* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download HUMAN GENETICS ARCHITECTURE LEARNING OBJECTIVES
Tay–Sachs disease wikipedia , lookup
Y chromosome wikipedia , lookup
Gene expression profiling wikipedia , lookup
Human genome wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Biology and consumer behaviour wikipedia , lookup
Neocentromere wikipedia , lookup
Oncogenomics wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
Genome evolution wikipedia , lookup
Genetic engineering wikipedia , lookup
Epigenetics of human development wikipedia , lookup
Genomic imprinting wikipedia , lookup
Heritability of IQ wikipedia , lookup
Gene expression programming wikipedia , lookup
Frameshift mutation wikipedia , lookup
Skewed X-inactivation wikipedia , lookup
Human genetic variation wikipedia , lookup
History of genetic engineering wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Population genetics wikipedia , lookup
Behavioural genetics wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Neuronal ceroid lipofuscinosis wikipedia , lookup
X-inactivation wikipedia , lookup
Point mutation wikipedia , lookup
Medical genetics wikipedia , lookup
Dominance (genetics) wikipedia , lookup
Public health genomics wikipedia , lookup
Genome (book) wikipedia , lookup
Microevolution wikipedia , lookup
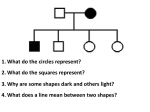
HUMAN GENETICS ARCHITECTURE LEARNING OBJECTIVES At the end of lecture students should be able to • • • • Review Genes and Human Diseases. Define Mutations. Review principals relating to the effects of gene mutations including : Point mutations within coding sequences Mutations within noncoding sequences Deletions and insertions Trinucleotiode-repeat mutations HUMAN GENETICS HUMAN CHROMOSOMES LINKAGE ANALYSIS • • • Traces the segregation of the trait through a family Traces the segregation of the chromosomes through a family Statistically measures the correlation of the segregation of the trait with the segregation of the chromosome SINGLE-NUCLEOTIDE POLYMORPHISMS (SNPS) One of the most common types of variation Multiple gene hypothesis: Range of phenotypes can be accounted for by cumulative effect of many alleles. Polygenes: Additive allele; nonadditive allele 1. phenotypic traits can be measured eg. weight or height 2. two or more loci (genes) could account for phenotype in an additive or cumulative way 3.each loci may be occupied by an additive allele, which contributes a constant amount to the phenotype, or a nonadditive allele which does not 4. The contribution by each allele may be small and is approx equal 5. Together the alleles contribute to a single phenotypic character with substantial variation. MULTIFACTORIAL TRAITS • Multifactorial traits are determined by multiple genetic and environmental factors acting together • Multifactorial = complex traits = quantitative traits • Most traits that vary in the population, including common human diseases with the genetic component, are complex traits • Genetic architecture of a complex trait = specific effects and combined interactions of all genetic and environmental factors. MUTATIONS • • • In molecular biology and genetics, mutations are changes in a genomic sequence: the DNA sequence of a cell's genome or the DNA or RNA sequence of a virus. They can be defined as sudden and spontaneous changes in the cell. Mutations are caused by radiation, viruses, transposons and mutagenic chemicals, as well as errors that occur during meiosis or DNA replication. MODES OF INHERITANCE • Autosomal Dominant – Huntington disease • Autosomal Recessive – Cystic fibrosis • X-linked – Duchenne muscular dystrophy • Mitochondrial – Leber Optic atrophy • Additive – HLA-DR in multiple sclerosis • Combinations of the above – RP (39 loci), Nonsyndromic deafness GENETIC DISORDERS • Genetic disorder is an illness caused by abnormalities in genes or chromosomes, especially a condition that is present from before birth. Most genetic disorders are quite rare and affect one person in every several thousands or millions. SINGLE GENE DISORDER • A single gene disorder is the result of a single mutated gene. There are estimated to be over 5000 human diseases caused by single gene defects. AUTOSOMAL DOMINANT INHERITANCE • • Autosomal traits are associated with a single gene on an autosome (non-sex chromosome)—they are called "dominant" because a single copy—inherited from either parent—is enough to cause this trait to appear. This often means that one of the parents must also have the same trait, unless it has arisen due to a new mutation. Examples of autosomal dominant traits and disorders are Huntington's disease, and achondroplasia. AUTOSOMAL RECESSIVE INHERITANCE • • Autosomal recessive traits is one pattern of inheritance for a trait, disease, or disorder to be passed on through families. For a recessive trait or disease to be displayed two copies of the trait or disorder needs to be presented. The trait or gene will be located on a non-sex chromosome. AUTOSOMAL RECESSIVE INHERITANCE • • • Because it takes two copies of a trait to display a trait, many people can unknowingly be carriers of a disease. From an evolutionary perspective, a recessive disease or trait can remain hidden for several generations before displaying the phenotype. Examples of autosomal recessive disorders are albinism, Cystic Fibrosis, Tay-Sachs disease. X-LINKED • • • • X-linked genes are found on the sex X chromosome. X-linked genes just like autosomal genes have both dominant and recessive types. Recessive X-linked disorders are rarely seen in females and usually only affect males. This is because males inherit their X chromosome and all X-linked genes will be inherited from the maternal side. Fathers only pass on their Y chromosome to their sons, so no X-linked traits will be inherited from father to son. X-LINKED • Females express X-linked disorders when they are homozygous for the disorder and become carriers when they are heterozygous. • X-linked dominant inheritance will show the same phenotype as a heterozygote and homozygote. • Just like X-linked inheritance, there will be a lack of male-to-male inheritance, which makes it distinguishable from autosomal traits. • One example of a X-linked trait is Coffin-Lowry syndrome, which is caused by a mutation in ribosomal protein gene. • This mutation results in skeletal, craniofacial abnormalities, mental retardation, and short stature. Y-LINKED INHERITANCE • • • Y-linked inheritance occurs when a gene, trait, or disorder is transferred through the Y chromosome. Since Y chromosomes can only be found in males, Y linked traits are only passed on from father to son. The testis determining factor, which is located on the Y chromosome, determines the maleness of individuals. Besides the maleness inherited in the Y-chromosome there are no other found Ylinked characteristics. QUANTITATIVE INHERITANCE • Quantitative traits = phenotypes differ in quantity rather than type (such as height) • In a genetically heterogeneous population, genotypes are formed by segregation and recombination • Variation in genotype can be eliminated by studying inbred lines = homozygous for most genes, or F1 progeny of inbred lines = uniformly heterozygous • Complete elimination of environmental variation is impossible MULTIFACTORIAL AND POLYGENIC(COMPLEX) DISORDERS Multifactorial disorders include heart disease and diabetes. • • • Although complex disorders often cluster in families, they do not have a clear-cut pattern of inheritance. This makes it difficult to determine a person’s risk of inheriting or passing on these disorders. Complex disorders are also difficult to study and treat because the specific factors that cause most of these disorders have not yet been identified. THANK YOU