* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Translation I
Signal transduction wikipedia , lookup
Cell nucleus wikipedia , lookup
Protein moonlighting wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Protein structure prediction wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
List of types of proteins wikipedia , lookup
Gene expression wikipedia , lookup
Biosynthesis wikipedia , lookup
Genetic code wikipedia , lookup
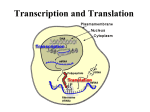
Translation I Gerhard Wagner BCMP200 12/2/2002 [email protected] Overview • Lecture 1 – Events of translation: • initiation, elongation, termination, folding – Machinery involved in translation • tRNAs, synthetases, ribosomes, translation factors – General regulatory mechanisms • Lecture 2 – Initiation • Lecture 3 – Elongation, termination, protein folding – regulation – Methods for studying translation Translation - big picture Initiation: Recruit fMet-tRNAMet, mRNA, large particle Elongation: Synthesize protein Termination: Stop synthesis, release protein Events of Translation • Initiation (very different between prokaryotes and eukaryotes) – – – – – – Dissociation of ribosome Binding of initiation factor (IF1/eIF1A) to A-site of small subunit Binding of other factors Recruitment of f-Met-tRNAMet to P-site of small particle Recruitment of mRNA to small particle Binding of large particle - dissociation of initiation factors • Elongation (similar between pro- and eukaryotes) – – – – Entry of aa-tRNA to A-site Peptide-bond formation Translocation of mRNA and tRNAs to P and E sites Entry of next aa-tRNA to A-site etc. • Termination (similar between pro- and eukaryotes) – Entry of release factor to recognize termination codon – Exit of polypeptide and release factors • Protein folding Players of translation • • • • • Ribosome (RNA, proteins) mRNA tRNA Aminoacyl-tRNA synthetases Translation factors (initiation, elongation,termination) Differences between eubacteria and eukaryotes Bacteria Eukaryotes • Ribosome: 30S+50S -> 70S • Few initiation factors: • Ribosome: 40S+60S-> 80S • Many initiation factors – IF-1(eIF1A), IF-2(eIF5B), IF-3 (?) • Elongation factors – EF1A (EF-Tu), EF1B (EF-Ts), EF2 (EF-G) • Release factors – RF-1, RF2, RF3 • Ribosome recycling factor – RRF • mRNA is not capped • Direct binding of 30S particle next to initiation codon (AUG) at ShineDalgarno sequence, 5’-AGGAGGU3’ • Translation coupled to transcription – eIF1, eIF1A, eIF2, eIF2B, eIF3, eIF4A, eIF4B, eIF4E, eIF4F, eIF4G, eIF4H, eIF5, eIF5B, eIF6 • Elongation factors – eEF1, eEF2 • Release factors – eRF1, eRF3 • Most mRNA is capped at 5’ end and polyadenylated at 3’ end • 40S particle is recruited to 5’ cap structure or poly(A) tail or an internal ribosome entry site (IRES) • Translation in always (?) in cytoplasm apart from transcription tRNA • Up to 50 (eukaryotes), or 30-35 (bacteria) different tRNAs • Cloverleaf structure • Unusual bases - covalent modification after transcription but before tRNAs leave nucleus • Acceptor arm: • 7 base pairs followed by xCCA-3’ aa attached to 2’ or 3’-OH of terminal