. Further analysis using pull-down assays shows that the interaction between PABP molecules, mediated by the C terminus, does not require the PABC domain and is enhanced by the presence of RRM 4. In vivo, fusion proteins containing parts of the PABP C terminus fused to the viral coat protein MS2 have an enhanced ability to prevent the expression of chloramphenicol acetyltransferase reporter mRNAs containing the MS2 binding site at distal distances from the cap. Altogether, our results identify a proline- and glutamine-rich linker located between the RRMs and the PABC domain as being strictly required for PABP/PABP interaction, cooperative binding to poly(A) and enhanced translational repression of reporter mRNAs in vitro and in vivo. 46358 Identification of a C-terminal PABP-PABP Interaction Domain EXPERIMENTAL PROCEDURES Cyclin Reporter Plasmids—The cyclin mRNAs described in this study were based on the Xenopus cyclin A cDNA in the plasmid pTZ18R, which contained a tract of 61 A residues, located 41 nucleotides from the 5⬘ end of cyclin mRNA (25). In this and in the following constructs, the cyclin cDNA is missing the end of its 3⬘-UTR (⬃80 nt) and it lacks a 3⬘ poly(A) tail (we have seen that the presence or absence of such a tail does not affect translation inhibition/stimulation by poly(A)). To reduce the A tract length, this plasmid was digested with StuI and XbaI and the longer fragment, devoid of the original A tract, was ligated to pairs of oligonucleotides designed to reduce the number of A residues to 20 or 10 nucleotides (5⬘ primers: 5⬘-CCTA20T-3⬘ and 5⬘-CCTA10T-3⬘). To reduce the distance between the transcription initiation site and the start of the A tract, the original plasmid was digested with EcoRI and StuI, treated with T4 DNA polymerase, and religated (distance cap/A tract of 10 nucleotides). To increase the distance, the starting plasmid was digested with KpnI and BamHI and ligated with the 62-bp KpnI/ BamHI fragment from plasmid Bluescript II KS⫹ from Stratagene. This plasmid was then digested with XhoI and ClaI, treated with T4 DNA polymerase, and then either religated (distance 86) or ligated with either one or two copies of the 40-nt StuI/SacII fragment obtained from the human PABP cDNA 5⬘-UTR (distances 126 and 166). All plasmid constructions were confirmed by sequencing. In Vitro Transcription and Translation—Transcriptions were performed as described previously (25), using T7 RNA polymerase with SalI (cyclin) or BamHI (PABP) linearized plasmid DNAs. For the translation reactions, the rabbit reticulocyte cell-free system (Promega) was treated with micrococcal nuclease to render it mRNA-dependent. In vitro transcribed RNAs were translated at the final concentrations indicated in the figures. Protein synthesis was assayed at 30 °C with [35S]methionine (Amersham Biosciences) and the radiolabeled products analyzed by 15% SDS-polyacrylamide gel electrophoresis and autoradiography. When used, polynucleotides were pre-incubated with the reticulocyte mix for 15 min on ice, prior to addition of mRNA, followed by incubation at 30 °C for 1 h. For the translations with the added proteins, mRNAs were pre-incubated with the GST fusion proteins and ribonuclease inhibitor (final concentration of ⬃200 units/ml in the translation reaction) on ice for 15 min in the presence of Escherichia coli rRNA (2.5 mg/ml) and poly(G) or poly(C) (at 2 mg/ml final) as nonspecific competitors, prior to the addition of the translation extract. All the translation results shown are representative of various experiments performed with different batches of mRNAs and proteins, and the data were highly reproducible in trend from experiment to experiment. Repeating the experiments with the same mRNA/protein batches produced identical results. GST-PABP Expression Plasmids—The plasmid encoding human PABP (construct P10 – 636, missing the first nine codons), cloned into the BamHI site of pGEX2T, has been described (25). Clones encoding the PABP variants P10 –584, P10 –370, and P372– 636 were obtained by PCR using an annealing temperature of 50 °C for 30 cycles. As 5⬘ primer for variants P10 –584 and P10 –370, the oligonucleotide 6291 (5⬘-CGGGATCCCTGCGGGCAGCCG-3⬘), which anneals immediately upstream of the PABP translation initiation codon, was used. The 3⬘ oligonucleotides, 3771 and 3772, which are complementary to sequences coding for conserved motifs present at the end of the fourth RRM (3⬘ end of P10 –370) and in the second half of the C terminus (3⬘ end of P10 –584), have been described previously (39). To clone fragments P10 –584 and P10 –370 into the pGET2T expression vector, the PCR fragments were digested with NcoI (which cuts on the second methionine at amino acid position 10) and BamHI, prior to in-filling and ligation into the in-filled BamHI-cut pGEX2T vector (Amersham Biosciences). The P372– 636 PCR fragment was amplified with the oligonucleotides 7736 (5⬘-CCGGGATCCGAAGAGCGCCAGGCT-3⬘) and 7737 (5⬘-CGGAATTCTAGATATTTTTCTTCGGTG-3⬘) and inserted into the BamHI/EcoRI sites of the pGEX2T vector. The P10 –370 plasmid was further digested with EcoRI and the larger fragment gel religated to yield the P10 –190 plasmid. P237– 636 was the result of digesting the plasmid coding for wild type PABP with NcoI/HindIII followed by infilling and religation of the plasmid-containing fragment. P10 –542 was obtained by site directed mutagenesis of the P10 – 636 plasmid to replace the glutamic acid at position 543 with a stop codon. Mutagenesis was performed with the QuikChangeTM site-directed mutagenesis kit (Stratagene). Likewise, P237–542 was obtained from P237– 636 by the introduction of the same mutation. All plasmid constructions were confirmed by sequencing. Protein expression and purification has been described previously (25). Band-shift Assays—The RNA probes used in the band-shift assays were synthesized from linearized plasmids using T7 RNA polymerase and [␣-32P]ATP (Amersham Biosciences). Band-shifts were performed as described by Walker et al. (40) with minor modifications. Briefly, 10-l reactions were carried out in microtiter plates. The labeled probes were initially denatured by incubation at 95 °C for 3 min and then, following rapid cooling, were incubated with different concentrations of the recombinant proteins for 15 min on ice in the presence of binding buffer (5 mM Hepes, pH 7.2, 1.5 mM MgCl2, 2.5% glycerol, and 0.5 mM Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 (21) and also plays a role in the nuclear export of PABP bound to newly synthesized poly(A)-containing RNA (22). PABP expression is regulated at the translational level. PABP mRNA is a member of the 5⬘-terminal oligopyrimidine tract mRNA family (23), which includes mRNAs encoding components of the protein synthesis machinery. Translation of these mRNAs is linked to the growth status of the cell, and this regulation strictly requires a short sequence of polypyrimidines at their 5⬘ end (reviewed in Ref. 24). Control of PABP expression is also achieved through an autoregulatory mechanism whereby PABP binds to an A-rich tract present in the 5⬘-UTR of its mRNA and represses its own synthesis, when in excess over poly(A)⫹ RNA (25–28). The A-rich tract, 50 –70 nt long, is found in almost all PABP mRNAs (25, 29, 30). In the human mRNA, the 61-nt-long tract contains sets of 5– 8 A residues interspersed by 3– 6 pyrimidine residues and is located 73 nucleotides from the cap (23). Repression by PABP may be relieved when there is an increase in the intracellular poly(A)⫹ mRNA, or an increase in the length of pre-existing poly(A) tails. Addition of exogenous poly(A) mimics this effect and specifically stimulates PABP synthesis in vitro in the absence of mRNA synthesis (25), whereas overexpression of PABP in vivo leads to the translational repression of endogenous PABP mRNA (27, 28). The prototype mechanism