* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Stereoselective reactions of the carbonyl group
Fischer–Tropsch process wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
Asymmetric hydrogenation wikipedia , lookup
Kinetic resolution wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Elias James Corey wikipedia , lookup
Discodermolide wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Stille reaction wikipedia , lookup
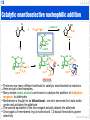
Aldol reaction wikipedia , lookup
Ene reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Hydroformylation wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
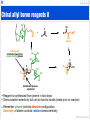
1 Stereoselective reactions of the carbonyl group • We have seen many examples of substrate control in nucleophilic addition to the • • carbonyl group (Felkin-Ahn & chelation control) If molecule does not contain a stereogenic centre then we can use a chiral auxiliary The chiral auxiliary can be removed at a later stage Me Me Me O Ph O O L L Mg O O Me MeMgBr –78°C Me H Me Me O OH H Me Me H Me O Ph O 98% de Me • Opposite diastereoisomer can be obtained from reduction of the ketone • Note: there is lower diastereoselectivity in the second addition as the nucleophile, ...‘H–’ is smaller Me Me Me Me O Ph O O Me KBH(i-OPr)3 O Me Me Ph O OH Me H 90% de Advanced organic 2 Chiral auxiliary in synthesis Me Me O Me Me Ph O O RMgBr –78°C Me Me O Ph O Me OH LiAlH4 HO Me OH Me Me Me O3 –78°C O Me O Me (–)-frontalin 100% ee • The chiral auxiliary, 8-phenylmenthol, has been utilised to form the pheromone, frontalin Advanced organic 3 Sulfoxides as chiral auxiliaries hydride directed to si face lithium can chelate 2 oxygens O Tol S LiAlH4 O R O HO H Li R H AlH3 O Raney Ni S R HO H R Tol Me 80% de O S Tol R DIBAL Tol S O O i-Bu Al i-Bu HO H H R H Al i-Bu i-Bu oxygens are not directly tethered O Raney Ni S Tol HO H R Me 70% de dipoles directed in opposing directions • Underrated chiral auxiliary - easily introduced, performs many reactions & can be readily removed • One auxiliary can give either enantiomer of product • Selectivity dependent on whether reagent can chelate two groups Advanced organic 4 Chiral reagents • Clearly, chiral reagents are preferable to chiral auxiliaries in that they function • • independent of the substrate’s chirality or on prochiral substrates A large number have been developed for the reduction of carbonyls Most involve the addition of a chiral element to one of our standard reagents Me H B + Me O Me H O B + RL Me Me 9-BBN•THF (+)-α-pinene RS Me alpine borane® proceeds via boatlike transition state can be reused Me H OH Me + RL B O RS Me Me H Me O alpine borane® (CH2)4Me small group as linear Me RS H OH (CH2)4Me 86% ee RL selectivity governed by 1,3-diaxial interactions Advanced organic 5 Chiral reagents II • Alpine-borane is very good for reactive carbonyls (aldehydes, conjugated ketones • • • etc) It is less efficient with other ketones so a more Lewis acidic variant has been developed Addition of the chloride makes the boron more electrophilic Transition state similar to previous slide Me H O BCl Me + Me H OH Me Me Me Me 2 (+)-Ipc2BCl 98% ee • And here is another... O Me B H Me + Me H OH Me Me Me Me Me >99% ee Advanced organic 6 Binol derivative of LiAlH4 1. (S)-BINAL–H 2. MOM–Cl O Me H OMOM Me SnBu3 O O SnBu3 93% ee OEt Al H Li (R)-BINAL–H • Reducing reagent based on BINOL and lithium aluminium hydride • Selectivity is thought toL arise from a 6-membered transition state (surprise!!) (R ) adopts the pseudo-equatorial position and the small • Largest substituent S substituent (R ) is axial to