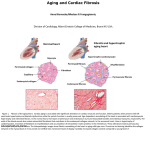
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Print - Circulation Research
Signal transduction wikipedia , lookup
Cell growth wikipedia , lookup
Cytokinesis wikipedia , lookup
Endomembrane system wikipedia , lookup
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cell encapsulation wikipedia , lookup
List of types of proteins wikipedia , lookup
Tissue engineering wikipedia , lookup
Circulation Research MAY 1976 VOL. 38 NO. 5 An Official Journal of the American Heart Association BRIEF REVIEWS Macromolecules of the Extracellular Compartment of Embryonic and Mature Hearts FRANCIS J . M A N A S E K , D.M.D. Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 THE FORM and function of the heart are the final result of an integration of cells, tissues, and extracellular material. The structure of many extracellular macromolecules is known, but studies of the cardiac extracellular matrix are still inadequate to provide a comprehensive understanding of it. The purpose of this review is to summarize the present knowledge of the cardiac extracellular matrix and to focus specific attention on this physiologically important compartment. It does not purport to be a detailed review of extracellular molecules; several excellent recent reviews exist.1"3 The three general types of macromolecules—collagen, glycosaminoglycan, and glycoprotein—which are characteristic of extracellular matrices probably differ qualitatively and quantitatively between different matrices and as a function of different ages, both pre- and postnatal, in the life of an animal. These three general classes of macromolecules will be described briefly after a review of some structural characteristics of the extracellular matrix. lamina (synonyms, basement lamina; basal lamella) is a subunit of the basement membrane which is visible, in most cases, only with the electron microscope (for terminology, see References 6 and 7) although some unusually thick basal laminae such as renal glomerular membrane and Descemet's membrane are prominent even at the light microscope level. To date all basal laminae whose composition has been determined appear to contain type IV collagen and glycoprotein,8 but there may be great variability both quantitatively and qualitatively. Sometimes the distinction between basement membrane and basal lamina is not made and the structure is simply called basement membrane. Confusion has resulted from inappropriate use of these terms and particularly when cell coats are called "basement membranes." In the heart, basal laminae surround capillaries, underlie the endocardium, and are present along the inner surface of the embryonic myocardium, whereas myocytes are surrounded by cell coats. Microscopic Appearance of Extracellular Space Chemical Composition of Extracellular Matrix Ground substance is the material occupying the apparently clear regions interspersed between cells and tissues. Under appropriate conditions of tissue preservation, ground substance is preserved, and elicits the metachromatic properties of certain biological stains such as toluidine blue or Alcian blue, and thus indicates the presence of polyanions. Embedded in ground substance are collagen and elastin fibrils: this results in a complex, multiphasic system. Cell surfaces are covered by a layer of extracellular material quite distinct from the ground substance.4 This layer, considered to be glycoprotein on the basis of histochemical staining and called the cell coat or external lamina, envelops cells without regard for polarity and is found on mesenchymal cells as well as epithelial cells. It also has been called the glycocalyx.* Cell surface coats should not be confused with basement membranes. Basement membranes are complex extracellular layers of collagen, glycosaminoglycan, reticular fibers (a form of collagen), and glycoproteins that underlie the basal surface (as opposed to apical, or free surface) of epithelia. The basal Molecular collagen (tropocollagen) is a protein that consists of three discrete subunits called a chains. The presence of covalent linkages between a chains reduces their extractability (as in the case of bone collagen). Lysine, OH-lysine, and their aldehyde derivatives are involved principally in cross-linking. There are two general kinds of a chains, termed a\ and al. Only one type of al chain has been detected, but there are at least four known a l chains, designated respectively al(I), al(II), al(III), al(IV). These distinctions are based on differences in molecular weight, posttranslational characteristics and both amino acid composition and sequence (for discussion see Reference 1). Different types of collagen are distinguishable on the basis of their a chain composition. Type 1 collagen has two a 1(1) chains and one al chain and is designated |al(I)] 2 a2. All the other known collagens consist entirely of a l chains. Different collagens are not uniformly distributed in extracellular matrix (Table I). It is important to stress that collagen need not assume the fibrillar, cross-banded form that occurs when tropocollagen molecules are assembled in a linear, near quarter staggered array. Thus, absence of histologically demonstrable collagen bundles or fibrils cannot be equated with the absence of From the Department of Anatomy. The University of Chicago, Chicago, Illinois. Original work reported in this review was supported by Grant HL 13831-04 and by the Louis Block Fund, The University of Chicago. 