for translational repression of mRNAs by 5⬘-UTR-bound proteins is that described for ferritin mRNA. Regulation of ferritin mRNA translation is mediated by a stem-loop structure called the IRE (iron regulatory element) and its binding factor, the iron regulatory protein (IRP; for reviews, see Refs. 31 and 32). The IRE-IRP complex represses ferritin mRNA translation by precluding the recruitment of the small ribosomal subunit (33). To be effective in vivo and in the rabbit reticulocyte lysate cell-free system, the IRE motif must be located within 60 nucleotides of the mRNA cap structure (34, 35). When placed further from the cap, the IRE-IRP complex delays productive scanning of the ribosomal subunit along the mRNA 5⬘-UTR, but without greatly compromising overall translation (36). In contrast, in plant or yeast extracts, which lack endogenous IRP, a cap-distal IRE-IRP complex can arrest ribosomal scanning and efficiently inhibit translation (36, 37). As to PABP-mediated translational repression, recent evidence indicates that, in HeLa cells, PABP bound to the A-rich tract in the reporter mRNA 5⬘-UTR, up to 200 nt from the 5⬘ cap, stalls the migration of the 40 S ribosomal subunit along the mRNA, preventing it from reaching the AUG and forming the elongating 80 S complex (38). In this study, we examined the requirements of the PABP molecule to act as a translational repressor of reporter mRNAs containing A-tracts in their 5⬘-UTR. We confirm the lack of positional requirement for effective inhibition by wild type PABP (38). Moreover, we observe that PABP variants lacking the C terminus are much less efficient in repressing the translation of mRNAs carrying cap-distal A-tracts. These PABP variants have affinities for poly(A) similar to that of the fulllength protein, but have a reduced ability to associate cooperatively to poly(A). We go on to show a correlation between the presence of the fourth RRM and part of the C terminus, but not including the PABC domain, and the ability of PABP to associate cooperatively to poly(A), multimerize, and enhance PABPmediated translational repression. Identification of a C-terminal PABP-PABP Interaction Domain 15 min at 37 °C, and the clarified supernatant was assayed for CAT and -galactosidase activities using standard methods (44). For the RNA expression analysis, 5-fold transfection reactions were carried out using the MSC-91 reporter plasmid with the highest concentration of effector plasmid tested. Approximately 15% of the transfected cells were processed and assayed for CAT and -galactosidase. The remaining transfected cells were extracted with TRIzol (Invitrogen), and the total RNA was digested with RNase-free DNase I (Amersham Biosciences) and then run on denaturing formaldehyde gels prior to Northern blotting. The following DNAs were used as probes: the 0.7-kb XbaI/HindIII fragment of pET-MS2, the 1.6-kb HindIII/BamHI fragment of pSV2-CAT, the 2.4-kb NcoI/SmaI fragment of pGEX2TPABP, and the 3.5-kb ScaI/NotI fragment of pCMV containing the -galactosidase gene. Probes were labeled with [␣-32P]dCTP using the Megaprime kit (Amersham Biosciences). RESULTS Requirements for Translational Regulation of Reporter mRNAs Containing 5⬘-UTR A-tracts—To study the role of the A-rich region of the PABP mRNA 5⬘-UTR in PABP-mediated translation repression, we previously used mRNA reporters containing A-rich sequences followed by the coding region of Xenopus cyclin A (25). Here, we first examined the effect of varying the length of the A-tracts upstream of the cyclin reporter mRNAs, by comparing the effect of 10, 20, and 61 A residues in the 5⬘-UTR. The longest tract is the same length as the entire A-rich region in the human PABP mRNA leader region. The assay is based upon the ability of endogenous reticulocyte PABP to repress translation of the A-tract containing reporter mRNAs. We have shown that the presence of the 5⬘-UTR A-tract is associated with a reduced translation efficiency of the reporter mRNAs, as a result of bound PABP, as compared with control RNAs devoid of the adenylate sequence. Addition of exogenous poly(A) stimulates the translation of these mRNAs in the reticulocyte system by titrating the PABP bound to their 5⬘-UTR (25). An internal control mRNA, which lacks any regulatory elements in the leader region, ⌬(1–263) PABP, is included in the translation assay (25). Translation of cyclin mRNAs containing in their 5⬘-UTR either 10, 20 or 61 A residues, at a fixed position relative to the cap (41 nt), was assayed in the absence or presence of increasing amounts of added poly(A) (Fig. 1A). First, we noted that, in the absence of poly(A) competitor, increasing the chain length of A residues progressively reduced the efficiency of translation of cyclin mRNAs, relative to the control mRNA, presumably reflecting enhanced PABP binding. The 10-A-containing cyclin mRNA was not affected by the addition of poly(A), except at the highest doses, which inhibited the translation of the various cyclin reporters and the control RNA. As has been described previously (25, 45), inhibition of translation by poly(A) presumably occurs through a depletion of the lysate PABP, because this inhibition can be reversed by addition of the purified protein (45). The translation of both the 20- and 61-A-containing cyclin mRNAs was stimulated at intermediate poly(A) levels, with similar degrees of stimulation observed for both mRNAs (Fig. 1B). These data are entirely consistent with the finding that PABP can bind oligo(A) sequences as short as 12 nt (11, 12) in vitro. Next, we investigated the importance of the position of the adenylate tract, relative to the 5⬘ end of the mRNA, to the translation of reporter mRNAs. Cyclin RNAs were obtained, which contained 61 A nucleotides placed at various distances from the cap, ranging from 10 to 166 nt. Stimulation of translation by added poly(A) was observed in all five distance reporter mRNAs, although the degree of enhancement was somewhat less efficient in the most cap-distal mRNAs, with A-tracts at 126 and 166 nt from the cap (Fig. 1 (C and D) and data not shown). This difference possibly reflects the presence of additional sequences in the cap-distal mRNAs, or a reduced efficiency of the translation of the mRNAs con- Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 dithiothreitol), 250 g/ml competitor E. coli rRNA, 100 mM KCl, 1 mg/ml BSA, and RNase inhibitor (2.5 units). Heparin was then added to 5 mg/ml followed by 2 l of loading buffer (48.5% glycerol and 0.5% each of bromphenol blue and xylene cyanol). Samples were then immediately run on nondenaturing polyacrylamide gels (acrylamide/bisacrylamide ratio of 60:1) in 0.5⫻ TBE, the gels having been previously pre-run for 15–30 min at 200 –300 volts. Electrophoresis was performed at 4 °C, followed by autoradiography. Pull-down Assays—The glutathione-Sepharose beads (Amersham Biosciences) were initially pre-washed with binding buffer (BB: 150 mM KCl, 5 mM MgCl2, 10 mM Hepes, pH 7.2, 0.2% Nonidet P-40) and saturated with 10 mg/ml BSA in BB for 30 min at 4 °C, followed by two more washes with BB. Approximately 20 l of the beads were incubated with 4 g of the GST-PABP variants in a final volume of 100 l for 30 min at room temperature. The beads were then washed two times with BB containing 4 mg/ml BSA and incubated with 10 l of translation extract for 30 min at room temperature and washed twice again with BB. Proteins bound to the beads were eluted by addition of SDS-PAGE sample buffer and the samples run on 15% SDS-PAGE, followed by Coomassie Blue staining and autoradiography. Labeled cyclin A and PABP were synthesized in the TNT T7 coupled reticulocyte lysate system (Promega) using 5 g/ml amounts of each plasmid. The labeled PABP deletions were obtained by linearizations of the wild type PABP cDNA with HindIII (1–237), MscI (1–358), or Tth111I (1– 432) (the