A by ClassI and ClassII aa-tRNA synthetases, respectively TYC arm forms one continuous helix with acceptor arm • D arm (dihydro-uridine) interacts with TYC loop via unusual Hbonds • V loop short in Class I tRNAs, long in Class II. • Anticodon arm contains base triplet that pairs with mRNA codon 3’ TYC D 5’ V Anticodon arm TYC loop D loop acceptor arm C C 5’ V loop anticodon loop 3’ A Aminoacyl-tRNA synthetases • Synthetase attaches aa to tRNA in a two-step process: – • • • • adenylation of aa 20 aa-tRNA synthetases, one for each aa. Bacteria have often fewer synthetases, and one synthetase attaches different amino acids to tRNA. Another enzyme then chemically modifies the incorrectly attached aa so that it corresponds to the anticodon of the tRNA Two classes of aa-tRNA synthetases Class I binds minor groove of acceptor arm, Class II binds major groove of acceptor arm (there are newly found exceptions) aa-tRNA synthetases have been engineered to incorporate unusual amino acids (P. Schultz, S. Yokoyama) Asp-tRNA-synthetase O H2N CH O + ATP C CH O CH3 ribose P O H2N O adenine CH3 AMP + 2Pi C O ribose A of tRNA aa-tRNA synthase Complex of ClassI Tyr-tRNA synthetase with tRNAtyr Fig. 3. Interactions between tyrosyl-tRNA synthetase and tRNAtyr. (A) The C-terminal domain (orange) binds in the elbow between the long variable arm and the anti-codon stem of the tRNA (red backbone, green bases). The anti-codon stem loop interacts with both the C-terminal domain and the -helical domain (pink). The tRNA makes no contact with the catalytic domain of the same subunit (cyan). (B) The unusual conformation of the anti-codon triplet in which Ade-36 is stacked on Gua-34, while Psu-35 bulges out. (C) Base-specific interactions of Asp-259 from the -helical domain with Gua-34 and Asp-423 from the C-terminal domain with Psu-35. Tyr-tRNA synthase complex with tRNATyr mRNA • Linear in bacteria - can circularize in eukaryotes (via Pabp, eIF4G and eIF4E) • In bacteria, ribosome is recruited to AUG codon via a Shine-Dalgarno sequence – 5’ AGGAGGU-(X)3-10-AUG 3’ • In eukaryotes, mRNA is usually capped and poly-adenylated - a consensus sequence is found around the initiation codon -ACCAUGG(Kozak sequence) O N HN 2HN Me N 5’ N HO O O O O O P O P O P O O O O OH Base 5’ O HO OH • 5’end-5’UTR-AUG-coding region-stop codon-3’UTR-poly(A)tail Capped mRNA • • Capping happens right after transcription, after about 25 nucleotides have been synthesized Capping by three enzymes: – – – – – • • • Phosphatase removes one phosphate from 5’ end Guanyl transferase adds a GMP in reverse linkage (5’ to 5’ instead of 5’ to 3’) Methyl transferase adds a methyl to the guanosine Some RNAs are also methylated at the second nucleotide All three enzymes bind to the phosphorylated RNA polymerase tail Cap addition distinguishes mRNA from other RNAs and helps to direct the ribosome to mRNA Cap is recognized by the cap-binding complex (CBC), consisting of two proteins, CBP80 and CBP20. Cap is stacked between two tyrosines Y20 and Y43 of CBP20. Bindin is achieved via the p-stacking effect. CBC stabilizes the