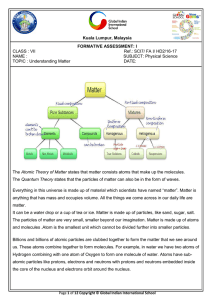
2.1-Properties of Matter
... A physical property is a quality or condition of a substance that can be observed or measured without changing the substance’s composition. In other words, observing the property doesn’t change the identity of the substance. Water is still Water. Paper is still Paper, etc. ...
... A physical property is a quality or condition of a substance that can be observed or measured without changing the substance’s composition. In other words, observing the property doesn’t change the identity of the substance. Water is still Water. Paper is still Paper, etc. ...
ChemThink Particulate Nature of Matter
... 1. What is the particulate nature of matter? ______________________________________________________________________ ______________________________________________________________________ 2. A substance composed of only one type of atom is called a(n) ____________________ 3. How are the atoms behavin ...
... 1. What is the particulate nature of matter? ______________________________________________________________________ ______________________________________________________________________ 2. A substance composed of only one type of atom is called a(n) ____________________ 3. How are the atoms behavin ...
Chemistry Notes
... Properties - properties you can test for or sense without changing the substance Examples: density color taste pH magnetism boiling point melting point odor ...
... Properties - properties you can test for or sense without changing the substance Examples: density color taste pH magnetism boiling point melting point odor ...
Review Sheet Filled Out
... Electrons closest to the nucleus have the least amount of energy Electrons farthest away from the nucleus have the most energy – valence e Have a negative charge Have insignificant mass and volume Reside in the 99.996% of the atom outside the nucleus Can’t tell where an electron is at any ...
... Electrons closest to the nucleus have the least amount of energy Electrons farthest away from the nucleus have the most energy – valence e Have a negative charge Have insignificant mass and volume Reside in the 99.996% of the atom outside the nucleus Can’t tell where an electron is at any ...
Physical Properties
... The Representation of Matter In chemistry we use chemical formulas and symbols to represent matter. ...
... The Representation of Matter In chemistry we use chemical formulas and symbols to represent matter. ...
class notes packet - Social Circle City Schools
... Chromatography is a method for analyzing complex _______________ such as _____ by separating them into the chemicals fro which they are made. Attraction to a medium will take light particles up the medium as heavier particles stay low on the medium When can this be used? ...
... Chromatography is a method for analyzing complex _______________ such as _____ by separating them into the chemicals fro which they are made. Attraction to a medium will take light particles up the medium as heavier particles stay low on the medium When can this be used? ...
Unit 2: Mixture and Matter Study Guide Ch 2 Vocab to know: Matter
... What’s The Matter Video PowerPoint Notes Mixture and Matter Concept Map and Worksheet ...
... What’s The Matter Video PowerPoint Notes Mixture and Matter Concept Map and Worksheet ...
... states: the ground state with E0=0 and an exited state with E1=ε=4x10-20J. a. In the beginning ¼ of the atoms were in the exited state. What is the temperature of the crystal? b. The crystal is placed in thermal contact with a reservoir at a temperature of 300K. How much energy will flow between the ...
Grade 9 Academic Science – Chemistry
... Water has very unusual characteristic physical properties (e.g., water has a high Heat Capacity (...it takes a lot of energy to raise the temperature of 1gm of water 1°C. In other words, water changes temperature more slowly than other compounds); many other compounds dissolve in water (...water is ...
... Water has very unusual characteristic physical properties (e.g., water has a high Heat Capacity (...it takes a lot of energy to raise the temperature of 1gm of water 1°C. In other words, water changes temperature more slowly than other compounds); many other compounds dissolve in water (...water is ...
Chromatography
... Fractional distillation separates by using the boiling points at which several fractions of the compound will ...
... Fractional distillation separates by using the boiling points at which several fractions of the compound will ...
PowerPoint
... • Called suspensions and colloids • Not uniform throughout – Ex. Chocolate chip cookie, gravel, soil ...
... • Called suspensions and colloids • Not uniform throughout – Ex. Chocolate chip cookie, gravel, soil ...
Matter 1. ______ is anything that has ______ and takes up ______
... b. an __________ is the smallest unit of an element that maintains the chemical identity of that element. An ____________ is a substance that cannot be broken down into simpler substances in a chemical reaction. An element contains only ________ __________kind of atom. A _____________ is a substance ...
... b. an __________ is the smallest unit of an element that maintains the chemical identity of that element. An ____________ is a substance that cannot be broken down into simpler substances in a chemical reaction. An element contains only ________ __________kind of atom. A _____________ is a substance ...
View - Workshops+SJCOE Workshop Management
... other objects). For example, a model showing that gases are made from matter particles that are too small to see and are moving freely around in space can explain many observations, including the inflation and shape of a balloon; the effects of air on larger particles or objects (e.g., leaves in win ...
... other objects). For example, a model showing that gases are made from matter particles that are too small to see and are moving freely around in space can explain many observations, including the inflation and shape of a balloon; the effects of air on larger particles or objects (e.g., leaves in win ...
Atomic arrangement, short and long range order, point. Direction
... longrange order. In an ideal gas the arrangementof an atom at any point in space is independent of the arrangement of other atoms. Thus, bothlong-range and shortrange order are absent in the ideal gas, but liquids and amorphous solidsexhibit sho rt-rangeordera certain regularity in the arrangement o ...
... longrange order. In an ideal gas the arrangementof an atom at any point in space is independent of the arrangement of other atoms. Thus, bothlong-range and shortrange order are absent in the ideal gas, but liquids and amorphous solidsexhibit sho rt-rangeordera certain regularity in the arrangement o ...
Chemistry 101 2007
... 1.1 The Atomic and molecular Perspectives of Chemistry. Chemistry is the study of the properties and behavior of matter. A property is a characteristic that allows us to recognize a particular type of matter. All the matter in the world is comprised of about 116 elements. ...
... 1.1 The Atomic and molecular Perspectives of Chemistry. Chemistry is the study of the properties and behavior of matter. A property is a characteristic that allows us to recognize a particular type of matter. All the matter in the world is comprised of about 116 elements. ...
worksheer format 11-12
... Plasma is a state of matter in which all matter is ionized and it occurs in the form of ions and electrons. Since, we know that for the ionization of the element or matter to occur energy is required to pull the electron from the attraction of the nuclear charge, high energy is required. So for the ...
... Plasma is a state of matter in which all matter is ionized and it occurs in the form of ions and electrons. Since, we know that for the ionization of the element or matter to occur energy is required to pull the electron from the attraction of the nuclear charge, high energy is required. So for the ...
State of matter

In physics, a state of matter is one of the distinct forms that matter takes on. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. Many other states are known, such as Bose–Einstein condensates and neutron-degenerate matter, but these only occur in extreme situations such as ultra cold or ultra dense matter. Other states, such as quark–gluon plasmas, are believed to be possible but remain theoretical for now. For a complete list of all exotic states of matter, see the list of states of matter.Historically, the distinction is made based on qualitative differences in properties. Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place. Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely. Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place. Matter in the plasma state has variable volume and shape, but as well as neutral atoms, it contains a significant number of ions and electrons, both of which can move around freely. Plasma is the most common form of visible matter in the universe.The term phase is sometimes used as a synonym for state of matter, but a system can contain several immiscible phases of the same state of matter (see Phase (matter) for more discussion of the difference between the two terms).