numbers indicate the amino acids retained from full-length PABP), followed by transcription with T7 RNA polymerase in the presence of the cap analogue and translation in the rabbit reticulocyte lysate. In some cases, prior to their use in the pull-down assays, the reticulocyte lysates were treated with either 100 units/ml cobra venom RNase (RNase V1; Amersham Biosciences) for 30 min at 37 °C or with 250 units/ml micrococcal nuclease for 20 min at 20 °C in the presence of 1.5 mM CaCl2 followed by addition of EGTA to 6 mM. Both RNases efficiently degraded commercial poly(A) (data not shown). When specified, the lysates were also supplemented with E. coli rRNA (250 g/ml), poly(A) (5 or 20 g/ml), or poly(C) (20 g/ml). Pull-downs with the His-tagged proteins were carried out as described above but using the Ni-NTA resin (Qiagen). Labeled human eIF4G was obtained by subcloning the XbaI/HindIII fragment from plasmid pSK-HFC1 (41) into the same sites of the pBluescript KS vector (Stratagene), followed by linearization with HindIII and in vitro transcription and translation as described above. His-tagged human eIF4A from plasmid pET(His6-eIF4A) (42) and MS2/PABP-(372– 636) were expressed and purified on Ni-NTA resin as recommended. MS2 Tethering Constructs—The reporter plasmids MSC-15 and MSA-15 were obtained by digestion of the MSC-GH and MSA-GH plasmids (43) with XbaI/HindIII and ligation of the larger fragment to the BamHI/HindIII fragment encoding the CAT gene from plasmid pSV2-CAT. Both the vector and insert DNAs were in-filled with Klenow DNA polymerase prior to ligation. The MSC-91 plasmid was constructed by inserting a BamHI/XbaI insert from the polylinker region of plasmid pcDNA3 (Invitrogen) into the BamHI site of MSC-15. In this case, the BamHI ligation was performed first, followed by the in-filling of the protruding ends and a second round of ligation. To produce the effector plasmids, the DNA fragments encoding the various PABP deletions originally in the pGEX2T vector were first cloned into the pET-MS2 plasmid (21) to generate MS2-PABP fusions. Subcloning the in-filled BamHI PABP fragments from P372– 636 and P237–584 into the BamHI site of pET-MS2, also in-filled, yielded MS2/ PABP-(372– 636) and MS2/PABP-(237–584), respectively. The resulting plasmids were then digested with XbaI/EcoRV, in-filled, and the inserts transferred to the EcoRV site of the eukaryotic expression vector pcDNA3, under the control of the cytomegalovirus promoter. All cloning steps and the final coding frames were confirmed by sequencing. Transfection Assays—Approximately 4 ⫻ 105 human embryonic kidney cells (293-EBNA-BCRJ) were grown in each well of the 24-well dishes with Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal calf serum, streptomycin (100 g/ml), and penicillin (60 g/ml). Subconfluent cultures were transfected in DMEM without serum and antibiotics. Transfections were performed with 250 ng of reporter plasmid, 20 ng of pCMV control plasmid (Clontech), and 2.5, 5, or 10 ng of effector plasmid (for the first two concentrations of the effector plasmid DNA, the difference to 10 ng was supplemented with DNA from the empty pcDNA3 vector). The plasmids were transfected along with LipofectAMINE (Invitrogen) as recommended by the manufacturer, and the transfected cells were allowed to grow for ⬃36 h in DMEM plus 10% fetal calf serum and antibiotics. Prior to harvesting, the cells were washed twice with phosphate-buffered saline. Cells were lysed with lysis buffer (100 mM Tris-HCl, pH 7.8, 0.5% Triton X-100) for 46359 46360 Identification of a C-terminal PABP-PABP Interaction Domain taining longer UTRs in the reticulocyte lysate. We conclude that PABP-mediated repression in vitro requires more than 10 As in the leader region, and that it can operate even when the A-tract is distal to the cap. This result is in agreement with recently published data (38), as well as with the distance of 73 nucleotides from the cap to the A-tract in the human PABP mRNA (23). RRM 3/4 and the C Terminus of PABP Are Required for Efficient Cap-distal Repression—To assess the contribution of the different domains of PABP, including the C terminus, to its activity as a repressor of translation, a series of different deletions in the PABP gene were prepared and the resulting proteins fused to GST were expressed in E. coli. Fig. 2A shows a scheme describing the different deletions obtained during this study, each named according to the number of the amino acids retained from the wild type protein. The various proteins were purified by affinity chromatography and quantified by SDSPAGE (see Fig. 2B for a selection of the various proteins used in this study, as well as Fig. 5A (upper panel)). Wild type GST-PABP (P10 – 636) and the variant with the whole of its C terminus deleted (P10 –370) were initially tested in in vitro translation assays with the cyclin reporter mRNAs described in Fig. 1. First, the RNAs with differing lengths of A-tracts were tested with the full-length GST-PABP (P10 – 636) and the P10 –370 variant. Both proteins were able to repress the translation of RNAs containing either 61 or 20 A nt. As expected, neither significantly affected the translation of the reporter containing 10 A nt in the 5⬘-UTR (data not shown), in agreement with the results in Fig. 1A. In contrast, when the RNAs containing variations in the distance between the cap and the 61-nt A-tract were translated in the presence of increasing amounts of PABP variants, a significant difference could be seen between the two proteins (Fig. 3, A and B). Full-length PABP efficiently repressed the translation of all the distance constructs up to 126 nucleotides, but the PABP variant lacking the C terminus (P10 –370) was a much less effective repressor at A-tract distances greater than 41 nucleotides. This difference was not at the level of RNA binding, as P10 – 636 and P10 –370 were equally able to bind 32P-labeled probes consisting of the 5⬘-UTR of the different cyclin mRNAs in a gel-shift assay (Fig. 3C). Strikingly, the PABP variant P10 –190, which contains only the first two RRMs required and sufficient for specific binding to poly(A) (12, 14), could only effectively repress the translation of the reporter RNA containing the most cap-proximal A-tract, at the highest doses tested (Fig. 3, D and E). We conclude that efficient repression of cap-distal A-tract mRNA requires some or all of RRM3/4 and is greatly enhanced by the C terminus. Mapping the Domains Required for Cooperative Binding of PABP to Poly(A)—The PABP C terminus confers homodimerization activity on PABP, bound to poly(A), as shown by bandshift assays in which labeled A23 probes (sufficient to bind only one PABP molecule) formed multiple complexes at saturating concentrations of PABP, but not of a variant only containing the four RRMs (12). This band-shift strategy was used to assay for homodimerization of various GST-PABP proteins in the presence of a labeled poly(A) probe corresponding to the 61-ntlong PABP 5⬘-UTR A-rich sequence (Fig. 4A). From the work of Kuhn and Pieler (12), we reasoned that this sequence is capable of binding three (possibly four) PABP molecules. Upon incubation with increasing concentrations of GST-PABP, (ranging from 4 to 260 nM), we observed multiple shifted complexes indicative of sequential filling of binding sites in a cooperative manner (Fig. 4B, P10 – 636). We estimate the Kd of the PABP proteins bound to the labeled probe to vary between 5 and 15 nM, which are very similar values to those obtained previously (12, 13, 46). To map the region involved in the PABP/PABP interaction, GST fusion proteins containing Cterminal deletions (P10 –584, P10 –542, and P10 –370) were used in comparison to GST-PABP. In full-length PABP and the variants lacking part of or the complete PABC domain (P10 – 584 and P10 –542 respectively), binding