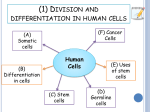
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Cancer Prone Disease Section Beckwith-Wiedemann syndrome Atlas of Genetics and Cytogenetics
Deoxyribozyme wikipedia , lookup
Epigenetics in learning and memory wikipedia , lookup
Designer baby wikipedia , lookup
Birth defect wikipedia , lookup
Saethre–Chotzen syndrome wikipedia , lookup
RNA silencing wikipedia , lookup
Genome (book) wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
Cancer epigenetics wikipedia , lookup
Medical genetics wikipedia , lookup
Frameshift mutation wikipedia , lookup
Gene expression profiling wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
DiGeorge syndrome wikipedia , lookup
Epitranscriptome wikipedia , lookup
Non-coding RNA wikipedia , lookup
Gene expression programming wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Microevolution wikipedia , lookup
Oncogenomics wikipedia , lookup
X-inactivation wikipedia , lookup
Down syndrome wikipedia , lookup
Long non-coding RNA wikipedia , lookup
Primary transcript wikipedia , lookup
Epigenetics of human development wikipedia , lookup
Point mutation wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Atlas of Genetics and Cytogenetics in Oncology and Haematology OPEN ACCESS JOURNAL AT INIST-CNRS Cancer Prone Disease Section Mini Review Beckwith-Wiedemann syndrome Marcel Mannens DNA-diagnostics laboratory, University of Amsterdam, Academic Medical Center Department of Clinical Genetics PO Box 22700 1100 DE Amsterdam, the Netherlands (MM) Published in Atlas Database: November 2000 Online updated version : http://AtlasGeneticsOncology.org/Kprones/BeckwithWiedemannID10037.html DOI: 10.4267/2042/37714 This work is licensed under a Creative Commons Attribution-Noncommercial-No Derivative Works 2.0 France Licence. © 2001 Atlas of Genetics and Cytogenetics in Oncology and Haematology Identity Clinics Alias Exomphalos-macroglossia-gigantism triad Inheritance Incidence of 7/105; given the variable expression of the symptoms, the actual frequency is likely to be higher; generally there is sporadic occurrence of the syndrome (85%); inheritance is mostly maternal (imprinting) with a more severe phenotype after maternal transmission. Note Clinically and genetically heterogeneous; three distinct regions on 11p15 have been associated with BWS (BWSCR1/2/3); BWSCR2 seems to be particularly associated with hemihypertrophy. Phenotype and clinics Multiple features that occur variably; most prominent is the EMG triad (exomphalos-macroglossia-gigantism): apart from the abdominal wall defects and pre- and postnatal growth abnormalities, earlobe pits or creases, facial nevus flammeus, hypoglycemia, renal abnormalities and hemihypertrophy (unilateral overgrowth) are frequently seen. Patient with Beckwith-Wiedemann syndrome. The face shows the enlarged tongue (macroglossia), the ear the typical earlobe creases Marcel Mannens. Atlas Genet Cytogenet Oncol Haematol. 2001; 5(1) 62 Beckwith-Wiedemann syndrome Mannens M Neoplastic risk Protein Description: Untranslated. Expression: Highly expressed in endodermal and mesodermal embryonic tissues; in adult brain, only in the pons and globus pallidus; in adult tissues, expression is primarily confined to skeletal and cardiac muscle; other tissues are down-regulated postnatal but re-expressed in tumours that express the gene during embryogenesis. Function: Putative tumour suppressor; proposed regulatory function for IGF2 expression (under debate). Mutations Germinal: Hypermethylated in 10-20% of sporadic BWS cases; familial transmission unclear yet; loss of imprinting (LOI) can be induced in deletion mouse models. Somatic: Hypermethylated in 10-20% of sporadic BWS cases mostly somatic events due to UPD in mosaic form; LOI in tumours. The increased risk for childhood solid tumours is 7.5% (thousand fold increase); tumours most frequently seen are nephroblastoma (Wilms tumour), adrenocortical carcinoma, rhabdomyosarcoma and hepatoblastoma; clinical risk factors are hemihypertrophy and nephromegaly; genetic risk factors are uniparental disomy (UPD) and H19/IGF2 