minimise 1,3-diaxial interactions O O Al O H RS Li Et RL O Advanced organic 7 Chiral reagents: allylation Ti Cl OH Ph Ph O H Ph Ph O O Me Me MgBr Ti OH Ph Ph O H O Ph Ph Ph H OH H O O Me Me 95% ee • Stereoselective reactions other than reduction can be performed with chiral reagents • In the above reaction the standard Grignard reagent is reacted with the titanium • complex to give the chiral allylating reagent Note: the chirality is derived from tartaric acid (again) Advanced organic 8 Chiral allyl boron reagents L RZ O + L R O RE B L B O RZ R L L L OH H2O2 NaOH B R R RZ RE RE RZ RE • Allyl boron reagents have been used extensively in the synthesis of homoallylic alcohols • Reaction always proceeds via coordination of Lewis basic carbonyl and Lewis acidic • • boron This activates carbonyl as it is more electrophilic and weakens B–C bond, making the reagent more nucleophilic Funnily enough, reaction proceeds by a 6-membered transition state H L R RE H B O RZ disfavoured H L H L vs RE R RZ B O H OH H L RE R RZ OH R RZ RE E-alkene gives anti product Z-alkene gives syn product • Aldehyde will place substituent in pseudo-equatorial position (1,3-diaxail strain) • Therefore alkene geometry controls the relative stereochemistry (like aldol rct) Advanced organic 9 Chiral allyl boron reagents II Me OH O B + Me Et Me Me H Et Me 92% ee 2 crotyl group orientated away from pinene methyl groups Me Me Me H H H Et Me H B Me Me O Et Me OH Me substituent pseudoequatorial • Reagent is synthesized from pinene in two steps • Gives excellent selectivity but can be hard to handle (make prior to reaction) • Remember pinene controls absolute configuration Geometry of alkene controls relative stereochemistry Advanced organic 10 Other boron reagents RZ Me RE B Me Me RZ O i-PrO2C RE O i-PrO2C 2 attacks on si face of RCHO B RZ Ts attacks on si face of RCHO tartaric acid derivative N Ph RE B N Ts Ph attacks on re face of RCHO • A number of alternative boron reagents have been developed for the synthesis of homoallylic alcohols • These either give improved enantiomeric excess, diastereoselectivity or ease of handling / practicality • Ultimately, chiral reagents are wasteful - they need at least one mole of reagent for • each mole of substrate End by looking at chiral catalysts Advanced organic 11 Catalytic enantioselective reduction OMe O CBS catalyst (10%) BH3•THF MeO H OH MeO MeO 93% ee • An efficient catalyst for the reduction of ketones is Corey-Bakshi-Shibata catalyst (CBS) • This catalyst brings a ketone and borane together in a chiral environment • The reagent is prepared from a proline derivative • The reaction utilises ~10% heterocycle and a stoichiometric amount of borane and • works most effectively if there is a big difference between each of the substituents on the ketone The mechanism is quite elegant... H N Ph Ph B O Me CBS catalyst proline derivative H BH3 Ph N Ph H3B B O Me active catalyst Advanced organic 12 Mechanism of CBS reduction • interaction of amine & borane activates borane • it positions the borane • it increases the Lewis acidity of the endo boron H catalyst turnover H Ph N BH3•THF Ph Ph N H3B B O Me B O Me Ph O H OH RL RS RL Ph coordination of aldehyde activates aldehyde and places it close to the borane Ph O Ph RS N B Me H B O H O Ph RS H N B Me H B O H RS H RL RL chair-like transition state largest substituent is pseudo-equatorial Advanced organic 13 Catalytic enantioselective nucleophilic addition O O H + C5H11 Zn C5H11 Me H OH (–)-DAIB (2%) O Me C5H11 >95% ee NMe2 OH Me (–)-DAIB Me Me Me Me Me C5H11 N Me O Zn C H 5 11 Zn C H 5 11 Me Me Me Me Me N C5H11 O Zn Ar vs. O Zn C5H11 