of one molecule to the Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 1. Translational derepression by poly(A) of cyclin reporter mRNAs containing different poly(A) tracts in their 5ⴕ-UTR. Capped cyclin mRNAs (0.5 g/ml) containing A-tracts in their 5⬘-UTRs were translated in nuclease-treated rabbit reticulocyte lysate pre-incubated with increasing concentrations of poly(A) (0, 0.25, 0.5, 1, 2, 4, 8, 16, and 32 g/ml). These mRNAs were translated alongside the ⌬(1–263) PABP mRNA (0.25 g/ml) as an internal control. The cyclin mRNAs did not contain a 3⬘ poly(A) tail, and the distance between the A tract and the cyclin initiator AUG remained constant (70 nucleotides). A, translation profile of cyclin mRNAs with varied lengths of A-tracts (10, 20, or 61 A residues), at a fixed distance of 41 nucleotides between the cap and the A-tract. B, results from A were quantitated by densitometry and the intensities of the cyclin bands plotted as a function of the poly(A) concentration defining as 1 (dashed line) the value obtained in the absence of poly(A). C, translation of mRNAs containing 61-nt A-tracts in their 5⬘-UTR, located at varying distances from the cap (10, 41, 86, and 126 nucleotides) in reticulocyte lysate pre-incubated with increasing concentrations of poly(A) as in A and B. D, same as B but plotting the results shown in C. Identification of a C-terminal PABP-PABP Interaction Domain 46361 probe was an intermediate stage followed at higher concentrations by binding of two, three, or even more molecules. In contrast, P10 –370, although having an affinity to poly(A) similar to that of these proteins, differed significantly from them in that multiple binding of the protein to the probe RNA was substantially impaired. Thus, even at the highest concentration of P10 –370, a significant proportion of the probe was bound by only one molecule and two molecules seemed to be the most that could bind the probe (Fig. 4B). Similar results were observed in the case of an A61 RNA probe, although here we noted an increased efficiency of cooperative PABP/PABP interactions, which prevented detection of intermediate complex formation (data not shown). In the translation repression assay, the new variant P10 – 542, although lacking the PABC domain, was observed to be nearly as active as the full-length protein in the repression of reporter mRNAs with the A tract distal to the cap, with a reduction in repression efficiency only at the 126-nt distance (Fig. 4C). We conclude that the PABP C terminus, excluding the PABC domain, is involved in cooperative PABP/PABP binding in the presence of poly(A) and enhances PABP activity as repressor of translation. PABP C Terminus, the Fourth RRM, and Poly(A) Are Required and Sufficient for Specific PABP/PABP Interaction— The experiments above point to the region between amino acids 370 –542 as being involved in mediating PABP/PABP interactions and enhancing translational repression. To confirm these results, we used pull-down assays in which PABP variants were bound to glutathione-Sepharose beads and subsequently incubated in the presence or absence of poly(A) with 35S-labeled full-length PABP translated in rabbit reticulocyte lysate. A second 35S-labeled protein, cyclin A, was included in the reaction as a control to monitor for nonspecific bead binding. Following incubation, and washing, the proteins bound to the beads were analyzed by SDS-PAGE, and Coomassie Blue staining and autoradiography. The top panel in Fig. 5A shows that approximately equal amounts of GST-PABP variants were bound and eluted from the beads. As shown in the middle and bottom panels of Fig. 5A, weak and equal cyclin protein binding is observed in all lanes (this nonspecific binding was consistently enhanced following ribonuclease treatment, for unknown reasons). In the presence of poly(A), all N-terminal GST-PABP variants containing at least RRM1/2 pull down S-labeled PABP. Comparison with an aliquot of the initial translation mix (Retic lane) allows us to estimate that approximately 20% of the 35S-labeled PABP is bound by GST-PABPs. Variants P237–542 and P237– 636, which include a complete RRM4 plus the 370 –542 part of the C terminus, identified as necessary for cooperative binding to poly(A) (Fig. 4), or the whole of the C terminus, respectively, also interact with the translated PABP. In contrast, variant P372– 636, which only consists of the PABP C terminus, interacts very weakly. This level of binding was well above background binding; as shown in the control lanes, labeled PABP did not bind immobilized GST or glutathione-Sepharose beads. In the absence of poly(A), and with the addition of cobra venom RNase to degrade any remaining excess RNA, most of the fusion proteins, including full-length GST-PABP, lose the ability to interact with 35SPABP. Surprisingly, the P237–542 and P237– 636 variants still retain the capacity to bind to the labeled protein, suggesting that the removal of the first three RRMs from at least one of the binding molecules relieves an inhibitory mechanism, which prevents PABP/PABP binding in the absence of poly(A). The same results were obtained in several independent experiments, some using micrococcal nuclease in place of cobra venom RNase (data not shown). To rule out the possibility that the P237– 636 and P237–542 proteins bind full-length PABP through an RNA bridge (at least when poly(A) is present), we tested their ability to bind 32 P-labeled A61 RNA probes using band-shift assays (performed as described in Fig. 4B). The P372– 636, P10 – 636, and P10 –370 PABP variants were also included in this experiment as controls (Fig. 5B). Only the proteins containing functional RRMs 1 and 2 bound the A-tract probe, indicating that the P237– 636 and P237–542 variants bound full-length PABP directly. We next tested the ability of GST-PABP variants to bind truncated PABP proteins, labeled in vitro, to determine the minimal optimal interaction region. Three different mRNAs, coding for sequential C-terminal PABP deletions, which lack the PABC domain, the whole of the C terminus, and the fourth RRM plus the C terminus, respectively (see Fig. 2A for exact positions of the deletions), were initially co-translated in the rabbit reticulocyte lysate. Following translation, the reaction was treated with micrococcal nuclease, to remove any remain35 Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 2. Different GST-PABP fusion proteins used in this study. A, scheme of the GST-tagged PABP fusion proteins used in this study (named according to the amino acids remaining from the full-length human PABP). The position of the GST tag as well as the four RRMs and the PABC domain in PABP are indicated (see “Experimental Procedures” for details of the different proteins). Highlighted also are the positions of the sites for the enzymes HindIII (H), MscI (M), and Tth111I (T), which cut after the codons for amino acids 237, 358, and 432, respectively. B, Coomassie Blue-stained SDS-polyacrylamide gel of selected purified GST-PABP fusions used in this study. The various C-terminal and N-terminal deletions are shown on the left and right panels, respectively, with P10 – 636 and P10 –370 in both for comparison. On the left are indicated sizes in kDa of protein molecular mass markers. The dots indicate the position of the full-length, GST-PABP proteins. 