* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Darifenacin Hydrobromide
National Institute for Health and Care Excellence wikipedia , lookup
Discovery and development of cephalosporins wikipedia , lookup
Drug interaction wikipedia , lookup
Discovery and development of ACE inhibitors wikipedia , lookup
Discovery and development of TRPV1 antagonists wikipedia , lookup
CCR5 receptor antagonist wikipedia , lookup
Toxicodynamics wikipedia , lookup
NMDA receptor wikipedia , lookup
5-HT2C receptor agonist wikipedia , lookup
Discovery and development of beta-blockers wikipedia , lookup
Discovery and development of antiandrogens wikipedia , lookup
5-HT3 antagonist wikipedia , lookup
Psychopharmacology wikipedia , lookup
Nicotinic agonist wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Neuropharmacology wikipedia , lookup
Discovery and development of angiotensin receptor blockers wikipedia , lookup
Dydrogesterone wikipedia , lookup
Cannabinoid receptor antagonist wikipedia , lookup
Darifenacin Hydrobromide
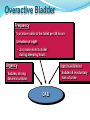
Overactive Bladder
Frequency
• 8 or more visits to the toilet per 24 hours
Urination at night
• 2 or more visits to toilet
during sleeping hours
Urgency
Urge Incontinence
• Sudden, strong
desire to urinate
• Sudden & involuntary
loss of urine
OAB
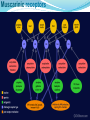
Muscarinic receptors
Treatment
Antimuscarinic drugs
First-generation----e.g Oxybutynin
(primarily selective)
Second-generation---- e.g Tolterodine
(balanced selective M2/M3)
New drugs----e.g Darifenacin
(M3 selective receptor antagonis)
Darifenacin hydrobromide
----A potent and competitive M3 selective
receptor antagonist
a white to almost white, crystalline powder.
Brand name : Emselex
Synonyms: (S)-2-{1-[2-(2,3-Dihydrobenzofuran-5yl)Ethyl]-3-Pyrrolidinyl}-2,2-Diphenylacetamide
Hydrobromide
HBr
Manufacturer: Novartis & Pfizer
Oct.28.2004—European Commission has granted
Marketing Authorization Emselex for the treatment
of overactive.
Dec.23.2004—FDA has approved darifenacin HBr for
the treatment of OAB.
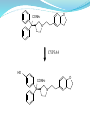
The structure-activity relationship
Part A imidazole ring being
replaced by alkyl will
significantly affect anticholinergic receptor activity
and selectivity;
Part B for the connecting
part, usually two carbon
anticholinergic activity at
best. carbon chain
growes, activity will
decrease;
The structure-activity relationship
Part C of the amide or H
atom, anticholinergic
activity and M3 receptor
selectivity better, Nmonosubstituted, N, Ndisubstituted or ester is
anticholinergic activity
decreases even without
anticholinergic activity.
The structure-activity relationship
Part D benzene
generally no replaced if
heterocyclic or alkyl
replace it activity
decreases, one of
benzene may be
replaced by six atom
ring : Oxybutynin.
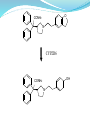
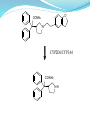
Synthesis:
Darifenacin
Darifenacin is a competitive muscarinic receptor
antagonist labeled for the treatment of overactive
bladder with symptoms of urge urinary
incontinence, urgency, and frequency.
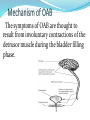
The symptoms of OAB are thought to
result from involuntary contractions of the
detrusor muscle during the bladder filling
phase.
M2,M3 muscarinic receptors with a
ratio of 3:1 exist in human bladder
tissue. Although the density of M2
receptors is much greater than that of
the M3 subtype, M3 receptor subtype
is predominant in the mediation of
bladder contraction.
M3 are responsible for urinary bladder
contraction,gastrointestinal smooth muscle
contraction, saliva production, and iris
sphincter regulation.
Darifenacin has been shown to have high
affinity and selectivity for the muscarinic M3
receptor, with low selectivity for the other
muscarinic receptor subtypes.
Effect
Help to reduce the incidence of urinary
incontinence .
Increase urinary bladder reserves .
Reduce the frequency of urination.
Reduce the oppressive sense of eager to
urinate and urinary urgency sense.
dry mouth
constipation
blurred vision
heat prostration
others
Pharmacokinetic properties
Darifenacin is metabolised by CYP3A4
and CYP2D6.
CYP3A4
CYP2D6
Absorption
first-pass metabolism
7.5 mg daily doses
15 mg daily doses
ENABLEX® 7.5 mg
(N = 68 EM, 5 PM)
AUC2
4
(ng.h/
mL)
Cma
Cavg Tma
x
(ng/ x
(ng/
mL) (h)
mL)
ENABLEX®15 mg
(N = 102 EM, 17 PM)
t1/2
(h)
E
12.43
29.24 2.01 1.22 6.49
M
(5.64)
(15.47) (1.04) (0.64) (4.19)
a
P 67.56
M (13.13)
AUC24
(ng.h/
mL)
Cma
Cavg Tma
x
(ng/ x
(ng/
mL) (h)
mL)
t1/2
(h)
12.05
88.90
5.76 3.70 7.61
(12.37)
(67.87) (4.24) (2.83) (5.06)
b
4.27 2.81 5.20
157.71
9.99 6.58 6.71
19.95 c
7.40 d
(0.98) (0.55) (1.79)
(77.08) (5.09) (3.22) (3.58)
Distribution
lipophilic base
98% bound to plasma proteins
Vss =163 litres
Metabolism
Darifenacin metabolism (oral
administration)
CYP3A4 CYP2D6 in the liver
CYP3A4 in the gut wall
three main metabolic routes
monohydroxylation in the
dihydrobenzofuran ring
dihydrobenzofuran ring opening
N-dealkylation of the pyrrolidine
nitrogen
O
CONH2
N
CYP3A4
HO
O
CONH2
N
O
CONH2
N
CYP2D6
OH
CONH2
N
O
CONH2
N
CYP2D6/CYP3A4
CONH2
NH
Excretion
Following administration of an oral
dose of 14C-darifenacin solution to
healthy volunteers, approximately
60% of the radioactivity was
recovered in the urine and 40% in
the faeces. Only a small percentage
of the excreted dose was unchanged
darifenacin (3%).
Efficacy against urinary frequency
NICE states that :
there is NO evidence of clinically
important efficacy differences among
antimuscarinic drugs
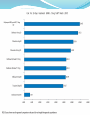
Adverse events
Selected adverse events
Selection of antimuscarinic drug for treatment of
OAB
NICE states that :
Non-proprietary, immediate-release
oxybutynin, which is the most costeffective of the available options, should
be offered as first-line antimuscarinic if
bladder training is ineffective.
If not tolerated, darifenacin, solifenacin,
tolterodine, trospium or an extendedrelease or transdermal formulations of
oxybutynin should be considered.
MEMBERS
0440101
0440118
0441707
0440220
0440211
Cui Sisi
Wang Lutai
Lu Lisha
Wang Yan
Gao Wei