* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Fatty acid synthesis wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Phosphorylation wikipedia , lookup
Free-radical theory of aging wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Mitochondrion wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Coenzyme Q10 wikipedia , lookup
Metalloprotein wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Biochemistry wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Electron transport chain wikipedia , lookup
Citric acid cycle wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
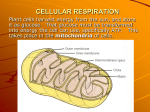
Pyruvate Carboxylase Reversing the final steps O- O OO- P H O O- O P H O CH2OPO32O O- O- O OH OH OH OH OH OH OH O O P O-O P OH -O OH CH2OPO32O OH OH OH OH O- O- O OH O- OH OH Inverse regulation of glycolysis and gluconeogenesis Hexokinase IV in liver Hexokinase I in muscle HxkIV Km = 10 mM When blood glucose drops below 5 mM, F6P inhibits it. This way liver does not compete with muscle for glucose Regulation of phosphofructokinase Electron transport and oxidative phosphorylation Glucose is completely oxidized to CO2 through the enzymatic reactions of glycolysis and the citric acid cycle. The redox equation for this process is: C6H12O6 + 6O2 ---> 6CO2 + 6H2O ΔG°’ = -2823 kJ.mol-1 Which can be represented by two half reactions: C6H12O6 + 6H2O ---> 6CO2 + 24H+ + 24e- glucose is oxidized 6O2 + 24H+ + 24e- ---> 12H2O molecular oxygen is reduced In living systems the electron transfer process connecting these two half reactions occurs through a multistep pathway that harnesses the liberated free energy to form ATP. The 12 electron pairs involved in glucose oxidation are not transferred directly to O2. Rather they are transferred to coenzymes NAD+ and FAD to form NADH and FADH2 10 NADH : 20 e2 FADH2 : 4 e- The sites of electron transfer that form NADH and FADH2 in glycolysis and the citric acid cycle are represented in the figure. The electrons are extracted from the cofactors by reoxidation and then join the electron-transport chain, in this process, protons are expelled from the mitochondrion. The free energy stored in the resulting pH gradient drives the synthesis of ATP from ADP and Pi (inorganic phosphate) through oxidative phosphorylation. Reoxidation of NADH ~ 3 ATP Reoxidation of FADH2 ~ 2 ATP A total of 38 ATP are produced per each molecule of glucose completely oxidized to CO2 and H2O (including the 2 ATP made in glycolysis and the 2 ATP made in the citric acid cycle) NAD+ and FAD coenzymes are reduced during glucose oxidation Mitochondria is the site of eukaryotic oxidative metabolism 0.5 m in diameter and 1 m long (about the size of a bacterium) The outer membrane contains porin, a protein that forms pores and allows free difussion of up to 10 kD molecules The inner membrane is a lot more dense and is permeable only to O2, CO2 and H2O. Contains numerous transport proteins that control metabolite passage. Mitochondrion is not a regular shaped organelle it is a dynamic organelle that is reticulated throughout the cell Electrons enter the electron transport chain onto Q Glycerol phosphate shuttle NADH:Q Oxidoreductase Succinate dehydrogenase Fatty acid metabolism Reduced state has more protons than the oxidized state! Redox loops pumps out four protons! The glycerophosphate shuttle mainly occurs in rapidly metabolizing tissues. NADH from TCA cycle are generated in mitochondrial matrix NADH from glycolysis are generated in cytoplasm Problem: No way to transport NADH into the mitochondrion to