* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter 7: Cellular Respiration and Fermentation
Isotopic labeling wikipedia , lookup
Gaseous signaling molecules wikipedia , lookup
Carbon sink wikipedia , lookup
Mitochondrion wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biosequestration wikipedia , lookup
Phosphorylation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Electron transport chain wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Biochemistry wikipedia , lookup
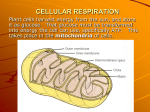
Chapter 9: Cellular Respiration AP Biology Oxidation and Reduction • E is gained by the transfer of e’s • The relocation of e-’s releases the stored E and the E ultimately makes ATP Redox Reactions • Oxidation: the loss of e-’s from a substance • Reduction: the gain of e-’s by a substance • E must be added to pull e-’s from an atom Redox Reactions • A redox reaction that relocates an e- from a less electronegative atom to a more electronegative atom loses potential E Hydrogen • In most redox reactions, it is not only the e- that is transferred but in most biological reactions the whole hydrogen atom is transferred Carbohydrates and Fats • Contain high levels of hydrogen and their electrons • There is a barrier that keeps sugar from combining immediately with O2 • This barrier is reduced inside the body with the help of enzymes Cellular Respiration C6H12O6 + 6O2 6H2O + 6CO2 + E Introduction • Cellular Respiration is a catabolic pathway. –Does not directly perform cellular work??? –Redox reactions!! –Importance of Hydrogen?? Introduction • Does glucose react instantaneously? Spontaneously? • Activation barrier/ Enzymes(lots of them) – Lowers activation energy – Allows breakdown to proceed spontaneously Cellular Respiration • What is a potential problem with the breakdown of glucose as a spontaneous reaction?? – Hint: TNT, Gasoline • How does the cell prevent this problem? – Does not release energy all at once – Multi- step process catalyzed by specific enzymes Cellular Respiration • With each step, electrons are released with a proton (hydrogen atom) • Each hydrogen is transferred to a coenzyme (NAD+) • And eventually to Oxygen Nicotinamide Adenine Dinucleotide • NAD+ • Derivative of the vitamin niacin • Coenzyme – Oxidizing agent • Will be reduced to NADH – (2 electrons and Hydrogen) – Dehydrogenase-removes two hydrogen from substrate Nicotinamide Adenine Dinucleotide Glycolysis: • • • • • Occurs in the cytoplasm Net gain of 2 ATP and 2 NADH Start: 6 carbon glucose End: 2- 3 carbon pyruvate molecules Ten steps- two phases – Energy investment phase – Energy payoff phase Glycolysis: • Energy investment stage: • Glucose phosphorylated by ATP(2) – Unstable – Splits • Each 3 carbon molecule is phosphorylated again – Inorganic phosphate comes from cytosol not ATP Glycolysis: • Energy Payoff Phase: • Each 3 carbon molecule reduces NAD+ to NADH • Each 3 carbon molecule gives up its 2 phosphates to ADP to form 4 ATP. Glycolysis: Summary • Reactants: – – – – Glucose NAD+ ATP (2) ADP (2) • • • • Products: Pyruvate (2) NADH (2) ATP (4) Mitochondria Structure • Double membrane organelle – Outer membrane: very permeable – Inner membrane: • selectively permeable – pyruvate (yes) – NADH (no) • contains electron transport proteins • Cristae- inner foldings – Increase surface area • similar to plasma membrane of bacteria Mitochondria Structure • Double membrane organelle – Matrix: inside inner membrane • Protein rich solution: enzymes • In between cristae Citric Acid Cycle: – Krebs Cycle • Tricarboxylic Acid Cycle – Matrix – Reactants: • 2 molecules of pyruvate • Each makes a circuit through the cycle – One glucose = two pyruvate = two turns of the cycle – Products: • For each pyruvate – 3 NAD+ – 1 FAD+ – 1 ADP + P 