332 CIRCULATION RESEARCH VOL. 38, No. 5, MAY 1976 TABLE I. Chain Composition and Major Distribution of Different Collagen Types Chain composition Major locations (mature tissues) Typel [al(l)] s o2 Skin, bone, tendon Type 11 (al(II)la Cartilage Type III Type IV [«I(IID], tal(IV)], Skin, dentin, aorta Some basal laminae such as lens capsule, Descemet's membrane. renal glomerular and alveolar membranes Collagen type Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 collagen per se, nor can the appearance of fibrils be used to determine onset of synthesis of molecular collagen. Collagen types (Table 1) can be identified by denaturing extracted tropocollagen and identifying a chains according to a variety of criteria including size, level of proline hydroxylation, and amino acid composition. Most useful is the technique whereby collagen a chains are broken at specific sites by cyanogen bromide into peptides. CNBr peptides derived from unknown a chains can be compared to those of known standards. Glycosaminoglycans (mucopolysaccharides): Glycosaminoglycans (GAGs) are major constituents of ground substance. The GAG molecule is a protein-polysaccharide. As a matter of convenience we can divide GAGs into sulfated and unsulfated species. Polysaccharide moieties of different GAGs are long, unbranched chains of repeating dissacharide subunits. Carboxylate or ester sulfate groups situated along the length of the polysaccharide chain result in an overall net negative charge on the molecule. The polyanionic nature of GAG polysaccharides results in their histological demonstrability by means of metachromasia. In physiologically normal situations these negatively charged polysaccharides are associated with cations (counterions), generally Na + . Glycoproteins, like GAGs, consist of protein and CHO moieties. Unlike those of GAGs, glycoprotein carbohydrate moieties can be branched and contain a greater variety of carbohydrates. Noncollagenous glycoproteins are major consistuents of basal laminae 8 - 9 and probably also of cell coats (glycocalyx or external lamina). 4 Relatively little is known about the distribution and composition of embryonic matrix glycoproteins. 10 Some glycoproteins are associated with cell surfaces and may provide a degree of specificity to cell surfaces which may be important in cell-cell interactions during ontogeny."- l2 Extracellular Matrix in the Fully Developed Heart COLLAGEN Fibrillar collagen occupies 4% of the extracellular space. 13 Native cardiac collagen is virtually nonextractable in NaCl or dilute acid. 14 Bovine myocardial collagen (unspecified age) may be composed of two genetically distinct molecules: [al(I)]ja2and (a\), with the latter a l chains most similar to ar 1(1 II). Alpha chains analogous to al(II) or the basal lamina collagen, al(IV), were not detected in this analysis14 of total myocardial collagen. This is surprising since basal Typical morphological appearance in situ Fibrillar; "typical" 640 A periodicity Fibrillar; may or may not demonstrate periodicity Fibrillar Never fibrillar; may be filamentous component of basal laminae laminae are abundantly present in heart tissue. Myocardia from different regions of the heart were not examined separately. VALVULAR COLLAGEN Heart valves are derived largely from endocardium, via endocardial cushions. 15 ' 17 Morris and McClain 19 showed that valvular collagen (veal calf atrioventricular values) is not extractable in 1 M NaCl or 0.5 M acetic acid. On the basis of CNBr peptide analysis some homology between valvular collagen and the [al(II)] s collagen of cartilage was suggested. This apparent molecular homology is intriguing in light of the report of presumably pathological hyaline cartilage formation within human aortic valve19 as well as the normal presence of the fibrocartilaginous corpora Arantii within each leaflet. Absence of detectable amounts of type IV collagen in valvular tissue is surprising. The endothelium covering the valvular surface has an unusually thick basal lamina,20 and the techniques employed should have been adequate to recover and detect type IV collagen. GLYCOSAMINOGLYCANS IN VALVE TISSUE Early compositional analyses of bovine valves21- " showed the presence of hyaluronate and confirmed the presence of chondroitin sulfate and dermatan sulfate. The autoradiographic studies of Mancini et al. 23 showed that adult rat valves incorporate inorganic sulfate-35 and suggested the synthesis of sulfated GAG. Autoradiographic analysis of sulfate incorporation in guinea pig valves24 showed a decrease in both incorporation of sulfur-35 and turnover of sulfated extracellular