* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Proteins Hwk KEY
Biosynthesis wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Gene expression wikipedia , lookup
Paracrine signalling wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Signal transduction wikipedia , lookup
Expression vector wikipedia , lookup
Magnesium transporter wikipedia , lookup
Point mutation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Homology modeling wikipedia , lookup
Protein purification wikipedia , lookup
Interactome wikipedia , lookup
Western blot wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Metalloprotein wikipedia , lookup
Protein–protein interaction wikipedia , lookup
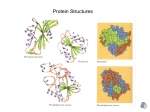
IB Bio Year 1 / IHS PROTEINS Homework, Neil 9e KEY Directions: after reading 5.4, answer the following questions in your journal. Please rephrase the questions in the answers. 1. Draw a “generalized” amino acid (this means using “R” to stand for its side chain). (Image credit: education-portal.com) Remember, the amino and carboxyl groups do ionize under certain conditions. 2. Explain how the 20 different amino acids found in living things are separated into 3 groups. They are separated by the fundamental “personalities” (chemical properties) of their side chains or R groups: 9 have nonpolar side chains (making them hydrophobic) 6 have polar side chains (making them hydrophilic) 5 have side chains that ionize (acquire electrical charges), making them really hydrophilic). Of these 5, 2 of them are acidic (acquire negative charges when they lose H), and 3 are basic (acquire positive charges when they gain H). 3. State the functions of these 8 proteins: cellulase, collagen, hemoglobin, immunoglobulin, insulin, rhodopsin, rubisco, and silk. (Use the index!) Proteins cellulase collagen hemoglobin immunoglobulin insulin rhodopsin rubisco silk 4. Functions Secreted by bacteria and fungi to hydrolyze cellulose molecules so the component glucose molecules can be absorbed for energy Structural component of connective tissue and bone Reversibly binds oxygen so it can be delivered to the tissues from the lungs; found inside red blood cells Also known as antibodies, these proteins bind to and inactivate antigens (molecules on germs) Hormone secreted by the pancreas that promotes the uptake of glucose and the synthesis of glycogen A “hybrid” molecule consisting of a protein (opsin) and a second molecule (retinal) within the cells of your retina; absorbs light and sets into motion the biochemical processes that result in vision. Short for Ribulose Bisphosphate Carboxylase, this enzyme participates in one of the sugarmaking reactions of photosynthesis Spiders use this stretchy, strong protein to build their webs (among other things) For each level of protein structure, list the bonds that stabilize the protein. (Hint: use the Exploring figure 5.20) Level of structure Primary structure Secondary structure Tertiary structure Bonds that stabilize that level Covalent peptide bonds Hydrogen bonds between the O and H atoms of the polypeptide’s backbone R group interactions: hydrogen bonds, ionic bonds (+/-) and charge repulsions (+/+ and -/-), disulfide bridges, hydrophobic interactions/van der Waals Quaternary structure 5. Same as for tertiary structure Why does a denatured protein lose its function? Every protein has a “job” (function) because of its shape. For instance, immunoglobulins bind (grab onto) parts of germs, hemoglobin binds oxygen, enzymes grab substrates, and so on. If a protein is exposed to high heat or abnormal pH or salt conditions that interfere with the bonds holding it in secondary, tertiary, or quaternary structure, its shape is lost – and its function as well.