Document
... Plasma is a mixture of positively charged ions and free electrons. When a gas is heated to a very high temperature, most of the electrons are separated from their atoms, and the gas becomes a plasma. Plasma is considered to be a fourth state of matter because its properties are different from those ...
... Plasma is a mixture of positively charged ions and free electrons. When a gas is heated to a very high temperature, most of the electrons are separated from their atoms, and the gas becomes a plasma. Plasma is considered to be a fourth state of matter because its properties are different from those ...
home work #4
... 1. __________ solids, liquids, and gases 2. __________ the amount of space an object takes up 3. __________ anything that takes up space and has mass 4. __________ something that can be observed about an object, such as the color, size, shape, or sound 5. __________ the measure of how much material ...
... 1. __________ solids, liquids, and gases 2. __________ the amount of space an object takes up 3. __________ anything that takes up space and has mass 4. __________ something that can be observed about an object, such as the color, size, shape, or sound 5. __________ the measure of how much material ...
Matter
... with a lot of solute is a concentrated solution. A solution with very little solute is a dilute solution. A solution that has as much solute as it can hold is called a saturated solution. Solutes can be solids, liquids, or gases. A suspension is a kind of mixture that separates if it is left alone f ...
... with a lot of solute is a concentrated solution. A solution with very little solute is a dilute solution. A solution that has as much solute as it can hold is called a saturated solution. Solutes can be solids, liquids, or gases. A suspension is a kind of mixture that separates if it is left alone f ...
Chapter 4 – Matter - Chemistry at Winthrop University
... Formed between metals and non-metals, and also with polyatomic ions (electrically charged molecules) ...
... Formed between metals and non-metals, and also with polyatomic ions (electrically charged molecules) ...
Matter Exam Study Guide
... on a separate piece of paper. 1. What is the definition of matter? 2. What is the definition of mass? 3. What is the definition of volume? 4. What is the definition of density? 5. What are the four states of matter discussed in this class? 6. Define a solid. 7. Define a Liquid. 8. Define a gas. 9. D ...
... on a separate piece of paper. 1. What is the definition of matter? 2. What is the definition of mass? 3. What is the definition of volume? 4. What is the definition of density? 5. What are the four states of matter discussed in this class? 6. Define a solid. 7. Define a Liquid. 8. Define a gas. 9. D ...
Export To Word
... SOURCE AND ACCESS INFORMATION Contributed by: Courtney Benedix Name of Author/Source: Lin Jarrette District/Organization of Contributor(s): Leon Is this Resource freely Available? Yes License: Attribution-NonCommercial 3.0 Unported ...
... SOURCE AND ACCESS INFORMATION Contributed by: Courtney Benedix Name of Author/Source: Lin Jarrette District/Organization of Contributor(s): Leon Is this Resource freely Available? Yes License: Attribution-NonCommercial 3.0 Unported ...
Chapter 1_chemh
... Change of state: physical change from one state to another. ●Solid: definite volume definite shape ●Liquid: definite volume indefinite shape ...
... Change of state: physical change from one state to another. ●Solid: definite volume definite shape ●Liquid: definite volume indefinite shape ...
Physical and Chemical Properties
... the liquid form. • Basically the range at which the solid changes its state •The melting point of into a liquid. water is 0 degrees ...
... the liquid form. • Basically the range at which the solid changes its state •The melting point of into a liquid. water is 0 degrees ...
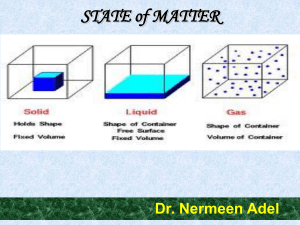
STATE of MATTER
... • It is state of matter that have properties between those of a conventional liquid and those of a solid crystal. • Liquid crystal may flow like a liquid, but its molecules may be oriented in a crystal-like way like a solid. • Liquid crystals can be divided into Thermotropic Lyotropic and Metallotro ...
... • It is state of matter that have properties between those of a conventional liquid and those of a solid crystal. • Liquid crystal may flow like a liquid, but its molecules may be oriented in a crystal-like way like a solid. • Liquid crystals can be divided into Thermotropic Lyotropic and Metallotro ...
This famous round building was made for sports
... measure mass (heavier objects); equal to 1,000 ...
... measure mass (heavier objects); equal to 1,000 ...
Science 9
... The cup should contain only salt. The water evaporated. Since a new substance was not formed, a physical change has taken place. ...
... The cup should contain only salt. The water evaporated. Since a new substance was not formed, a physical change has taken place. ...
Notes. - Net Start Class
... Gas particles are independent of each other and move in a straight line. Change in direction occurs only with collisions. Gases assume the shape and volume of their container. ...
... Gas particles are independent of each other and move in a straight line. Change in direction occurs only with collisions. Gases assume the shape and volume of their container. ...
Chapter 3 PowerPoint Notes
... matter in which the material has no definite shape and no definite volume (Takes the shape and volume of its container). • Molecules/Atoms are spread apart and can be compressed. ...
... matter in which the material has no definite shape and no definite volume (Takes the shape and volume of its container). • Molecules/Atoms are spread apart and can be compressed. ...
Unit 01 Qual Chem
... Physical Change = a change that does not alter the identity of a substance (shape, size, state) Chemical Change = a change in which one or more substances are converted into substances with different chemical properties ...
... Physical Change = a change that does not alter the identity of a substance (shape, size, state) Chemical Change = a change in which one or more substances are converted into substances with different chemical properties ...
State of matter

In physics, a state of matter is one of the distinct forms that matter takes on. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. Many other states are known, such as Bose–Einstein condensates and neutron-degenerate matter, but these only occur in extreme situations such as ultra cold or ultra dense matter. Other states, such as quark–gluon plasmas, are believed to be possible but remain theoretical for now. For a complete list of all exotic states of matter, see the list of states of matter.Historically, the distinction is made based on qualitative differences in properties. Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place. Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely. Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place. Matter in the plasma state has variable volume and shape, but as well as neutral atoms, it contains a significant number of ions and electrons, both of which can move around freely. Plasma is the most common form of visible matter in the universe.The term phase is sometimes used as a synonym for state of matter, but a system can contain several immiscible phases of the same state of matter (see Phase (matter) for more discussion of the difference between the two terms).