Solute
... Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, io ...
... Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, io ...
Matter
... • smallest particle of element that retains properties of element • smallest particles of element that can undergo a chemical reaction ...
... • smallest particle of element that retains properties of element • smallest particles of element that can undergo a chemical reaction ...
matter (physical science)
... For example: Baking cookies will cause chemical changes, sugar will caramelize and the baking powder will cause gas bubbles to be formed rusting steel wool, oxygen reacts with iron Tarnishing pennies, oxygen reacts with copper Burning a candle, paraffin wax reacts with oxygen to produce carbon, carb ...
... For example: Baking cookies will cause chemical changes, sugar will caramelize and the baking powder will cause gas bubbles to be formed rusting steel wool, oxygen reacts with iron Tarnishing pennies, oxygen reacts with copper Burning a candle, paraffin wax reacts with oxygen to produce carbon, carb ...
States of Matter WebQuest
... 27. Use the chart to identify the state of matter described by the following. Many of these have more than one answer! Write solid, liquid, or gas in the spaces below. not easily compressible rigid – particles are locked into place flows easily ...
... 27. Use the chart to identify the state of matter described by the following. Many of these have more than one answer! Write solid, liquid, or gas in the spaces below. not easily compressible rigid – particles are locked into place flows easily ...
Chemical Changes in Matter Worksheet
... For each of the following reactions, replace words with formulas and symbols. Be sure to include coefficients and physical states. 1. Two atoms of solid lithium react with two molecules of liquid water to produce two units of aqueous lithium hydroxide and one molecule of hydrogen gas. ...
... For each of the following reactions, replace words with formulas and symbols. Be sure to include coefficients and physical states. 1. Two atoms of solid lithium react with two molecules of liquid water to produce two units of aqueous lithium hydroxide and one molecule of hydrogen gas. ...
Chapter 18 Resource: Matter
... 5. The (chemical, physical) properties of an element determine how the element will change when it reacts with another element. 6. An example of matter is (air, heat). 7. A difference in the (mass, atomic) number of atoms means the atoms are of ...
... 5. The (chemical, physical) properties of an element determine how the element will change when it reacts with another element. 6. An example of matter is (air, heat). 7. A difference in the (mass, atomic) number of atoms means the atoms are of ...
PROPERTIES_OF_MATTER
... • Elements in compounds are joined by chemical bonds. – Examples of chemical bonds: • Ionic bond (electrostatic attraction that binds oppositely charged ions) – Usually composed of metal and nonmetal atoms ...
... • Elements in compounds are joined by chemical bonds. – Examples of chemical bonds: • Ionic bond (electrostatic attraction that binds oppositely charged ions) – Usually composed of metal and nonmetal atoms ...
Chemical Principles – by Steven Zumdahl (5 ) Chapter 1
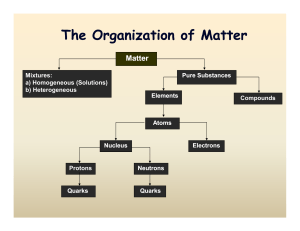
... Substance is a material that cannot be separated by physical means into two or more materials. (with different properties) Elements are substances that cannot be decomposed by chemical reactions. Elements (atoms) are the simplest forms of matter (in chemistry). Each element is represented by a symbo ...
... Substance is a material that cannot be separated by physical means into two or more materials. (with different properties) Elements are substances that cannot be decomposed by chemical reactions. Elements (atoms) are the simplest forms of matter (in chemistry). Each element is represented by a symbo ...
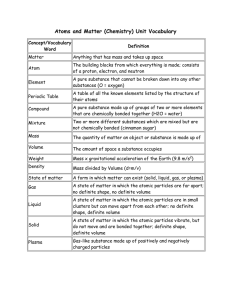
Yr9 Chem Terms def
... Particles too small to see which make up all matter Term used to include anything which has mass and occupies space Pure substances made of only 1 type of atom 3 Elements A tiny particle containing 2 or more atoms in a fixed ratio and 4 Molecules joined by chemical bonds 5 Periodic Table A table of ...
... Particles too small to see which make up all matter Term used to include anything which has mass and occupies space Pure substances made of only 1 type of atom 3 Elements A tiny particle containing 2 or more atoms in a fixed ratio and 4 Molecules joined by chemical bonds 5 Periodic Table A table of ...
FREEZING – is the change of a liquid to a solid. Freezing occurs
... Phase of matter is an important physical property of matter. Matter can exists in four phases: 1) Solid ...
... Phase of matter is an important physical property of matter. Matter can exists in four phases: 1) Solid ...
states of matter - Haiku for Ignatius
... A plasma is a very good conductor of electricity and is affected by magnetic fields. Plasmas, like gases • Plasma is the have an indefinite common state shape and an of matter indefinite volume. ...
... A plasma is a very good conductor of electricity and is affected by magnetic fields. Plasmas, like gases • Plasma is the have an indefinite common state shape and an of matter indefinite volume. ...
PSR 181-186 asnwers
... 8. Neutrons have a ___neutral______ charge and they are found in the _nucleus__. 9. In order to react, molecules must __collide______ with sufficient force. 10. When the temperature goes up, molecules move ___faster______. This makes a chemical reaction happen ___faster______. 11. Complete the chart ...
... 8. Neutrons have a ___neutral______ charge and they are found in the _nucleus__. 9. In order to react, molecules must __collide______ with sufficient force. 10. When the temperature goes up, molecules move ___faster______. This makes a chemical reaction happen ___faster______. 11. Complete the chart ...
People Search for Review
... 1. What is the difference between a mixture and a compound? Give an example of each. ...
... 1. What is the difference between a mixture and a compound? Give an example of each. ...
State of matter

In physics, a state of matter is one of the distinct forms that matter takes on. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. Many other states are known, such as Bose–Einstein condensates and neutron-degenerate matter, but these only occur in extreme situations such as ultra cold or ultra dense matter. Other states, such as quark–gluon plasmas, are believed to be possible but remain theoretical for now. For a complete list of all exotic states of matter, see the list of states of matter.Historically, the distinction is made based on qualitative differences in properties. Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place. Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely. Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place. Matter in the plasma state has variable volume and shape, but as well as neutral atoms, it contains a significant number of ions and electrons, both of which can move around freely. Plasma is the most common form of visible matter in the universe.The term phase is sometimes used as a synonym for state of matter, but a system can contain several immiscible phases of the same state of matter (see Phase (matter) for more discussion of the difference between the two terms).