C4H9 H Me Me Me C5H11 O Zn H O Zn N Me C5H11 C4H9 Ar disfavoured • There are now many different methods for catalytic enantioselective reactions • Here are just a few examples... • Many simple amino alcohols are known to catalyse the addition of dialkylzinc • • • reagents to aldehydes Mechanism is thought to be bifunctional - one zinc becomes the Lewis acidic centre and activates the aldehyde The second equivalent of the zinc reagent actually attacks the aldehyde Once again a 6-membered ring is involved and 1,3-diaxial interactions govern selectivity Advanced organic 14 Catalytic chiral Lewis acid mediated allylation O + Ph SnBu3 cat. (5%), DCM, rt, 7h H OH O Cl Ph H O N 88% 62% ee Rh N Cl Bn Bn cat. derived from amino acid (via the alcohol) • A variety of chiral Lewis acids can be used to activate the carbonyl group • These can result in fairly spectacular allylation reactions (higher ee than this example) • A problem frequently arises with crotylation • Often the reactions proceed with poor diastereoselectivity favouring either the syn • • or anti diastereoisomer regardless of geometry of the starting crotyl reagent This is because the reactions proceed via an open transition state (just to confuse you most actually favour syn! I use this example as the comparisons were easy to find...) O + Ph Me SnBu3 H E:Z 95 : 5 2 : 98 cat. (10%), DCM, rt, 72h O H OH Ph Me anti (ee) : syn (ee) 71 (57) : 29 (8) 63 (60) : 37 (7) R M H SnBu3 open transition state Advanced organic 15 Catalytic chiral Lewis base mediated allylation O + R RE Lewis base catalyst (LB) SiCl3 RE R H H H OH RZ RE R RZ LB Cl Si LB O Cl Cl RZ • An alternative strategy is the use of Lewis bases to activate the crotyl reagent • Reaction proceeds via the activation of the nucleophile to generate a hypervalent • • silicon species This species coordinates with the aldehyde, thus activating the aldehyde and allowing the reaction to proceed by a highly ordered closed transition state As a result good diastereoselectivities are observed and the geometry of nucleophile controls the relative stereochemistry Me H H N O P N N Me RE = Me 86% ee anti/syn 99/1 N O 5 () N P N H Ph Me N Ph Me H Me RZ = Me 95% ee syn/anti >19/1 H RE = Me 98% ee anti/syn >99/1 O RZ = Me 98% ee syn/anti 40/60 Me RE = Me 86% ee anti/syn 97/3 N N O O RZ = Me 84% ee syn/anti 99/1 Advanced organic 16 Organocatalysts O cat. (10%) NaOH Ph Et Br Et S Et S Ph Et Ph H Et O Et S Ph Ph O Ph Ph 92% ee 85% de (trans) • As we have seen with many stereoselective reactions, small organic molecules are • • • now finding considerable use as enantioselective catalysts Above is a simple example of epoxide formation The reaction proceeds by SN2 displacement of the bromide by the sulfide and subsequent deprotonation to give the reactive ylide Nucleophilic attack & cyclisation give the epoxide and regenerate the sulfide catalyst Advanced organic 17 Organocatalysts II H H NH HN N H N OTf N Ar HN Boc + R NO2 Ar cat. (10%) H P(O)Ph2 NO2 R 90% ee 90% de H Ar N Boc H O N O R • Acts as a ‘chiral proton’ • Protonation of the imine forms a highly electrophilic species • Aza-Henry reaction can then proceed to give the new amine Advanced organic 18 Bifunctional organocatalysts CF3 S F3C R NO2 N + Ar P(O)Ph2 H N H N H Me N Me P(O)Ph2 NO2 Ar cat. (10%) DCM, rt R 76% ee CF3 CF3 S S F3C HN N H O R N H N H N Me H Me O H F3C N N H H Me O O N N H Me NDpp R H Ar • Thio(urea) acts as a Lewis acid to activate & position the nitro substrate • Pendent amine functionality deprotonates the nitro α-C–H and presumably an electrostatic interaction shields the bottom face of the nitro enolate Advanced organic