46362 Identification of a C-terminal PABP-PABP Interaction Domain Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 3. Translation repression by GST-PABP variants of the cyclin mRNAs containing 5ⴕ-UTR A-tracts. The cyclin reporter mRNAs described in Fig. 1C were translated along with the ⌬(1–263) PABP mRNA (control) in the presence of increasing concentrations of GST-PABP proteins. A, comparison of the translation of the different mRNAs in the presence of increasing concentrations of full-length GST-PABP P10 – 636 (0, 1.6, 3.3, 6.5, 13, and 26 g/ml) or the P10 –370 variant (0, 1.4, 2.8, 5.6, 11.3, and 22.5 g/ml). B, the autoradiograms shown in A were quantitated and analyzed as in Fig. 1, but here for each experiment the ratio between the intensities of the cyclin and the control PABP bands was plotted as a function of the GST-PABP concentration, normalizing as 1 (dashed line) the value obtained in the absence of GST-PABP (non-normalized cyclin/control ratios of 3.05, 2.95, 1.2, and 0.55 for the 10-, 41-, 86-, and 126-nt distance mRNAs, respectively). C, band-shift analysis with the 5⬘-UTR of the cyclin mRNAs used in the translation assays. The various cyclin DNA constructs tested in A and B, linearized after the cyclin ATG (StyI site), were transcribed in the presence of [␣-32P]ATP. The resulting labeled RNAs (10,000 cpm or ⬃0.7 ng) were incubated with either wild type GST-PABP or the P10 –367 variant (14 g/ml final concentration) and the protein-RNA complexes analyzed on 4% nondenaturing polyacrylamide gels. ORF, open reading frame. D, translation repression assay of PABP variant P10 –190 (0, 2.6, 5.3, and 10.5 g/ml) with the 10-, 41-, and 86-nt distance cyclin mRNAs tested in A. E, the results from D were quantitated and plotted as in B (non-normalized cyclin/control ratios of 3.2, 2.8, and 2.15 for the 10-, 41-, and 86-nt distance mRNAs, respectively). The stimulation observed for the 86-nt distance in the last concentration (10.5 g/ml) is not reproducible and is only observed in cyclin/control ratio because of a small decrease in the control PABP band in the last lane of the experiment. In all translation assays, the concentration of the control mRNA was 0.25 g/ml. Cyclin mRNA concentrations: 10 and 41 at 2.5 g/ml; 86 and 126 at 3.75 g/ml. ing RNA, and excess ribosomal RNA was added as nonspecific competitor, after the addition of EGTA to quench micrococcal nuclease activity. The reaction was then divided into two aliquots, and either poly(A) or poly(C) was added prior to incubation with various bead-bound GST-PABPs (Fig. 5C). In the presence of poly(A), the three GST-PABPs bring down the three labeled proteins at equivalent levels, compared with their ini- tial reticulocyte levels. Indeed, as observed previously (Fig. 5A), P237– 636, although not able to bind poly(A) (Fig. 5B), is as efficient as the P10 –370 and P10 – 636 proteins that do bind poly(A) (Fig. 5C). Even the 1–237 labeled protein that lacks the entire C terminus and the fourth RRM is brought down in similar proportions to the longer proteins. (We also note that the minor labeled proteins, truncated versions of PABP arising Identification of a C-terminal PABP-PABP Interaction Domain 46363 from the use of internal AUGs or from premature termination, interact with immobilized PABP.) We cannot rule out a direct interaction between the various proteins but it is more likely that the added poly(A), which allows multiple molecules of PABP to associate, and the endogenous rabbit reticulocyte PABP, act as bridges for any poly(A)-binding protein, resulting in their association with the GST-PABP variants. In the presence of poly(C), as observed above after RNase treatment, neither the wild type nor the C terminus lacking variant (P10 – 370) can bind any of the labeled protein, whereas P237– 636 could relatively efficiently bring down the largest of these proteins (1– 432), which retains the first 50 amino acids of the C terminus. With further deletions (protein 1–358, which loses the remaining C terminus), this binding is severely impaired (3-fold reduction in binding), although some minor binding can still be seen even with the labeled protein 1–237 (⬃15% of that observed for protein 1– 432). Identical results were obtained with the GST variant P237–542 (data not shown). Together, these pull-down experiments confirm that the region of the C terminus immediately after the fourth RRM, but excluding the PABC domain, plays a significant role in mediating the interaction between PABP molecules. A role for RRM4 in this self-association is also implied by our findings, because it is required in the GST fusion for maximal interaction (compare P372– 636 and P237–542/636; Fig. 5A) and its removal from the labeled PABP deletion abolishes most of the remaining binding activity to the P237– 636 variant in the presence of poly(C) (Fig. 5C). Alternatively, the residual activity observed between P237– 636 and the labeled protein 1–237 may indicate minor binding to the other RRMs, even in the absence of RRM4. This latter observation is consistent with the overall sequence similarity between RRMs 2, 3, and 4 of ⬃45– 50% in human PABP, with RRM1 being more divergent. In summary, gel-shift assays and pull-down experiments indicate that RRM4 and the part of the C terminus, up to but not including PABC, mediates PABP self-association. This interaction is dramatically enhanced in the presence of poly(A); in the absence of poly(A), only truncated versions of PABP, lacking RRM1/2, retain the ability to bind other PABP molecules. Tethered PABP C Terminus Enhances Translation Repression by MS2 of mRNA Containing MS2 Binding Sites in Their 5⬘-UTR—To investigate the role of the C terminus in enhancing translation repression by PABP in vivo, we undertook a tethered strategy whereby MS2 binding sites were inserted in the 5⬘-UTR of CAT reporter genes and, in a second plasmid, the MS2 protein was fused to either the PABP C terminus (amino acids 372– 636) or its fourth RRM plus the proximal part of the C terminus (amino acids 237–542 missing the PABC domain), or used alone (Fig. 6A). The MS2 protein is known to repress translation/expression of reporter genes containing MS2 binding sites in their 5⬘-UTR, as long as it is placed within the first 40 or so nucleotides after the cap. It is not very efficient at preventing translation if the distance between the cap and the MS2 binding site is increased further (43, 47). Fig. 6 (B and C) Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 4. Cooperative binding to poly(A) by PABP does not require the PABC domain. A, A-rich tract from the 5⬘-UTR of the human PABP mRNA used as probe in the assays. B, band-shifts, performed by incubating the 32P-labeled A-rich probe with increasing concentrations of the PABP proteins P10 – 636, P10 –584, P10 –542, and P10 –370 and analysis on a 4% nondenaturing polyacrylamide gel. Protein concentrations used were 0, 0.38, 1.5, 6, and 24 g/ml. The arrows indicate the different protein-RNA complexes. C, translation repression assay with the P10 –542 variant. The 10-, 41-, 86-, and 126-nt distance cyclin mRNAs (Fig. 1C), as well as the control ⌬(1–263) PABP mRNA, were translated as described in Fig. 3 in the presence of increasing concentrations of P10 –542 (0, 7.5, and 30 g/ml). D, results from C were quantitated and plotted as a function of the recombinant protein concentration. 