imprinting defects. Treatment In general surgical correction of the abdominal wall defects and macroglossia; monitoring the glycemia during the first 3 days and early treatment of hypoglycemia (deleterious for central nervous system) is of importance to avoid further complications; frequent screening for tumour development. Prognosis Clinical features tend to become less with ageing; tumour risk decreases strongly after the 4-7th year of birth. IGF2 (insulin-like growth factor 2 (somatomedin A)) Cytogenetics Alias: IGF-II, somatomedin A, Hs.75963 Location: 11p15.5 DNA/RNA Transcription: 1356 bp mRNA, paternally expressed, maternal imprint. Protein Description: 180 amino acids, 20,14 kDa (unprocessed). Expression: IGF2 has the highest levels of expression in tissues that are affected by prenatal overgrowth in BWS; the main source of expression is liver; expression depends on promoter usage; P1 is exclusively active in adult liver, whereas P3 and P4 exert their action in liver prenatal; P2 is only active in certain tumour cell lines. Localisation: Secreted. Function: Embryonal growth factor, mitogen. Homology: Belongs to the insulin/IGF/relaxin family. Mutations Germinal: Hypomethylated; LOI in sporadic BWS cases; familial transmission unclear yet; BWS phenotype can be induced in igf2 overexpressing mouse models. Somatic: Hypomethylated, LOI in sporadic BWS cases; mostly somatic events due to UPD in mosaic form; LOI in tumours. Inborn conditions Paternal duplications of chromosome region 11p15, maternal translocations involving chromosome region 11p15.3-p15.5. Cytogenetics of cancer Apart from chromosome 11 aberrations, multiple chromosomes are involved in tumour development; promising prognostic indicators in Wilms tumour might be chromosome 1p and 16q aberrations; tther molecular abnormalities associated with an adverse outcome in Wilms tumour are 22q allele loss or P53 aberrations. Other findings Note In 10-20% of BWS cases, uniparental disomy of chromosome region 11p15 is seen, mostly in a mosaic form. Genes involved and proteins H19 Alias: D11S813E, D11S878E, ASM, ASM1 Location: 11p15.5 Note Imprinted, maternally expressed, untranslated mRNA. DNA/RNA Description: The human H19 gene is 2.7 kb long and includes 4 small introns; maternally expressed, paternal imprint. Atlas Genet Cytogenet Oncol Haematol. 2001; 5(1) CDKN1C (cyclin-dependent kinase inhibitor 1C) Alias: KIP2, P57KIP2, P57, CDKN5 Location: 11p15.5 63 Beckwith-Wiedemann syndrome Mannens M ZNF215 DNA/RNA Description: 1511 bp messenger, preferentially maternally expressed (paternal imprint). Protein Description: 316 amino acids; 32,177 kDa, CDK inhibitory domain, PAPA repeat, conserved C-terminal domain. Expression: It is expressed in the heart, brain, lung, skeletal muscle, kidney, pancreas and testis; high levels are seen in the placenta, low levels in liver. Localisation: Nuclear. Function Summary: Cyclin-dependent kinase inhibitor 1C is a tight-binding inhibitor of several G1 cyclin/Cdk complexes and a negative regulator of cell proliferation; mutations of CDKN1C are implicated in sporadic cancers and Beckwith-Wiedemann syndrome suggesting that it is a tumour suppressor candidate; in BWS however, no evidence for tumour association was found. Homology: p21CIP1 CdK inhibitor gene family. Mutations Germinal: Mostly maternal, nucleotide substitutions, small deletions. Somatic: CDKN1C mutations are described in tumour formation; mouse mutation-models reveal part of the BWS phenotype in particular the abdominal-wall defects. Alias: zinc finger protein 215, BAZ2 Location: 11p15.4 DNA/RNA Description: mRNA of 3480 bp, 9 exons, at least 5 splice variants; exon 9 runs antisense of a second gene: ZNF214. Transcription: Imprinted in a tissue specific manner, the maternal allele being preferentially expressed. Protein Description: 517 amino acids, 60,048 kDa; KRABA domain; similarities to a KRABB domain; SCAN box; nuclear localisation signal KKKR; 2 x 2 zinc-fingers. Expression: Widely expressed at low levels; expression is highest in testis; splice variants of ZNF215 show tissue specific expression. Localisation: