* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Physical Properties
Chemical industry wikipedia , lookup
Fluorochemical industry wikipedia , lookup
Al-Shifa pharmaceutical factory wikipedia , lookup
Registration, Evaluation, Authorisation and Restriction of Chemicals wikipedia , lookup
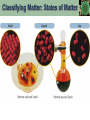
Chemical weapon proliferation wikipedia , lookup
Host–guest chemistry wikipedia , lookup
Chemical plant wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Chemical bond wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Chemical weapon wikipedia , lookup
Chemical Corps wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Transition state theory wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Biochemistry wikipedia , lookup
Safety data sheet wikipedia , lookup
Self-assembled monolayer wikipedia , lookup
Chemical potential wikipedia , lookup
Abiogenesis wikipedia , lookup
Organic chemistry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Condensed matter physics wikipedia , lookup
Drug discovery wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Computational chemistry wikipedia , lookup
Physical organic chemistry wikipedia , lookup
History of molecular theory wikipedia , lookup
History of chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Jeffrey Mack California State University, Sacramento Chemistry and Its Methods • Hypothesis: A tentative explanation or prediction based on experimental observations. • Law: A concise verbal or mathematical statement of a behavior or a relation that seems always to be the same under the same conditions. • Theory: a well-tested, unifying principle that explains a body of facts and the laws based on them. It is capable of suggesting new hypotheses that can be tested experimentally. 2 Chemistry and Its Methods 3 • Experimental results should be reproducible. • Furthermore, these results should be reported in the scientific literature in sufficient detail that they can be used or reproduced by others. • Conclusions should be reasonable and unbiased. • Credit should be given where it is due. Qualitative Observations 4 • No numbers involved • Color, appearance, statements like “large” or “small: • Stating that something is hot or cold without specifying a temperature. • Identifying something by smell • No measurements Qualitative Observations • A quantity or attribute that is measureable is specified. • Numbers with units are expressed from measurements. • Dimensions are given such as mass, time, distance, volume, density, temperature, color specified as a wavelength etc... 5 Classifying Matter: States of Matter 6 Classifying Matter: States of Matter 7 • In solids these particles are packed closely together, usually in a regular array. The particles vibrate back and forth about their average positions, but seldom does a particle in a solid squeeze past its immediate neighbors to come into contact with a new set of particles. • The atoms or molecules of liquids are arranged randomly rather than in the regular patterns found in solids. Liquids and gases are fluid because the particles are not confined to specific locations and can move past one another. • Under normal conditions, the particles in a gas are far apart. Gas molecules move extremely rapidly and are not constrained by their neighbors. The molecules of a gas fly about, colliding with one another and with the container walls. This random motion allows gas molecules to fill their container, so the volume of the gas sample is the volume of the container. States of Matter • SOLIDS — have rigid shape, fixed volume. External shape may reflect the atomic and molecular arrangement. –Reasonably well understood. • LIQUIDS — have no fixed shape and may not fill a container completely. –Structure not well understood. • GASES — expand to fill their container completely. –Well defined theoretical understanding. 8 Classifying Matter 9 Classifying Matter 10 Mixtures: Homogeneous and Heterogeneous • A homogeneous mixture consists of two or more substances in the same phase. No amount of optical magnification will reveal a homogeneous mixture to have different properties in different regions. • A heterogeneous mixture does not have uniform composition. Its components are easily visually distinguishable. • When separated, the components of both types of mixtures yields pure substances. Classifying Matter 11 Classifying Matter Pure Substances • A pure substance has well defined physical and chemical properties. • Pure substances can be classified as elements or compounds. • Compounds can be further reduced into two or more elements. • Elements consist of only one type of atom. They cannot be decomposed or further simplified by ordinary means. 12 Matter and its Representation What we observe… To what we can’t see! Chemical symbols allow us to connect… 13 The Representation of Matter In chemistry we use chemical formulas and symbols to represent matter. Why? We are “macroscopic”: large in size on the order of 100’s of cm. Atoms and molecules are “microscopic”: on the order of 10-12 cm 14 Elements • The elements are recorded on the PERIODIC TABLE • There are 117 recorded elements at this time. • The Periodic table will be discussed further in chapter 2. 15 Chemical Compounds Chemical compounds are composed of two or more atoms. 16 Chemical Compounds Molecule: Ammonia (NH3) Ionic Compound Iron pyrite (FeS2) 17 Chemical Compounds 18 • All Compounds are made up of molecules or ions. • A molecule is the is the smallest unit of a compound that retains its chemical characteristics. • Ionic compounds are described by a “formula unit”. • Molecules are described by a “molecular formula”. Molecular Formula • A molecule is the smallest unit of a compound that retains the chemical characteristics of the compound. • Composition of molecules is given by a molecular formula. H2O C8H10N4O2 - caffeine 19 Physical Properties Some physical properties: − Color − State (s, g or liq) − Melting and Boiling point − Density (mass/unit volume) Extensive properties (mass) depend upon the amount of substance. Intensive properties (density) do not. 20 21 Physical Properties Physical properties are a function of intermolecular forces. H Water (18 g/mol) liquid at 25oC Methane (16 g/mol) gas at 25oC H C H H O H H • Water molecules are attracted to one another by “hydrogen bonds”. • Methane molecules only exhibit week “London Forces”. Physical Properties Physical properties are affected by temperature (molecular motion). The density of water is seen to change with temperature. 22 Physical Properties Mixtures may be separated by physical properties: Physical Property Means of Separation Density Decantation, centrifugation Boiling point Distillation State of Matter Filtration Intermolecular Forces Chromatography Vapor pressure Evaporation Magnetism Magnets Solubility Filtration 23 Chemical Properties • Chemical properties are really chemical changes. • The chemical properties of elements and compounds are related to periodic trends and molecular structure. 24 Chemical Properties A chemical property indicates whether and sometimes how readily a material undergoes a chemical change with another material. For example, a chemical property of hydrogen gas is that it reacts vigorously with oxygen gas. 25 26 The Nature of Matter Gold Mercury Chemists are interested in the nature of matter and how this is related to its atoms and molecules. A Chemist’s View of Water Macroscopic H 2O (gas, liquid, solid) Particulate Symbolic 2 H2(g) + O2 (g) 2 H2O(g) Energy: Some Basic Principles Energy can be classified as Kinetic or Potential. • Kinetic energy is energy associated with motion such as: • The motion at the particulate level (thermal energy). • The motion of macroscopic objects like a thrown baseball, falling water. • The movement of electrons in a conductor (electrical energy). • Wave motion, transverse (water) and compression (acoustic). Matter consists of atoms and molecules in motion. 28 Energy: Some Basic Principles Potential energy results from an object’s position: • Gravitational: An object held at a height, waterfalls. • Energy stored in an extended spring. • Energy stored in molecules (chemical energy, food) • The energy associated with charged or partially charged particles (electrostatic energy) • Nuclear energy (fission, fusion). 29