molecules with age. An increase in hyaluronidase-resistant materials was correlated with age. No significant differences were noted between different valves. In addition to age-dependent changes in apparent synthesis, the composition of valvular GAGs changes with age. Moretti and Whitehouse 25 showed the presence of hyaluronate, chondroitin sulfate-6, and dermatan sulfate in both bovine and porcine valves. Using all valves from 2- to 6-day-old calves and 8- to 10-year-old cattle, they demonstrated a decrease in hyaluronate, a greater decrease in chondroitin sulfate, and a distinct increase in dermatan sulfate with age. The presence of hyaluronate, chondroitin sulfate, and dermatan sulfate in bovine valves was confirmed26 and later studies 27 suggested that dermatan sulfate might be associated with collagen, thereby decreas- CARDIAC EXTRACELLULAR ing the extractability of both the dermatan sulfate and the collagen. NONCOLLAGENOUS GLYCOPROTEINS Noncollagenous glycoproteins of the mature myocardium have not been characterized fully but are almost certainly part of the cell surface coat. 28 Because of their apparent importance in Ca 2+ flux29 it is unfortunate that more is not known about these molecules. Valve interstitium contains structural glycoproteins that may prove to be important in disease states. A review of this topic appeared recently.30 ELASTIN Elastic fibers have been identified in mature hearts by routine histological criteria at both the light and electron microscopic level. Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 The Embryonic Cardiac Extracellular Matrix The early tubular heart contains a prominent sleeve of extracellular matrix (cardiac jelly) located between the developing myocardium and the endocardium. Light microscopic studies of fixed material reveal the presence of thin strands of material in cardiac jelly. Migrating endocardial cushion cells are associated with these strands. 18 An electron microscopic study of a human embryo (crown-rump length, 9 mm) identified fibrillar matrix inclusions associated with endocardial cushion cells. 31 More recently a variety of morphological techniques have shown the association of filamentous material with rat endocardial cells.17 Studies using the electron microscope reveal a myocardial basal lamina that develops concomitantly with the onset of myofibril assembly and contraction. Fibrillar, cross-banded collagen is first detectable at about the same time (ca. 10 somites in chick development). Other electron microscopically detectable components 32 remain even more obscure. Clearly, microscopic examination is not adequate to define the composition of these regions. COMPOSITION OF EARLY EMBRYONIC EXTRACELLULAR MATRIX Relatively small tissue size makes compositonal analysis difficult and if careful staging of embryos is to be done, such analysis is virtually impossible. Recourse to histochemical techniques33 demonstrates the presence of acidic mucopolysaccharides, and early autoradiographic studies34 showed incorporation of sulfur-35 into sulfated matrix components. Autoradiography using staged embryos showed pronounced labeling of cardiac matrix in the chick using 3 S S O 4 " " or 3 H-glucosamine 3e and in the frog using 3S SO 4 ". 37 Other studies38- 3" also suggested the presence of glycosaminoglycans. Investigation of both total composition and labeled molecules with sulfur-3540-41 demonstrated the likely presence of chondroitin sulfate and heparan/heparin sulfate in the hearts of stage 16-19 (ca. 52 hours) chick embryos. A histochemical study42 identified hyaluronate (or an unsulfated carboxylic mucosubstance) and a number of sulfated GAGs in young rat hearts; concentration was greatest near the developing endocardium. The synthesis of hyaluronate, chondroitin, and undersulfated chondroitin sulfate (on the basis of charge/mass ratio compared to COMPARTMENT/Manasek 333 standard chondroitin sulfate, as determined electrophoretically) in hearts to stage 11(13 somite, ca. 45 hours embryos) and, in addition, fully sulfated chondroitin sulfate after stage 11 was recently shown.43 Heparan sulfate synthesis was not detected in these studies which used hearts that did not yet have endocardial cushions. Glycosaminoglycan synthesis during the remainder of cardiogenesis is poorly characterized. Hyaluronate synthesis was detected in 4-day-old hearts labeled in vitro 44 and sulfate incorporation was shown to vary as a function of age. 45 COLLAGEN An early compositional study48 demonstrated the presence of collagen in chick ventricles from day 10 of incubation to 21 days after hatching and suggested that prior to day 19 of incubation collagen accumulated at a rate slower than that of contractile proteins (measured as actomyosin). After day 19, collagen and actomysin increased at nearly identical rates. A later study47 detected collagen in hearts as early as 3 days of incubation, although 5-day-old hearts were the youngest that contained enough collagen to be measured quantitatively (their OH-proline analyses required 0.5 g total