46364 Identification of a C-terminal PABP-PABP Interaction Domain describes the scheme of the CAT reporters and the MS2 fusion proteins. Two different CAT constructs were made with the high affinity MS2 binding site MSC placed at 15 and 91 nucleotides from the transcription start site, MSC-15 and MSC-91 respectively, and a third construct contained the low affinity MS2 binding site MSA placed at the 15-nucleotide distance, MSA-15 (47). Plasmids encoding the reporter genes and the protein fusions were co-transfected in 293 cells along with a third plasmid encoding -galactosidase to serve as transfection control. After 30 –35 h of transfection, the cells were harvested and assayed for CAT and -galactosidase activities. To compensate for variations in transfection efficiency, the ratios between the CAT and -galactosidase activities were determined and for each set of transfections, this ratio was compared with a control transfection where no MS2 expression plasmid was included (Ctrl). In some experiments, RNA was extracted as well and used for Northern blots to confirm approximately equal expression and stability of the different CAT and MS2 genes (Fig. 7D). First, the cells were transfected with the MSC-15 CAT reporter plasmid along with increasing concentrations of the plasmids expressing the different MS2 fusion proteins (Fig. 7A). All three proteins led to a dose-dependent decrease in CAT activity, with a maximum reduction of ⬃40%. A minor enhancement of repression was observed by the fusion proteins containing the PABP C terminus. In cells transfected with the reporters con- taining the mutated MS2 binding site (MSA-15), no reduction in CAT activity was observed for MS2 on its own and less than 20% reduction for the MS2-PABP C terminus fusions, showing that expression of the MS2 fusions is not causing a general nonspecific inhibition of CAT expression (Fig. 7B). We then analyzed the effect of the fusion proteins on the reporter CAT mRNA bearing a cap-distal MS2 binding site (MSC-91, Fig. 7C). Although less than 10% reduction of CAT activity was observed even with the highest dose of the MS2 plasmid tested, both fusions containing the PABP fragments were still capable of efficiently (⬃40% reduction) repressing CAT expression. These effects were largely at the level of translation, as we did not observe corresponding changes in RNA levels (Fig. 7D). The tethered strategy has also been used by us to specifically investigate reduction in CAT mRNA abundance as well as translation in 293 cells mediated by PABP binding to its 5⬘-UTR (48). Full-length PABP fused to MS2, like the MS2/C terminus fusions described above, despite reducing the expression of the CAT construct MSC-91, does not affect the reporter mRNA levels. Initially the transfection results were quite surprising, as we previously showed that the GST-PABP C terminus (P372– 636) seemed to have only a limited ability for PABP-PABP binding in the absence of poly(A) (Fig. 5A). Although we expected the intracellular system to be comparable to a poly(A)-rich envi- Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 5. Delineation of the region required for the interaction between GST-PABP variants and 35S-labeled PABP. A, GST-PABP fusion proteins were immobilized on glutathione-Sepharose beads in the presence of BSA and incubated with 35S-labeled wild type PABP and cyclin A translated in the rabbit reticulocyte lysate. The proteins bound to the beads were eluted in SDS sample buffer and analyzed in SDSpolyacrylamide gels by Coomassie Blue staining and autoradiography. The dots indicate the position of the full-length, undegraded, GST-PABP proteins. The assay was performed in the presence of 5 g/ml poly(A) or after treatment of the reticulocyte lysate with RNase V1 to eliminate any residual RNA. The lane marked Beads represents the sample where no protein was added to the beads. B, GST-PABP variants were tested for their ability to bind poly(A) in band-shift assays, carried out as described in Fig. 4A but using a 32P-labeled 63-A RNA probe and 5% nondenaturing polyacrylamide gels. As positive controls, PABP variants P10 – 636 and P10 –370 were also included. C, 35S-labeled PABP deletions were obtained by linearizing PABP cDNA with various restriction enzymes (HindIII for 1–237, MscI for 1–358, and Tth111I for 1– 432; the numbers indicate the amino acids remaining in the labeled proteins) followed by transcription with T7 RNA polymerase and translation in the rabbit reticulocyte lysate. The translation reaction was then treated with micrococcal nuclease and incubated with E. coli rRNA and poly(C) or poly(A) (both at 20 g/ml) prior to incubation with the GST-PABP variants indicated. In A and C, the lane marked Retic indicates an equivalent amount of the translation products used in the assay. Identification of a C-terminal PABP-PABP Interaction Domain 46365 N-terminal His tag and tested whether MS2-PABP-(372– 636) immobilized on Ni-NTA beads interacts with 35S-labeled PABP. As shown in Fig. 7E, this protein does indeed bind full-length PABP, even in the absence of poly(A). Control assays were also performed using as the labeled protein a human eIF4G variant (41), which lacks the motif required for binding PABP while retaining those required to bind eIF4A (16). No interaction between MS2-PABP-(372– 636) and labeled eIF4G is observed (Fig. 7E). In contrast, recombinant His-tagged human eIF4A can only recover traces of the labeled PABP, while binding very efficiently to labeled eIF4G. These experiments then confirmed that the C terminus on its own, or at least when positioned after an adequate fusion protein, mediates PABP/ PABP interaction and can enhance the limited activity of the RNA-binding protein MS2 in blocking the translation of mRNAs by the small ribosomal subunit at cap-distal positions. DISCUSSION ronment, we examined whether the N-terminal MS2 portion stabilized the fusion protein (MS2-PABP-(372– 636)) and enhanced its ability to bind full-length PABP. Therefore, we took advantage of the fact that the MS2 half of the protein has an Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 6. Tethering approach to investigate the role in vivo of the PABP C terminus in enhancing translational repression. A, experimental strategy used to test the effect of MS2-PABP fusions on the translation of CAT reporter mRNAs containing a MS2 binding site in their 5⬘-UTR. B, scheme of the different plasmids encoding the reporter mRNAs. Three different reporter plasmids were used, two coding for a high affinity MS2 binding site positioned at 15 and 91 nt from the cap (MSC-15 and MSC-91, respectively) and a third coding for a MS2 binding site containing a C to A mutation (MSA-15), which drastically reduces the binding of the MS2 protein to the mRNA (47). C, MS2PABP fusions tested in the assay. The results presented in this report contribute in multiple aspects to the study of the poly(A)-binding protein, with implications both in PABP function/synthesis as well as control of mRNA translation in general. By examining the requirements for PABP to act as a translational repressor, we confirm that PABP can be effective in preventing translation by binding A-tracts at locations both proximal and distal to the cap. In the latter case, PABP very likely blocks the scanning of the 40 S ribosomal subunit along the mRNA, as has been recently shown (38). However, the ability to bind poly(A) is not sufficient for efficient translational repression at distal sequences, because removal of the PABP C terminus somewhat reduces its ability to inhibit translation (Figs. 3 and 4). A second significant contribution from this work is the identification, within the PABP C terminus, of the region required for PABP/PABP interaction and cooperative binding to poly(A) (Figs. 4, 5, and 7). Major implications from this observation will be discussed below. Translation of PABP mRNA is controlled by at least two independent mechanisms: autoregulation mediated by the PABP protein (25–27), and coupling to the cellular growth stage mediated by the polypyrimidine stretch in its 5⬘ end (23). In this study, by using cyclin reporter mRNAs with the A-tracts in their 5⬘-UTR, we were able to concentrate on one aspect of PABP mRNA translation, avoiding the influence of other sequences present in PABP mRNA, which may affect its translation or stability (28). The A-rich sequence required for PABP mRNA translation repression differs from other sequences that have a similar function such as the IRE, in that it does not easily assume a secondary or tertiary structure, which may prevent ribosomes from scanning along the mRNA. The A-rich sequence also does not need to be positioned proximally to the cap structure (this work and Ref. 38). We reasoned that specific properties of PABP might be required for it to be an effective repressor and decided to