Nuclear. Function: Putative transcription factor; ZNF215 was cloned from a region associated with hemihypertrophy, cardiac abnormalities, Wilms tumour and minor BWS features; as such the gene might be responsible for a distinct phenotype in BWS. Homology: Belongs to the Krueppel family of C2H2type zinc finger proteins. Mutations Germinal: Various amino acids substitutions found in BWS / hemihypertrophy patients; causal relationship with phenotype unclear. Somatic: In tumours no mutations found so far. KCNQ1OT1 (KCNQ1 overlapping transcript) References Alias: KCNQ1 overlapping transcript 1, LIT1, KvDMR1, KvLQT1-AS, Long QT intronic transcript 1 Location: 11p15.5 DNA/RNA Description: Maternally imprinted gene, > 80 kb RNA. Transcription: Intronic transcript 1, embedded in intron 9 (and 10) of KCNQ1, in opposite orientation; expressed in most human tissues and from the paternal allele, the maternal allele being imprinted through a specific methylation of a CpG island; abnormally expressed in patients with Beckwith-Wiedemann syndrome, independently of IGF2 imprinting; no abnormal imprinting in Wilms tumour. Protein Expression: Untranslated. Function: Unknown; it is postulated that KCNQ1OT1 might influence the expression of nearby imprinted genes such as CDKN1C or IGF2/H19. Mutations Germinal: Aberrant methylation in 50-80% of BWS patients not always 100% (might be due to UPD in some cases); inheritance unclear. Somatic: Unclear; there is no association between aberrant methylation and tumour development. Atlas Genet Cytogenet Oncol Haematol. 2001; 5(1) Beckwith J. Extreme cytomegaly of the adrenal fetal cortex, omphalocele, hyperplasia of kidneys and pancreas, and Leydig-cell hyperplasia: Another syndrome? Western Society for Pediatric Research (abst) Los Angeles 1963 (Nov 11). WIEDEMANN HR. [FAMILIAL MALFORMATION COMPLEX WITH UMBILICAL HERNIA AND MACROGLOSSIA--A "NEW SYNDROME"?]. J Genet Hum. 1964 Sep;13:223-32 Beckwith J. Macroglossia, omphalocele, adrenal cytomegaly, gigantism, and hyperplastic visceromegaly. Birth Defects 1969;5:188-96. Elliott M, Bayly R, Cole T, Temple IK, Maher ER. Clinical features and natural history of Beckwith-Wiedemann syndrome: presentation of 74 new cases. Clin Genet. 1994 Aug;46(2):168-74 Hoovers JM, Kalikin LM, Johnson LA, Alders M, Redeker B, Law DJ, Bliek J, Steenman M, Benedict M, Wiegant J, Lengauer C, Taillon-Miller P, Schlessinger D, Edwards MC, Elledge SJ, Ivens A, Westerveld A, Little P, Mannens M, Feinberg AP. Multiple genetic loci within 11p15 defined by Beckwith-Wiedemann syndrome rearrangement breakpoints and subchromosomal transferable fragments. Proc Natl Acad Sci U S A. 1995 Dec 19;92(26):12456-60 Schneid H, Vazquez MP, Vacher C, Gourmelen M, Cabrol S, Le Bouc Y. The Beckwith-Wiedemann syndrome phenotype and the risk of cancer. Med Pediatr Oncol. 1997 Jun;28(6):4115 64 Beckwith-Wiedemann syndrome Mannens M DeBaun MR, Tucker MA. Risk of cancer during the first four years of life in children from The Beckwith-Wiedemann Syndrome Registry. J Pediatr. 1998 Mar;132(3 Pt 1):398-400 Alders M, Ryan A, Hodges M, Bliek J, Feinberg AP, Privitera O, Westerveld A, Little PF, Mannens M. Disruption of a novel imprinted zinc-finger gene, ZNF215, in Beckwith-Wiedemann syndrome. Am J Hum Genet. 2000 May;66(5):1473-84 Li M, Squire JA, Weksberg R. Molecular genetics of Wiedemann-Beckwith syndrome. Am J Med Genet. 1998 Oct 2;79(4):253-9 Steenman M, Westerveld A, Mannens M. Genetics of Beckwith-Wiedemann syndrome-associated tumors: common genetic pathways. Genes Chromosomes Cancer. 2000 May;28(1):1-13 Lee MP, DeBaun MR, Mitsuya K, Galonek HL, Brandenburg S, Oshimura M, Feinberg AP. Loss of imprinting of a paternally expressed transcript, with antisense orientation to KVLQT1, occurs frequently in Beckwith-Wiedemann syndrome and is independent of insulin-like growth factor II imprinting. Proc Natl Acad Sci U S A. 1999 Apr 27;96(9):5203-8 Atlas Genet Cytogenet Oncol Haematol. 2001; 5(1) This article should be referenced as such: Mannens M. Beckwith-Wiedemann syndrome. Atlas Genet Cytogenet Oncol Haematol. 2001; 5(1):62-65. 65