be reoxidized! Solution: Use the malate-aspartate shuttle Complex I: NADH:CoQ oxidoreductase NADH + H+ + CoQ(ox) + 4H+(in) ---> NAD+ + CoQH2(red) + 4H+(out) ∆E = 0.360 V ∆G = -69.5 kJ/mol -0.030 -0.25 -0.30 -0.32 +0.045 -0.32 -0.30 Ubiquinone +0.045 What is FMN? Flavin mononucleotide What are FeS clusters? Complex II: succinate dehydrogenase Succinnate:CoQ oxidoreductase FADH2 + CoQ(ox) ---> FAD + CoQH2(red) ∆E = 0.085 V ∆G = -16.4 kJ/mol -0.031 -0.040 -0.030 -0.245 -0.060 -0.080 +0.045 Succinate -0.031 Heme b Complex III CoQH2(red) + 2cyt c(ox) + 2H+(in) ---> CoQ(ox) + 2cyt c(red) + 4H+(out) ∆E = 0.190 V ∆G = -36.7 kJ/mol +0.235 +0.215 +0.280 -0.030 +0.030 +0.045 CoQH2 + cyt c1(ox) ---> CoQ•- + cyt c1(red) + 2H+ (out) Cycle I CoQH2 + CoQ•- + cyt c1(ox) + 2H+(in) ---> CoQ + CoQH2 + cyt c1(red) + 2H+ (out) Cycle II CoQH2 + 2cyt c1(ox) + 2H+(in) ---> CoQ + 2cyt c1(red) + 4H+ (out) Net Reaction 4 protons pumped instead of 2 Ubiquinone = +0.045 Cytochrome c: an electron bottle neck Complex IV 4 cytochrome c2+ + O2 + 8H+(in) => 4 cytochrome c 3+ + 2H2O + 4H+(out) ∆E = 0.580 V ∆G = -112 kJ/mol +0.235 +0.245 +0.815 +0.385 +0.340 2H+ If 2 electrons enter at complex I 4 + 4 + 2 = 10 protons pumped out If 2 electrons enter at complex II or Glycerol dehydrogenase or fatty acid metabolism 4 + 2 = 6 protons pumped out Proton concentration gradient pH is lower in intermembrane space than in the mitochondrial matrix GA - GA0’ = RT ln [A] A(out) A(in) ∆GA = GA(in) - GA(out) = RT ln ( [A]in ) [A]out If the solute is charged there is another aspect of the equation: electrical potential Membrane potential = ∆ = (in) - (out) ∆GA = RT ln ( [A]in ) + ZAF ∆ [A]out Free energy is a combination of chemical and electrical potential ∆GA = RT ln ( [A]in ) + ZAF ∆ [A]out ∆G = 2.3RT [pH(in) - pH(out)] + ZF ∆ ∆ = 0.168V = 0.168 J•C-1 ∆pH = 0.75 [≈ 210,000V•cm-1!!!!] F = 96,485 C•mol-1 Z = +1 ∆G = 21.5 kJ•mol-1 ∆G of ATP synthesis = 40 to 50 kJ•mol-1 ATP synthase allows protons to flow back in Harnesses the free energy in the process The gamma subunit: the rotor Per glucose 10 NADH : 20 e2 FADH2 : 4 eIf 2 electrons enter at complex I 4 + 4 + 2 = 10 protons pumped out If 2 electrons enter at complex II or Glycerol dehydrogenase 4 + 2 = 6 protons pumped out Per glucose 120 protons 120/3 = 40 + 4ATP from glycolysis and TCA 44 ATP! Why only 38? ATP synthase is nearly 100% efficient: so why do you not get 1ATP per 3 H+? H+ transported -0.3ATP 2.7 ATP/ NADH H+ transported -0.3ATP 2.4 ATP/ NADH What about fatty acid biosynthesis, succinate dehydrogenase and glycerol phosphate shuttle? 1.5 ATP per pair of electrons... How is electron transport regulated? What is uncoupling? pH 8 pH 5 The c-subunits of F0 ATPase Electrons sometimes leak out of the chain onto molecular oxygen. As much 5% leak out onto oxygen Incompletely reduced oxygen is toxic Reactive oxygen species Superoxide •O2Peroxide O22Hydroxyl radical •OH Where does this occur? O2 O O O2- H3CO CH3 H3CO CH3 H3CO R H3CO R O O H B Superoxide comes from Q not Complex IV Why does this happen? The ox-tox hypothesis Oxygen is soluble in membranes and it can oxidize lipids, albeit slowly. Too much oxygen is toxic. Mitochondria detoxify oxygen by reducing it to water. This is how they were beneficial What about superoxide and peroxide? Maybe these are allowed to leak out as signal molecules that apprise the cell of the energetic state…. Maybe the generation of superoxide, which is not soluble in membrane is a way to detoxifying oxygen…… Mitochondria and apoptosis Cytochrome c is released from intermembrane space