mRNA and interacts with nuclear pore complex during export of mRNA. In cytoplasm, CBC is replaced with eIF4E that helps to recruit the ribosome to mRNA. Here the m7G is stacked between two tryptophanes (p-stacking ) Polyadenylation of mRNA - binding of Pabp and other factors • 3’-end is polyadenylated by CstF (cleavage stimulating factor), CPSF (cleavage and polyadenylation specificity factor) and PAP (poly(A) polymerase). • Poly(A) tail binds multiple copies of Pabp (poly(A)-binding protein) • Other factors bind mRNA, SR proteins, hnRNPs etc. bind to mRNA and make it ready for export • Some but not all of the attached proteins (CBC, Pabp) are exported with the mRNA • In cytosole, CBC is replaced with eIF4E for translation. mRNA structure • Eukaryotic mRNA can circularize by coupling the cap-binding protien eIF4E and Pabp to the scaffold protein eIF4G eIF4E m7GpppN-- AUG eIF4G Pabp AAAAAAAA • • 5’UTR sometimes contains long GC-rich regions that tend to form secondary structure and inhibit ribosome scanning. This is found particularly in mRNAs for growth-promoting proteins (growthfactors, oncogene products) and is thought to be a regulatory element to prevent uncontrolled cell growth. Some mRNAs contain secondary structures that allow for direct binding of the small ribosomal particle, aided by segments of eIF4G. This is called an internal ribosome entry site, IRES. Genetic Code and Codon Usage Escherichia coli [gbbct]: 11985 CDS's (3688954 codons) • Genetic Code is degenerate. – – – – • • • 64 codons 20 amino acids < 50 tRNAs 20 synthetases Wobble base pairing: in some tRNAs, 3rd base of anticodon can pair with different bases of codons Common aa have multiple codons and multiple tRNAs, rarest amino acids, trp and met are each encoded by only one codon Codon usage frequency varies with organism. Can be looked up at: http://www.kazusa.or.jp/codon/ • Important for expression of mammalian proteins in E.coli. Optimize codons for expression. fields: [triplet] [amino acid] [fraction] [frequency: per thousand] ([number]) UUU F 0.58 22.2 ( 81958) UUC F 0.42 16.0 ( 59150) UUA L 0.14 14.4 ( 53048) UUG L 0.13 13.0 ( 47827) UCU S 0.17 10.4 ( 38427) UAU Y 0.59 17.5 ( 64717) UGU C 0.46 5.2 ( 19357) UCC S 0.15 9.1 ( 33697) UAC Y 0.41 12.2 ( 44909) UGC C 0.54 6.1 ( 22348) UCA S 0.14 9.0 ( 33177) UAA * 0.61 2.0 ( 7408) UGA * 0.30 1.0 ( 3684) UCG S 0.14 8.5 ( 31383) UAG * 0.08 0.3 ( 996) UGG W 1.00 13.9 ( 51416) CUU L 0.12 11.9 ( 43948) CCU P 0.18 7.5 ( 27601) CAU H 0.58 12.5 ( 46295) CGU R 0.36 19.9 ( 73524) CUC L 0.10 10.2 ( 37561) CCC P 0.13 5.4 ( 19840) CAC H 0.42 9.3 ( 34207) CGC R 0.36 19.6 ( 72420) CUA L 0.04 4.2 ( 15655) CCA P 0.20 8.6 ( 31840) CAA Q 0.34 14.6 ( 53879) CGA R 0.07 3.8 ( 13999) CUG L 0.47 48.2 (177820) CCG P 0.49 20.8 ( 76842) CAG Q 0.66 28.4 (104717) CGG R 0.11 5.9 ( 21773) AUU I 0.49 29.8 (109873) ACU T 0.19 10.4 ( 38312) AAU N 0.49 20.7 ( 76457) AGU S 0.16 9.9 ( 36590) AUC I 0.39 23.6 ( 87131) ACC T 0.40 21.9 ( 80904) AAC N 0.51 21.4 ( 78873) AGC S 0.24 15.1 ( 55819) AUA I 0.12 7.0 ( 25709) ACA T 0.17 9.4 ( 34580) AAA K 0.74 35.3 (130185) AGA