tissue) and showed that collagen is formed faster than net weight increases, indicating that it constantly forms a greater proportion of the heart. Salt-soluble collagen remains constant (30 M8/g w e t weight) throughout development. However, since the total collagen per gram of tissue increases constantly, the finding that soluble collagen per gram remains constant can be interpreted only as indicating relatively less collagen is soluble as the heart matures. Soluble collagen was calculated to be about 6% at day 5, decreasing to 1.5% at day 21 (hatching) and was proportional to newly synthesized collagen, suggesting that newly synthesized collagen is soluble in neutral salt. These interpretations agree with the seemingly contradictory findings of McClain,14 who found that mature myocardial collagen was virtually insoluble in NaCl, and Johnson et al., 32 who found that newly synthesized collagen in the very early embryonic heart (made lathyritic) was soluble in neutral NaCl solution. To date there has been only one systematic study 32 of embryonic cardiac collagen synthesis at the time of initial heart formation. This investigation correlating microscopic and biochemical findings for the chick detected crossbanded collagen fibrils at about stage 10 (10 somites, 30 hours) and the synthesis of type I-like collagen (detected biochemically) in lathyritic specimens. Unfortunately total hydroxyproline synthesis was not measured, so efficiency of extraction (I M NaCl, 4°C, 3 days) was not determined. This deficiency in the study becomes important because synthesis of type IV collagen was not detected even though this study encompassed the time that myocardial basal laminae were being formed.32-48- *" Interestingly, type IV-like collagen was not detected in the compositional studies by McClain, who used mature myocardium 14 and valves.19 Data available to date suggest therefore that type IV collagen is not present in detectable amounts in the cardiac interstitium. Embryonic hearts used by Johnson et al. 32 were all prevalvular (pre-cushion). Thus it seems likely that the type I I-like valvular collagen is synthesized after these structures ap- 334 CIRCULATION RESEARCH pear. The decrease in collagen extractability which correlates with age may reflect the interaction of collagen with glycosaminoglycan. 27 NONCOLLAGENOUS CLYCOPROTEINS Embryonic cardiac myocytes are surrounded by a cell coat which is demonstrable by use of ruthenium red (a cationic dye that binds to polyanions 10 - 50 ) and which probably represents glycoprotein. It is not known if these surface coats change in composition during cardiogenesis. Differences in the staining properties of surface coats of different types of cells within the myocardium have been noted" but the significance of, and time of onset of, such differentiation is not known. Direct evidence for the synthesis of glycoprotein recently has been obtained for early embryonic hearts49- " but they have not yet been fully characterized. Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 ELASTIN It is not known when during development elastin is first synthesized. Origin of Cardiac Extracellular Macromolecules It is known that a vast number of non-"connective tissue" cell types make many extracellular macromolecules. Although it seems likely that fibroblasts produce some, if not most, of the collagen synthesized in mature hearts it should be stressed that the myocardium is a heterogeneous structure containing vascular endothelium, vascular smooth muscle, nerves, and fibroblasts in addition to cardiac muscle. It is not possible to determine, at the present time, whether any of these other cell types contribute significantly to the extracellular matrix. It would be most interesting to determine the source of the type III -tike a chains. There are ontogenic events in the chronology of myocardial cellular development that may provide indirect insight into the course of the different cardiac collagens. Type I-like collagen is synthesized at a time when the myocardium contains only developing muscle cells.32 The only other cell type present in the heart at these stages is primitive endocardium."• " • 5 4 It is most likely that the developing cardiac myocytes are elaborating the early type I-like collagen. This conclusion is consistent with phylogenetic evidence. Tunicate (hemichordates) hearts lack an endocardium yet have a submyocardial "cardiac jelly" 55 which contains cross-banded collagen fibrils (see Fig. 5, Reference 55). Anatomical considerations make it unlikely that this collagen was derived from any source other than the myocardium. Parenthetically, the rest of the tunicate cardiac matrix is probably also a myocardial product. Seemingly, endocardium is not necessary for the production of the rudimentary cardiac matrix. Endocardia! cushions, the precursors of valves, appear at discrete times in development. It is not known whether this corresponds to the onset of valvular ( a l ) 3 (presumably type I I-like) collagen synthesis. Later in development (in the chick, by about day 4) the other cell types rapidly make their appearance. 