use this activity as a tool to study the contribution of individual domains within PABP to its function. Full-length PABP represses the translation of cyclin mRNAs bearing A-tracts in their 5⬘-UTR, whether the A-tracts are proximal or distal to the cap, although with somewhat higher efficiency in the former case. On the other hand, repression by PABP variants lacking the C terminus is much more sensitive to the distance of the A-tract from the cap (Fig. 3). Thus, the C-terminal region of the PABP, although not interfering with binding to poly(A), is involved in the repression mechanism. Its role may be to increase the size of the complex bound to the RNA, not only by recruiting other PABP molecules but perhaps also by allowing the association of other proteins to create a multiprotein complex that blocks ribosomal scanning. Previous reports also hint at the contribution of the C terminus to PABP 46366 Identification of a C-terminal PABP-PABP Interaction Domain Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 7. Repression by MS2 of the CAT reporter mRNA bearing a cap-distal MS2 binding site is enhanced by tethering the C terminus of PABP. Plasmids encoding the different reporter mRNAs as well as the MS2-PABP fusions were co-transfected in 293 cells, and 30 –35 h later the cells were harvested and assayed for CAT and -galactosidase activity. Three different transfection reactions were performed for each experimental point, and the mean values and standard deviations were determined. The ratio of CAT to -galactosidase activity was set to 1 for the control cells transfected without any MS2 fusions. Results shown are representative of at least three independent transfection experiments. A, effect of the different MS2-fusions on expression of MSC-15 mRNA. Three different concentrations of the effector plasmids were used (2.5, 5, and 10 ng). B and C, same as A but using the plasmids encoding the MSA-15 and MSC-91 CAT mRNAs, respectively. D, Northern blots analysis comparing the mRNA levels of the CAT, -galactosidase (b-gal), and MS2 fusions in 293 cells transfected as in C with the CAT construct MSC-91. Probes used for the Northern blots are indicated. On the right panel are the results from the CAT/-galactosidase activity for the experiment used for the RNA analysis. This result is representative of several different experiments. E, pull-down assay to confirm binding of MS2-PABP-(372– 636) to 35S-labeled PABP. His-tagged MS2-PABP-(372– 636) was immobilized on Ni-NTA beads and incubated with 35S-labeled PABP or eIF4G (lanes 1 and 2). As a control, His-tagged human eIF4A was immobilized on Ni-NTA beads and incubated with 35S-labeled PABP or eIF4G (lanes 3 and 4). Ctrl, control. function. In Xenopus oocytes, a minimal PABP consisting of RRMs 1 and 2 did not seem to be able to bind efficiently to mRNAs or prevent their deadenylation when overexpressed in oocytes, in contrast to wild type PABP (49). In a HeLa-derived cell line, a similar PABP variant (as well as one containing the four RRMs but no C terminus) was still able to bind efficiently Identification of a C-terminal PABP-PABP Interaction Domain 46367 to poly(A) and shuttle to the cellular nucleus although its export back to the cytoplasm was impaired (22). The C-terminal region of PABP was previously reported to be involved in mediating protein/protein interactions, and more specifically PABP multimerization in the presence of poly(A) (12). The recent description of the PABC domain very much filled a gap in the knowledge about PABP function by mapping the motif involved in interactions with eRF3, PAIP1 and PAIP2 (19, 50, 51). Our results build upon these discoveries by confirming the participation of the C terminus in PABP/PABP interaction, while ruling out any participation of the PABC domain, and suggest a significant additional contribution of the fourth RRM. These results may relate to those obtained in yeast, where neither the first two RRMs nor the last 50 amino acids, which includes most of the PABC domain, are involved in mRNA stabilization by PABP (21). The fourth RRM is one of the least characterized of the PABP RRMs. An analysis of sequence conservation of RRMs from PABPs of distantly related organisms shows that RRM 4 is as conserved as RRM 2, which is known to be involved in such important functions as specific poly(A) binding and interaction with eIF4G. In contrast, RRM 3 is much less conserved. RRM 4 has been ascribed roles in cellular viability and in translation (see Introduction), but no interacting factors have yet been identified. Although its involvement in the protein/protein interactions necessary for PABP multimerization may in part explain its conservation (this work), it is also possible that it interacts with still unknown protein or RNA factors (18). The region of the C terminus bordered by RRM 4 and the PABC domain first came to notice when PABP sequences from yeast and human were compared, because of its high content of the amino acids proline and glutamine (52–54) (Fig. 8). Strikingly, in human PABP, roughly 1 in every 5 or 6 amino acids in this region is proline and 1 in every 3 is either glutamine, asparagine, or arginine, and there are no negatively charged amino acids in the entire 150-amino acid region. Alignments of the C terminus plus the fourth RRMs from PABPs of distantly related organisms shows no consistent sequence homology within this “linker” region, although the bias for the amino acids proline, glutamine, and, to a lesser extent, asparagine and arginine is maintained, as is the lack of acidic residues (Fig. 8). Until now, the strongest evidence pointing to the proline glutamine-rich linker region being involved in protein-protein interaction arose from a study of yeast PABP (Pab1p). Pbp1p (for Pab1p-binding protein) was obtained in a screen for factors that interact with the PABP C terminus. The specific interaction between Pab1p and Pbp1p does not require the last 24 amino acids of the PABP C terminus, which includes part of PABC (55). A likely candidate for mediating the Pab1p/Pbp1p interaction is a segment within the Pab1p linker region (italicized in Fig. 8) found to be partially homologous to two segments in the Pbp1p C terminus. Pab1p mutations that prevent the interaction map within this segment, which was proposed to be involved in homomultimerization (for both Pab1p/Pab1p or Pbp1p/Pbp1p multimers) or heteromultimerization (for Pab1p/Pbp1p multimers). Interestingly, removal of the RRMs from Pab1p actually enhances its binding to Pbp1p (55). This observation is reminiscent of the PABP pull-down assays described here, where removal of RRMs 1–3 appears to relieve an inhibitory constraint that prevents PABP/PABP interaction in the absence of poly(A). Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 FIG. 8. Sequence alignment of PABP regions containing RRM 4 and the C terminus from homologues of representative organisms. Protein homologues to the constitutive human PABP sequence (accession no. P11940) from selected organisms were chosen, and the regions encompassing RRM4 plus the C terminus (as defined in the human sequence) were aligned with the help of Blast and DNAstar software. Accession numbers: frog (Xenopus laevis), CAA40721; fly (Drosophila melanogaster), P21187; worm (Caenorhabditis elegans), CAA21572; plant (Arabidopsis thaliana), AAA61780; yeast (Saccharomyces cerevisiae), AAA34787. Residues identical to the human sequence are shaded. Human RRM 4 is boxed, and the PABC domain is overlined (19). The yeast Pab1p region involved in the interaction with Pbp1p (55) is in italics. The region in human PABP shown to be required for arginine methylation by CARM1 is underlined with double arrows indicating the two principal arginine targets (58). Single arrows indicate the last amino acids present in the truncated proteins shown in this study. Asterisks (*) indicate the prolines present in the human PABP linker region. 