3 NADH 1 FADH2 1 ATP Oxidation of Pyruvate: • Pyruvate must first be converted to Acetyl CoA – Pyruvate dehydrogenase – Each pyruvate molecule loses one Carbon and two Oxygen- Acetyl group – Acetyl group attaches to CoA molecule forming acetyl CoA – Reduction of NAD+ molecule to NADH • 2 total- one for each pyruvate Cyclic Nature of Citric Acid Cycle • CoA transfers 2 carbon molecule – Transfers 2 carbon acetyl group to 4 carbon oxaloacetate – Results = 6 carbon citrate – Start of cycle Cyclic Nature of Citric Acid Cycle • Citrate- goes through a series of oxidation reactions – Priming/ rearrangement stage • Prepares the 6 carbon citrate for energy extraction – Oxidized by NAD+ – Carbon dioxide Cyclic Nature of Citric Acid Cycle • Citrate also loses 2 carbon atoms (CO2) eventually returning to 4 carbon oxaloacetate again – – – – – Energy Extraction/ Acetyl group stage More reduction of NAD+ Reduction of FAD ATP produced- substrate level phosphorylation Cycle starts over again Important Features • NAD+ and FAD+ are reduced by the oxidation of an organic compound (transfer of H atom). • 1 ATP molecule is formed by substrate level phosphorylation during each turn of cycle (net per glucose = 2 ATP) • For each turn of the cycle, 3 Carbon atoms are lost to Carbon Dioxide – All 6 carbons exit the system by the end of the Kreb cycle. Oxidative Phosphorylation • Electron transport is coupled with ATP synthesis via chemiosmosis. • Over all drop in ΔG as electrons are transferred from NADH to Oxygen – Releases energy in manageable amounts • Create proton motive force – Drives the production of ATP Electron Transport Chain • Inner Mitochondrial membrane • Series (I - IV) of protein complexes – Complexes one – three have increasing affinity for electrons • Prosthetic groups: non-protein components essential to certain enzymes • Redox (downhill) reactions • Does not directly make ATP- eases the fall Prosthetic Groups: • FMN- flavin mono nucleotide– Gets reduced by NADH at complex I • CoQ- Ubiquinone– very hydrophobic – very mobile – Carries between complex I/II and complex III Prosthetic Groups: • Iron/Sulfur cluster– gets reduced by FADH2 at complex II – Transfer electrons between cytochromes • Cytochromes- transfers electrons to oxygen – Heme- Fe atom- carries electrons Chemiosmosis • The formation of a hydrogen ion gradient drives the cellular process of ATP synthesis – Proton motive force Chemiosmosis • Final protein complex = F0F1 protein – Catalyzed by ATP synthase – Oxidative phosphorylation: synthesis of ATP from ADP and Pi – 3 to 4 H+ to generate 1 ATP Animation • http://www.science.smith.edu/d epartments/Biology/Bio231/etc. html Fermentation If a cell runs out of O2, all the ecarriers are stuck in reduced form, halting system • Pyruvate produced by glycolysis acts as alternative acceptor of H from NADH, keeping glycolysis going to allow small ATP production Alcoholic Fermentation • Yeasts break down sugar into pyruvate. • Each pyruvate is dismantled into a molecule of CO2 and a 2C compound acetaldehyde • Acetaldehyde is reduced by accepting 2H's from NADH and H+ forming 2C alcohol ethanol (ethyl alcohol) Lactic Acid Fermentation • Occurs during strenuous exercise • Pyruvate from glycolysis is reduced by accepting hydrogens from NADH and H+ • Pyruvate converted into 3C compound, lactate Metabolic Energy Systesm: • Phosphagen pathway- high powered activities that last around 10 secs • Glycolytic pathway- moderately powered activities that last two minutes • Oxidative pathway- low powered pathways that last more than several minutes Respiration W/O O2 Anaerobic respiration: uses nitrate or sulfate as final electron acceptor Fermentation: the anaerobic breakdown of food molecules in which the final e- acceptor is an organic molecule