53 Two basic questions that need to be answered are: (1) Does the total collagen synthesized by each cell remain constant during this transition to a heterogene- VOL. 38, No. 5, MAY 1976 ous population? (2) Is this the time of appearance of the (a\)» collagen molecule? The use of cell culture techniques to answer these questions should be attempted with great caution. It is, of course, possible to grow cells in culture and assay for collagen synthesis. This approach would reveal whether a cell type made collagen in culture but could not reveal whether that same cell type made it in situ. It is hazardous to ascribe in situ equivalence to cell functions discovered in an in vitro experiment. Because of the difficulty of providing a suitable experimental control to the explanation procedure per se (the control, of course, is in situ), attempts to describe the molecular composition of a matrix solely by in vitro studies of synthesis are open to question. It is suggested here that minimum acceptable criteria for such use of cultured cells (or tissues) would require that the total product of the cells grown in vitro account for the total composition, qualitatively and quantitatively, of the normal matrix in situ. Note that this requires preexisting knowledge of normal matrix composition but permits circumvention, albeit by circumstantial argument, of the usual inability to show that cells make in situ what they make in culture. Glycosaminoglycans are produced by undifferentiated cardiac myoblasts as well as by immature cardiac myocytes 3S. 36, 43, 52 labeled in situ. Autoradiographic studies also demonstrate myocardial labeling in vitro in chick40 and in frog embryos. 37 Electron microscopic histochemistry has recently shown acidic glycosaminoglycans in both myocardium and endocardium. 58 The extensive studies by Okamoto and his colleagues31 implicate the rat endocardial cushion cells in production of sulfated matrix. Ueno57 has quantified sulfate incorporation by rat cushion, but noted that little label was seen over myocardial or noncushion endocardial cells. Seemingly, endocardial cushion tissue becomes a major source of matrix glycosaminoglycan as the myocardial secretory activity declines. The heparin (probably heparan sulfate) detected by Gessner and his colleagues 40 - 41 may represent a maturational change in matrix composition since it was not found in younger hearts. 43 It is not known when synthesis of valvular dermatan sulfate25 begins. It is also not known whether heparan sulfate synthesis continues through development and, if so, where it is localized. Heparan sulfate has not been detected in mature valves. Neither the cells producing dermatan sulfate nor heparan sulfate have been identified, although it may be presumed that valvular dermatan sulfate is synthesized either by the endocardium or the endocardial-derived cushion cells. Matrix contributions from visceral epicardium also are poorly understood. Fibrillar collagen appears in the subepicardial space during development 53 but no other data, compositional or synthetic, are available. Subendocardial connective tissue contains elastic fibers, fibrillar collagen with a periodicity of 400-580 A, and ground substance, 58 all of which may be the product of endocardium, fibroblasts, smooth muscle (origin unknown), or unidentified cells. The Role of Matrix in Congenital Cardiac Abnormalities There is a prevailing thought that some congenital abnormalities may result from abnormal matrix production. CARDIAC EXTRACELLULAR Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 Salicylates inhibit incorporation of 35SO«" into valve glycosaminoglycan59 and are teratogenic. 60 Other teratogenic agents such as 6-aminonicotinamide, aminopterin, and trypan blue reduce incorporation of sulfate when administered in vivo or, in the case of trypan blue, also depress sulfation in vitro. 61 These are all studies of incorporation and it is not known whether molar or mass synthesis was depressed since end-product specific activity was not determined. The possibility that the teratogens had a principal effect elsewhere and depressed sulfate incorporation only as a secondary, and possibly trivial, side effect was not ruled out. In contrast to the decrease in sulfate incorporation resulting from administration of chemical teratogens, Ueno 57 showed that after 14.1-meV fast neutron irradiation (130 rads, a cardioteratogenic dose), sulfate incorporation was actually increased over that of controls. There is no simple relationship between sulfur incorporation and cardiac malformations and no evidence exists proving that teratogens alter mass or molar synthesis of cardiac glycosaminoglycan. Pharmacological inhibition of collagen synthesis' 2 inhibits early cardiac morphogenesis but since it is impossible to prove that inhibition of collagen synthesis was the principal, causal lesion these studies were terminated. Studies on cells in vitro have implicated some extracellular components in the contraction