46368 Identification of a C-terminal PABP-PABP Interaction Domain Acknowledgments—Plasmids pSK-HFC1 and pET(His6-eIF4A) were a generous gift from I. Ali and R. Jackson. We thank A. Kornblight for the pSV2-CAT vector, M. Hentze for the MSC-GH and MSA-GH plasmids, and M. Wickens for the pET-MS2. We acknowledge members of the de Melo Neto, Standart, and Martins de Sa groups for helpful assistance and general discussion. REFERENCES 1. Sachs, A. B., Sarnow, P., and Hentze, M. W. (1997) Cell 89, 831– 838 2. Wickens, M., Anderson, P., and Jackson, R. J. (1997) Curr. Opin. Genet. Dev. 7, 220 –232 3. Preiss, T., and Hentze, M. W. (1999) Curr. Opin. Genet. Dev. 9, 515–521 4. Sachs, A. B. (2000) in Translational Control of Gene Expression (Sonenberg, N., Hershey, J. W. B., and Mathews, M. B., eds) pp. 447– 465, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY 5. Tarun, S. Z., Jr., and Sachs, A. B. (1996) EMBO J. 15, 7168 –7177 6. Wells, S. E., Hillner, P. E., Vale, R. D., and Sachs, A. B. (1998) Mol. Cell 2, 135–140 7. Caponigro, G., and Parker, R. (1996) Microbiol. Rev. 60, 233–249 8. Tucker, M., and Parker, R. (2000) Annu. Rev. Biochem. 69, 571–595 9. Bernstein, P., Peltz, S. W., and Ross, J. (1989) Mol. Cell. Biol. 9, 659 – 670 10. Wang, Z., and Kiledjian, M. (2000) Mol. Cell. Biol. 20, 6334 – 6341 11. Sachs, A. B., Davis, R. W., and Kornberg, R. D. (1987) Mol. Cell. Biol. 7, 3268 –3276 12. Kuhn, U., and Pieler, T. (1996) J. Mol. Biol. 256, 20 –30 13. Deardorff, J. A., and Sachs, A. B. (1997) J. Mol. Biol. 269, 67– 81 14. Deo, R. C., Bonanno, J. B., Sonenberg, N., and Burley, S. K. (1999) Cell 98, 835– 845 15. Kessler, S. H., and Sachs, A. B. (1998) Mol. Cell. Biol. 18, 51–57 16. Imataka, H., Gradi, A., and Sonenberg, N. (1998) EMBO J. 17, 7480 –7489 17. Otero, L. J., Ashe, M. P., and Sachs, A. B. (1999) EMBO J. 18, 3153–3163 18. Gray, N. K., Coller, J. M., Dickson, K. S., and Wickens, M. (2000) EMBO J. 19, 4723– 4733 19. Kozlov, G., Trempe, J. F., Khaleghpour, K., Kahvejian, A., Ekiel, I., and Gehring, K. (2001) Proc. Natl. Acad. Sci. U. S. A. 98, 4409 – 4413 20. Deo, R. C., Sonenberg, N., and Burley, S. K. (2001) Proc. Natl. Acad. Sci. U. S. A. 98, 4414 – 4419 21. Coller, J. M., Gray, N. K., and Wickens, M. P. (1998) Genes Dev. 12, 3226 –3235 22. Afonina, E., Stauber, R., and Pavlakis, G. N. (1998) J. Biol. Chem. 273, 13015–13021 23. Hornstein, E., Git, A., Braunstein, I., Avni, D., and Meyuhas, O. (1999) J. Biol. Chem. 274, 1708 –1714 24. Meyuhas, O., and Hornstein, E. (2000) in Translational Control of Gene Expression (Sonenberg, N., Hershey, J. W. B., and Mathews, M. B., eds) pp. 671– 693, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY 25. de Melo Neto, O. P., Standart, N., and Martins de Sa, C. (1995) Nucleic Acids Res. 23, 2198 –2205 26. Bag, J., and Wu, J. (1996) Eur. J. Biochem. 237, 143–152 27. Wu, J., and Bag, J. (1998) J. Biol. Chem. 273, 34535–34542 28. Hornstein, E., Harel, H., Levy, G., and Meyuhas, O. (1999) FEBS Lett. 457, 209 –213 29. Hotchkiss, T. L., Nerantzakis, G. E., Dills, S. C., Shang, L., and Read, L. K. (1999) Mol. Biochem. Parasitol. 98, 117–129 30. Bates, E. J., Knuepfer, E., and Smith, D. F. (2000) Nucleic Acids Res. 28, 1211–1220 31. Hentze, M. W., and Kuhn, L. C. (1996) Proc. Natl. Acad. Sci. U. S. A. 93, 8175– 8182 32. Rouault, T. A., and Harford, J. B. (2000) in Translational Control of Gene Expression (Sonenberg, N., Hershey, J. W. B., and Mathews, M. B., eds) pp. 655– 670, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY 33. Muckenthaler, M., Gray, N. K., and Hentze, M. W. (1998) Mol. Cell 2, 383–388 34. Goossen, B., Caughman, S. W., Harford, J. B., Klausner, R. D., and Hentze, M. W. (1990) EMBO J. 9, 4127– 4133 35. Goossen, B., and Hentze, M. W. (1992) Mol. Cell. Biol. 12, 1959 –1966 36. Paraskeva, E., Gray, N. K., Schlager, B., Wehr, K., and Hentze, M. W. (1999) Mol. Cell. Biol. 19, 807– 816 37. Koloteva, N., Muller, P. P., and McCarthy, J. E. (1997) J. Biol. Chem. 272, 16531–16539 38. Bag, J. (2001) J. Biol. Chem. 276, 47352– 47360 39. de Melo Neto, O. P., Walker, J. A., Martins de Sa, C., and Standart, N. (2000) Nucleic Acids Res. 28, 3346 –3353 40. Walker, J., de Melo Neto, O. P., and Standart, N. (1998) in Methods in Molecular Biology: Protein Synthesis: Methods and Protocols (Martin, R., ed) pp. 365–378, Humana Press, Totowa, NJ 41. Joshi, B., Yan, R., and Rhoads, R. E. (1994) J. Biol. Chem. 269, 2048 –2055 42. Pestova, T. V., Hellen, C. U., and Shatsky, I. N. (1996) Mol. Cell. Biol. 16, 6859 – 6869 43. Stripecke, R., Oliveira, C. C., McCarthy, J. E., and Hentze, M. W. (1994) Mol. Cell. Biol. 14, 5898 –5909 44. Bignon, C., Daniel, N., and Djiane, J. (1993) BioTechniques 15, 243–246 45. Grossi de Sa, M. F., Standart, N., Martins de Sa, C., Akhayat, O., Huesca, M., and Scherrer, K. (1988) Eur. J. Biochem. 176, 521–526 46. Gorlach, M., Burd, C. G., and Dreyfuss, G. (1994) Exp. Cell Res. 211, 400 – 407 47. Stripecke, R., and Hentze, M. W. (1992) Nucleic Acids Res. 20, 5555–5564 48. Melo, E. O., de Melo Neto, O. P., and Martins de Sa, C. (2003) FEBS Lett. 546, 329 –334 49. Wormington, M., Searfoss, A. M., and Hurney, C. A. (1996) EMBO J. 15, 900 –909 50. Roy, G., De Crescenzo, G., Khaleghpour, K., Kahvejian, A., O’Connor-McCourt, M., and Sonenberg, N. (2002) Mol. Cell. Biol. 22, 3769 –3782 51. Cosson, B., Couturier, A., Chabelskaya, S., Kiktev, D., Inge-Vechtomov, S., Philippe, M., and Zhouravleva, G. (2002) Mol. Cell. Biol. 22, 3301–3315 52. Adam, S. A., Nakagawa, T., Swanson, M. S., Woodruff, T. K., and Dreyfuss, G. (1986) Mol. Cell. Biol. 6, 2932–2943 53. Sachs, A. B., Bond, M. W., and Kornberg, R. D. (1986) Cell 45, 827– 835 54. Grange, T., de Sa, C. M., Oddos, J., and Pictet, R. (1987) Nucleic Acids Res. 15, 4771– 4787 55. Mangus, D. A., Amrani, N., and Jacobson, A. (1998) Mol. Cell. Biol. 18, 7383–7396 56. Hoshino, S., Imai, M., Kobayashi, T., Uchida, N., and Katada, T. (1999) J. Biol. Chem. 274, 16677–16680 57. Le, H., Browning, K. S., and Gallie, D. R. (2000) J. Biol. Chem. 275, 17452–17462 58. Lee, J., and Bedford, M. T. (2002) EMBO Rep. 3, 268 –273 59. Bedford, M. T., Frankel, A., Yaffe, M. B., Clarke, S., Leder, P., and Richard, S. (2000) J. Biol. Chem. 275, 16030 –16036 Downloaded from www.jbc.org at CAPES/MEC - UFPI on October 6, 2007 Evidence from various sources indicates that PABP activity can be regulated by different means. An example of such modulation has been observed in mammals, where binding of human eRF3 to PABP abolishes its ability to multimerize when binding to poly(A) (56). Phosphorylation of plant PABP has also been shown to play a role in regulating its binding to poly(A), because only the modified form binds poly(A) cooperatively (57). Theoretically, phosphorylation of the proline linker region could reduce its net positive charge and enhance PABP-PABP interaction. Recently, human PABP was identified as a substrate of the coactivator-associated arginine methyltransferase or CARM1 (58). Modification of target proteins by arginine methyltransferases has been shown to interfere with proteinprotein interactions, and, in one example, the arginine residues flank proline-rich motifs (59). Intriguingly, the region in PABP methylated by CARM1 has been mapped to the amino acids 384 – 478 (58), within the linker region that we show to be involved in PABP-PABP interaction (see Fig. 8). However, because the recombinant protein binds cooperatively to poly(A), at least for human PABP, neither methylation nor phosphorylation is probably required for the PABP-PABP interaction. Nevertheless in vivo, these modifications might play an important role in regulating poly(A)-binding and PABP-PABP interactions. Further work is still needed to elucidate these possibilities and to define precisely how the multiple PABP domains interact.