R 0.07 3.7 ( 13500) AUG M 1.00 26.4 ( 97325) ACG T 0.25 13.7 ( 50690) AAG K 0.26 12.5 ( 45938) AGG R 0.04 2.1 ( 7787) GUU V 0.28 19.8 ( 73179) GUC V 0.20 14.3 ( 52706) GUA V 0.17 11.6 ( 42768) GUG V 0.35 24.3 ( 89623) GCU A 0.18 17.1 ( 62923) GAU D 0.63 32.8 (120820) GGU G 0.35 25.4 ( 93737) GCC A 0.26 24.2 ( 89153) GAC D 0.37 19.2 ( 70721) GGC G 0.37 27.0 ( 99602) GCA A 0.23 21.2 ( 78120) GAA E 0.68 39.0 (144050) GGA G 0.13 9.6 ( 35295) GCG A 0.32 30.0 (110528) GAG E 0.32 18.7 ( 68998) GGG G 0.15 11.3 ( 41635) Homo sapiens [gbpri]: 50031 CDS's (21930294 codons) fields: [triplet] [amino acid] [fraction] [frequency: per thousand] ([number]) UUU F 0.46 17.1 (374332) UCU S 0.18 14.7 (323470) UAU Y 0.44 12.1 (264652) UGU C 0.45 10.1 (221863) UUC F 0.54 20.4 (448127) UCC S 0.22 17.5 (384476) UAC Y 0.56 15.5 (339473) UGC C 0.55 12.4 (271056) UUA L 0.07 7.3 (160731) UCA S 0.15 11.9 (260418) UAA * 0.28 0.8 ( 16884) UGA * 0.50 1.4 ( 30111) UUG L 0.13 12.7 (277774) UCG S 0.06 4.5 ( 98166) UAG * 0.22 0.6 ( 12911) UGG W 1.00 13.0 (284246) CUU L 0.13 12.9 (283480) CCU P 0.28 17.3 (380219) CAU H 0.41 10.6 (231860) CGU R 0.08 4.7 (102673) CUC L 0.20 19.5 (428574) CCC P 0.33 20.0 (439256) CAC H 0.59 15.0 (329569) CGC R 0.19 10.8 (236986) CUA L 0.07 7.0 (153837) CCA P 0.27 16.7 (367297) CAA Q 0.26 11.9 (261063) CGA R 0.11 6.3 (138297) CUG L 0.40 40.1 (880072) CCG P 0.11 7.0 (154028) CAG Q 0.74 34.4 (755209) CGG R 0.21 11.8 (257761) AUU I 0.36 15.8 (346233) ACU T 0.24 12.9 (283671) AAU N 0.46 16.7 (365457) AGU S 0.15 12.0 (263279) AUC I 0.48 21.3 (466577) ACC T 0.36 19.1 (419213) AAC N 0.54 19.3 (422697) AGC S 0.24 19.4 (424788) AUA I 0.16 7.2 (157385) ACA T 0.28 14.9 (325763) AAA K 0.42 24.0 (526117) AGA R 0.21 11.7 (255681) AUG M 1.00 22.3 (489160) ACG T 0.12 6.2 (135294) AAG K 0.58 32.5 (713826) AGG R 0.20 11.6 (254743) GUU V 0.18 10.9 (239795) GCU A 0.26 18.6 (408931) GAU D 0.46 22.1 (484271) GGU G 0.16 10.8 (237026) GUC V 0.24 14.6 (320190) GCC A 0.40 28.4 (622538) GAC D 0.54 25.7 (563848) GGC G 0.34 22.6 (495700) GUA V 0.11 7.0 (154102) GCA A 0.23 16.0 (350382) GAA E 0.42 29.0 (634985) GGA G 0.25 16.4 (358824) GUG V 0.47 28.7 (630151) GCG A 0.11 7.6 (165700) GAG E 0.58 40.3 (884368) GGG G 0.25 16.4 (360728) Ribosomes 80S (4.2M) 70S (2.5M) 60S (2.8M) 50S (1.6M) 30S (0.9M) 5S rRNA (120 nt) 23S rRNA (2900 nt) 34 proteins 16S rRNA (1540 nt) 21 proteins Prokaryotes 5S rRNA (120 nt) 28S rRNA (4700 nt) 5.8S rRNA (160 nt) ~49 proteins 40S (1.4M) 18S rRNA (1900 nt) 33 proteins Eukaryotes Distinct roles of ribosomal particles • Small particle gathers components, f-Met-tRNAMet, mRNA, initiation factors; is crucial for decoding • Some antibiotics (streptomycin) interfere with decoding process • Large particle joins after components have been assembled and performs protein synthesis, in the presence of small particle. • Large particle is target of macrolide antibiotics (erythromycin etc.) Ribosome structure • Ribosome was a main focus of structural biologists, and structure was solved in a 30-year effort • X-ray crystallography – Yonath (30S and 50S of eubacterium, work