and "spreading" of cultured heart cells63 and suggesting the dependency of some physiological parameters on matrix macromolecules. Matrix composition can change in several ways. A given cell type (e.g., cardiac muscle) might change its synthetic pattern, its degradative activity (qualitatively or quantitatively), or both. Changes in cell populations must be TABLE 2. Cardiac Matrix considered also, since different cell types (e.g., fibroblasts, cardiac muscle, endocardium) may make different matrix components at different rates, at different times. It is known that myocardial cell populations change during ontogenesis53 but little is known about cell population changes in the neonate, juvenile, or mature myocardium. We cannot ignore these questions if matrix dynamics are to be understood. Most important, cell population heterogeneity must be considered in establishing culture analogs for both physiological and biochemical investigation lest uncontrolled variables obscure interpretation. Conclusions The extracellular matrix of the heart is not a static, uniform compartment. Its composition seems to change even in mature hearts, and these changes may be important in controlling development and function at the level of both the cell and organ. For example, early in development the elastic properties of the matrix seemingly are essential for diastolic filling.' 5 - 64 At this stage the elasticity is probably largely a function of the hyaluronate present. *3 This suggests that a single type of extracellular molecule can play a critical role in early cardiac physiology. It is tempting to propose that continued presence of these molecules also contributes to the elasticity of the myocardium in older hearts. Comparison of the matrix of normal hearts and those with acquired disease may provide new insight into the management of heart disease. Similar comparisons between normal and congenitally malformed hearts may provide insight into the etiology of such abnormalities. Both qualitative and quantitative data are needed. The provocative observation Principal fate of labeled precursor* • fnll'ipen 3 synthesized' H-GlcN "SO.- synthesized Noncollagenous glycoprotein HA,Ch, UChS HA.Ch UChS Type 1 -like; other types (?) Yes (not characterized) HA.Ch, UChS, ChSt HA, Ch UChS, ChS Yes; type(s) not characterized Yes (not characterized) Older than 18somite chick; by implication. mammals also HA, ChS, HS(H?), DS (?), probably HA ChS, HS, DS Yes; type(s) not characterized or localized Yes (not characterized) Postnatal (mammals)t HA, ChS, HS(?), (?) ChS, HS, DS Presumably yes Yes Stages 9 - I I (7-13 somites, 29-45 hr) labeled in situ (chick) Stages 11-13 (1318 somites, 40-51 hr), labeled in situ (chick) 335 Macromolecules GlycosaminDevelopmental age COMPARTMENT/Manasek DS Cell types and tissues present in heart Differentiating myocytes; endocardium; no "connective tissue" cells Differentiated, functional cardiac myocytes; endocardium; no "connective tissue" cells Cardiac myocytes; endocardium; epicardium: vascular components; fibroblasts; valvular tissue Same • HA = hyaluronate, Ch => chondroitin, UChS = undersulfated chondroitin sulfate, DS _ dermatan sulfate, ChS = chondroitin sulfate, 'H-GlcN = D-glucosamineo-'H. t Similar glycosaminoglycans have been identified histochemically in rat heart, but no synthetic data are available. $ Synthesis has not been studied to any significant extent. These data are derived largely from studies of composition that have been assumed to represent synthesis also. CIRCULATION RESEARCH 336 that hyaluronidase, an enzyme known to hydrolyze specific glycosaminoglycans, can alter the course of experimental myocardial infarcts" suggests yet another way in which the matrix can influence myocardial responses previously thought to reside exclusively in myocardial cells. The regional distribution of different macromolecules also should be considered if their functions are to be elucidated. Finally, the dynamics of the cell populations that produce matrix must be fit into the overall picture. Table 2 is a brief summary of the present status of cardiac matrix studies; it should be considered principally as a demonstration of gaps in our knowledge of the acellular compartment of the heart. References Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 1. Gallop PM, Blumenfeld OO, Seifter S: Structure and metabolism of connective tissue proteins. Annu Rev Biochem 41: 617-672, 1972. 2. Schubert M, Hamerman D: A Primer on Connective Tissue Biochemistry. Philadelphia, Lea and Febiger, 1968. 3. Balazs EA (ed): Chemistry and Molecular Biology of the Intercellular Matrix, three volumes. New York, Academic Press, 1970. 4. Rambourg A, Le Blond CP: Electron microscope observations on the carbohydrate-rich cell coat present at the surface of cells in the rat. J Cell Biol 32: 27-53, 1967. 5. Bennett HS: Morphological aspects of the extracellular polysaccharides. J Histochem Cytochem 11: 15-23, 1963. 6. Bloom W, Fawcett DW: A Textbook of Histology. Philadelphia, W. B. Saunders, 1969, p. 88. 