since 1980) • • Schluenzen et al. Cell, 102, 615 (2000) 30 S particle at 3.3Å resolution Harms et al., Cell 107, 679 (2002) 50 S particle at 3.1 Å resolution – Steitz & Moore (50S) • • • • Ban et al. Cell, 93, 1105–1115, (1998) 9Å resolution Ban et al. Nature, 400, 841- (1999) 5Å resolution Ban et al. Science 289, 905-920 (2000) 2.4Å resolution Nissen et al. Science 289, 920-930 (2000) Ribosome activity – Ramakrishnan (30S) • • Clemons et al., Nature, 400, 833 (1999) 5.5 Å Carter et al. Science, 291, 498 (2001) 3.1 Å structure with IF1 bound – Noller (70S) • • Cate et al., Science 285 2095-2104 (1999) 7.8Å Yusupov et al. Science 292, 883-896 (2001) 5.5 Å • Cryo-EM – – Agarwal et al., PNAS 95, 6134 (1988) Stark et al. Cell 100, 301 (2000) 50S particle of bacterial ribosome (Steitz & Moore) • 5Å resolution structure • 23S RNA (2900 nucleotides), 5S RNA (120 nucleotides), 33 proteins • Location of some of the proteins • Deep active site cleft • Acceptor arms of aa-tRNAs must dive into cleft • Exit tunnel at the bottom of the large active-site cleft (initial evidence from work of Unwin et al., 1986, and Yonath and Wittman, 1987 • Positioning of elongation factor EF-G •Central protuberance (CP), L1 protein, crown view Ban et al., Nature 400, 841 (1999) Mapping of the tunnel with tungsten clusters 50S particle Ban et al. Cell, 93, 1105–1115, (1998) 9 Å resolution Ban et al. Nature, 400, 841 (1999) 5Å resolution Ban et al. Science 289, 905 (2000) 2.4Å resolution • All RNA and proteins defined • Catalytic cleft in center • Exit tunnel 30S particle (Ramakrishnan) • Clemons et al., Nature, 400, 833 (1999) 5.5 Å • Landmarks: – Head (H), Neck (N), Platform (P), Body (Bo), Shoulder (Sh) 30 S particle, Clemons et al. 5.5 Å Location of prominent helices and proteins 30S particle, location of initiation factor IF1 • • • • Clemons et al., Science, 2001 Carter et al. Fig. 2B 30S particle with IF1 bound Landmarks: – Head (H), Neck (N), Platform (P), Body (Bo), Shoulder (Sh) • Helix 44 and S12 interact with initiation factor IF1, which sits in the A-site IF1 S12 H44 30S particle with tRNA-binding sites Head Location of tRNA-binding sites S12 P Helix 44 E Neck Platform A Shoulder S12 H44 Body Carter et al., Science 2001 70S ribosome (Noller group) Cate et al., Science 1999 Location of tRNAs Cate et al., Science 1999 Model of Location of tRNAs in 50S particle (Nissen et al.) Acceptor arms in catalytic cleft Anticodon arms stick out Macrolide bound to exit channel (Hansen et al. Mol. Cell 10, 117 (2002) Ribosome is a ribozyme • First proposed by Crick, J. Mol. Biol. 38, 367(1968) • Affinity labeling experiments by Noller’s group (Barta et al., 1984) showed that U2584 and U2585 of a highly conserved internal loop from domain V of 23S rRNA are close to CCA-end of P-site • Ribosome can be depleted of many of its proteins and maintains peptidyl-transferase activity • Ribosome structures show that there is no protein near the site of peptide synthesis (closest distance is 18 Å) • Good evidence an proposals for the enzymatic mechanism are shown in Nissen et al. (2000).