7. Fawcett DW: An Atlas of Fine Structure. Philadelphia, W. B. Saunders, 1966, pp 353-364. 8. Kefalides N: Structure and biosynthesis of basement membranes. Int Rev Connect Tissue Res 6: 63-104, 1973. 9. Spiro, RG: Glycoproteins. Annu Rev Biochem 39: 599-638, 1970. 10. Manasek FJ: The extracellular matrix; a dynamic component of the developing embryo. Curr Top Dev Biol 10: 35-102, 1975. 11. Turner RS, Burger MM: The cell surface in cell interactions. Ergeb Physiol68: 121-155, 1973. 12. Moscona A A: Surface specification of embryonic cells: leclin receptors, cell recognition and specific cell ligands. In The Cell Surface in Development, edited by AA Moscona. New York, John Wiley and Sons, 1974. 13. Frank JS, Langer GA: The myocardial interstitium; its structure and its role in ionic exchange. J Cell Biol 60: 586 601, 1974. 14. McClain PE: Characterization of cardiac muscle collagen. J Biol Chem 249: 2303 2311, 1974. 15. Patten BM, Kramer TC, Barry A: Valvular action of the embryonic chick heart by localized apposition of endocardial masses. Anal Rec 102: 299-311, 1948. 16. Hay DA. Low FN: The fusion of dorsal and ventral endocardial cushions in the embryonic chick heart; a study in fine structure. Am J Anat 133: 1-23, 1972. 17. Markwald RR, Fitzharris TP, Adams Smith WN: Structural analysis of endocardial cylodifferentiation. Dev Biol 42: 160 180, 1975. 18. Morris SC, McClain PE; Heterogeneity in the cyanogen bromide peptides from striated muscle and heart valve collagen. Biochem Biophys Res Commun 47: 27 34, 1972. 19. Seemayer TA, Thelmo WL. Morin J: Cartilaginous transformation of the aortic valve. Am J Clin Palhol 60: 616 620, 1973. 20. Hibbs RG, Ellison JP: The atrioventricular valves of the guinea pig. II. An ultrastructural study. Am J Anat 138: 347-369, 1973. 21. Deiss WP, Leon AS: Isolation of a sulfate ester of hyaluronic acid from heart valves. J Biol Chem 206: 375-380. 1954. 22. Deiss WP, Leon AS: Mucopolysaccharides of heart valve mucoprotein. J Biol Chem 215: 685-689. 1955. 23. Mancini RE, Nunez C, Lusting ES: A radioautographic study on the specificity and sequence of the uptake of S " by the mucopolysaccharides of embryonic and adult connective tissues. J Histochem Cytochem 4: 444-445. 1956. 24. Hallgrimsson J, Friberg U, Burke JF: Changes with age in the metabolism of sulfated glycosaminoglycans in guinea pig cardiac valves. Exp Mol Pathol 12: 70 83. 1970. 25. Moretti A, Whitehouse MW: Changes in mucopolysaccharide composition of bovine heart valves with age. Biochem J 87: 396 402, 1963. 26. Lowther DA, Toole BP, Meyer FA: Extraction of acid mucopolysaccharides from heart valves. Arch Biochem Biophys 118: I - I I , 1967. VOL. 38, No. 5, MAY 1976 27. Toole BP, Lowther DA: Dermatan sulfate-protein; isolation from and interaction with collagen. Arch Biochem Biophys 128: 567-578, 1968. 28. Howse HD, Ferrans VJ, Hibbs RG: A comparative histochemical and electron microscopic study of the surface coatings of cardiac muscle cells. J Mol Cell Cardiol 1: 157-168, 1970. 29. Langer GA: Heart; excitation-contraction coupling. Annu Rev Physiol 35: 55-86, 1973. 30. Mori Y, Kanke Y: Biochemical study of cardiac valvular tissue. In Biochemistry and Pathology of Connective Tissue, edited by Y Otaka. Tokyo, V. Shoin, 1974. 31. Okamoto N, Satow Y, Ikeda T: An electron microscope observation on the heart of a 9 mm (C-R) human embryo. Hiroshima J Med Sci 17: 153-167, 1968. 3?. Johnson RC, Manasek FJ, Vinson W, Seyer J: The biochemical and ultrastructural demonstration of collagen during early heart development. Dev Biol 36: 252-271, 1974. 33. Ortiz EC: Estudio histoquimico de la gelatina cardiaca en el embrion de polio. Arch Inst Cardiol Mex 28: 244-262, 1958. 34. Johnston PM, Comar CL: Autoradiographic studies of the utilization of S"-sulfate by the chick embryo. J Biophys Biochem Cytol 3: 231-238, 1957. 35. Manasek FJ: Sulfated extracellular matrix production in the embryonic heart and adjacent tissues. J Exp Zool 174: 415-439, 1970. 36. Manasek FJ: Some comparative aspects of cardiac and skeletal "myogenesis. In Developmental Regulation, edited by S Coward. New York, Academic Press, 1973, pp 193-218. 37. Kosher RA, Searls R: Sulfated mucopolysaccharide synthesis during the development of Rana pipiens. Dev Biol 32: 50-68, 1973. 38. Thureson-Klein A, Klein RL: Cation distribution and cardiac jelly in early embryonic heart; a histochemical and electron microscopic study. J Mol Cell Cardiol 2: 31-40, 1971. 39. Satow Y, Okamoto N, Ueno T et al.: Fine structural localization of the pyroantimonate precipitable cation in the cardiac jelly of the rat embryonic tubular heart after neutron irradiation. Congenital Anomalies 13: 221-233, 1973. 40. Gessner IH, Bostrom H: In vitro studies on "S-sulfale incorporation into the acid mucopolysaccharides of chick embryo cardiac jelly. J Exp Zool 160: 283-289, 1965. 41. Gessner IH, Lorincz AE, Bostrom H: Acid mucopolysaccharide content of the cardiac jelly of the chick embryo. J Exp Zool 160: 291 298. 1965. 42. Markwald RR, Adams Smith WN: Distribution of mucosubstances in the developing rat heart. J Histochem Cytochem 20: 896-907, 1972. 43. Manasek FJ, Reid M, Vinson W et al.: Glycosaminoglycan synthesis by the early embryonic chick heart. Dev Biol 35: 332 348, 1973. 44. Holtzer H, Matheson DW: Induction of chondrogenesis in the embryo. In Chemistry and Molecular Biology of the Intercellular Matrix, vol 3, edited by EA Balazs. New York, Academic Press, 1970, pp 1753 1770. 45. Layton LL: Quantitative differential fixation of sulfate by tissues maintained in vitro. Cancer 3: 725 734, 1950. 46. Herrman H, Barry SR: Accumulation of collagen in skeletal muscle, heart and liver of chick embryo. Arch Biochem Biophys 55: 526 533. 1955. 47. Woessner JE, Bashey Rl, Boucek RJ: Collagen development in heart and skin of the chick embryo. Biochim Biophys Acla 140: 329 338. 1967. 48. Manasek FJ: Embryonic development of the heart. I. A light and electron microscopic study of myocardial development in the early chick embryo. J Morphol 125: 329 366. 1968. 49. Manasek FJ: Glycoprotein synthesis and tissue interactions during establishment of the functional embryonic chick heart. J Mol Cell Cardiol (in press). 50. Martinez-Palomo A: The surface coats of animal cells. Int Rev Cylol 29: 29-75, 1970. 51. Martinez-Palomo A. Benitez D. Alanis J: Selective deposition of lanthanum in mammalian cardiac cell membranes: ultrastructural and electrophysiological evidence. J Cell Biol 58: I 10. 1973. 52. Manasek FJ: The extracellular matrix of the early embryonic heart. In Developmental and Physiological Correlates of Cardiac Muscle, edited by M Lieberman, T Sano. New York, Raven Press, 1975, pp I 20. 53. Manasek FJ: Histogenesis of the embryonic myocardium. Am J Cardiol 25: 149 168. 1970. 54. Manasek FJ: Embryonic development of the heart. II. Formation of the epicardium. J Embryol Exp Morphol 22: 333 348. 1969. 55. Oliphant LW. Cloney RA: The ascidian myocardium: sarcoplasmic reticulum and excitation-contraction coupling. Z Zellforsch Mikrosk Anat 129: 395 412. 1972. 56. Gros D, Challice CE, Schrevel J: Detection de glycoproteines dans l'appareil de Golgi des cellules myocardiques et endocardiques du coeur embryonnaire de souris. J Microsc (Paris) 20: 303 306. 1975. 57. Ueno T: The association of sulpho-mucopolysaccharides with hislogenesis of endocardial swellings (cushion tissues) in normal and abnormal developing rat heart (in Japanese). Hiroshima Med J 21:619-644. 1973. CARDIAC EXTRACELLULAR COMPARTMENTjManasek 58. Lannigan RA, Zaki SA: Ultrastructure of normal atrial endocardium. Br Heart J 28: 785-795. 1966. 59. Bostrom H, Moretli A. Whitehouse M: Studies on Ihe biochemistry of heart valves. Biochim Biophys Acta 74: 213-221, 1963. 60. Gessner IH: Some biochemical and anatomic effects of sodium salicylale on the chick embryonic heart. In Pathophysiology of Congenital Heart Disease, edited by FH Adams. HJC Swan. VE Hall. Berkeley. University of California Press, 1970, pp 17 26. 61. Overman DA, Beaudoin AR: Early biochemical changes in the embryonic rat heart after leratogen treatment. Teratology 4: 183-190, 1971. 337 62. Johnson RC, Manasek FJ, Vinson WC, Seyer J: Collagen in early cardiogenesis. Anat Rec 175: 350a, 1973. 63. Gordon HP, Brice MA: Intrinsic factors influencing the maintenance of contractile embryonic heart cells in vitro. II. Biochemical analysis. Exp Cell Res 85: 311-318, 1974. 64. Barry A: The functional significance of the cardiac jelly in the tubular heart of the chick embryo. Anat Rec 102: 289-298. 1948. 65. Maroko PR, Libby P, Bloor CM et al.: Reduction by hyaluronidase of myocardial necrosis following coronary artery occlusion. Circulation 46: 430-437, 1972. Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 Macromolecules of the extracellular compartment of embryonic and mature hearts. F J Manasek Downloaded from http://circres.ahajournals.org/ by guest on June 15, 2017 Circ Res. 1976;38:331-337 doi: 10.1161/01.RES.38.5.331 Circulation Research is published by the American Heart Association, 7272 Greenville Avenue, Dallas, TX 75231 Copyright © 1976 American Heart Association, Inc. All rights reserved. Print ISSN: 0009-7330. Online ISSN: 1524-4571 The online version of this article, along with updated information and services, is located on the World Wide Web at: http://circres.ahajournals.org/content/38/5/331.citation Permissions: Requests for permissions to reproduce figures, tables, or portions of articles originally published in Circulation Research can be obtained via RightsLink, a service of the Copyright Clearance Center, not the Editorial Office. Once the online version of the published article for which permission is being requested is located, click Request Permissions in the middle column of the Web page under Services. Further information about this process is available in the Permissions and Rights Question and Answer document. Reprints: Information about reprints can be found online at: http://www.lww.com/reprints Subscriptions: Information about subscribing to Circulation Research is online at: http://circres.ahajournals.org//subscriptions/