* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Metabolism of Glucose C6H12O6+6O2 1 unit of Glucose 38 ATP
Point mutation wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Electron transport chain wikipedia , lookup
Mitochondrion wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Photosynthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
Phosphorylation wikipedia , lookup
Blood sugar level wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Butyric acid wikipedia , lookup
Microbial metabolism wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Biosynthesis wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
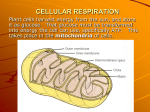
BIOCHEMISTRY – DR. WONG 2/19/2006 10:26:00 AM Metabolism of Glucose C6H12O6+6O2 1 unit of Glucose 38 ATP need 6 unit of O2 6 unit CO2 NAOH=3ATP FADH2=2ATP Net Chemical 1. Glycolysis 2. Intermediate 3. Kreb cycle We have many tiny steps to extract the energy, otherwise the 38 ATP would kill the cell. Recap of Glycolysis (9 steps): Glucose –you bring the glucose to the mitochondria, you have to provide ATP energy to the glucose to activate it (Glucose-6Phosphate. ‘6’ refers to the carbon atom). Phosphate is from ATP. We have to modify the structure into Fructose-6-Phosphate. Then into Fructose-1,6-diPhosphate Why do we have to change the glucose into Fructose? We need the second carbon to come off the ring to be available to react with the phosphate. (check recording on this) When they have enough energy, they split into two identical molecules*. into BPG Bisphosphoglycerate [DI for short, from DPG] BPG is so important because it has a special function, it is the one that gets the oxygen out of the hemoglobin. o Temperature o PH o CO2 level These all help BPG get O2 out of hemoglobin. Note: *Should be 2 identical reactions. *One 6C molecule turns into two 3C molecules. C6H12O6 o 3C molecule is small enough to pass through the outer membrane of the mitochondria, that’s why it splits. Pyruvate (3C) How much ATP can you extract from glycolysis only? 8 ATP Body lost 1 ATP from first step. Lost 1 more at the next step. -ATP -ATP +ATP +3ATP +ATP -2 ATP x210 ATP NET 8ATP We always chose 8 ATP as the answer to how much APT is gained from glycolysis. Pyruvate (3C) (Lack O2) Anaerobic NADH Aerobic (O2) 2 x CO2 C02 out Acetyl-CoA (2C) acetyl-coenzymeA This acetyl-CoA goes into the Kreb’s Cycle (citric acid cycle) This process enables the Glucose to pass through the inner membrane of the mitochondria. Matrix is called Matrix because it is very viscous/sticky. It’s filled with the intermediate chemicals and enzymes. At first two stages of the Kreb cycle, one CO2 is lost. It picks up 2C’s from the acetyl-c.A at 6C gives up one CO2 (and NADH) at 5C gives up CO2 (and NADH). It then gives up ATP, NADH, and FADH2. We need to know that the Kreb cycle is continuous until the mitochondria dies/stops. Also, the cycle will pick up 2 Carbon molecules from the acetylcoenzymeA. It gives up 2 carbon molecules each complete Kreb cycle. It gives up: o 3NADH 9 1 2 12 ATP o 1ATP o 1FADH2 “12 ATP” 1 Glucose 2x Kreb Cycle 2x 12ATP/cycle =2xATP To better understand this, look at the link Dr. Wong sent out on the e-mail. NOTES: Study where you see the Kreb cycle, the intermediate step. 2C can pass through the membrane? Pyruvate is intermediate step? Inside, outside of membrane? How many CO2 comes out per cycle? 2 Glycolysis Outer membrane Pyruvate Inner membrane 1C 2C Pyruvate 3C O2 Loss NADH (equivalent to ATP) Loss CO2 acetyl.cA O2 No O2 38 ATP “? ATP” 1 unit of Glucose in anaerobic pathway Gly 8 ATP. Lost 6 ATP. Body gains “2ATP” From Pyruvate to Lactate (lose 3ATP). Mental activity O2/anaerobic 38 Blood supply provides more O2 to skeletal muscle, giving more energy to you. 2ATP Stop So far, we’ve not seen oxygen really participate/get involved. Now we will see it in the Electron Transport System (ETS): the job of which is to convert e(-) acceptor into Energy Lipid Protein Glucose e(-) acceptors ETS/Oxidative Phosphorylation ½ O2 (one O atom) e(-) O2 molecules ETS is a chain reaction: O2+ NADH, FADH2 H20 Every electron needs 1 oxygen atom. The longer the distance the electron travels, the more energy that’s created. e.g. ABCDE, etc, this is a chain reaction. The line above is a chain reaction. How many electrons are generated during the metabolism of glucose? How many electron receptors are produced? 2. After the split they produce one NADH. x2=8 12 e(O2 Glycolysis 2 NADH Intermediate 2 NADH Kreb’s cycle 3 NADH 1 FADH2 4 e(-)acceptors ) acceptors 12 o-atoms 6 molecules e(-) NADHETS acetyl-c.A Free NADH lactate Note: Baroreceptors monitor how far the blood vessels are stretching. If you eat more, you have to break down more O2 atom. Not stable (not octet), it has to pick up e(-)/H+ from H2O to become stable. This leaves more free radicals in the body and causes chaos in the body. Eating more creates more free radicals. BIOCHEMISTRY – DR. WONG 2/19/2006 10:26:00 AM Nucleic Acids: I. DNA – deoxyribonucleic acid o Remove oxygen atom from ribose (a 5C sugar-carbohydrate) II. RNA – ribonucleic acid DNA RNA Double helix 3 different forms One-form MRNA (single strand) tRNA – anti-codon a.a. tRNA ribosomal ribosomes Located inside the nucleus Found anywhere inside the cell DNA to RNA transcription NUCLEUS to RER genetic materials Structure of nucleic acid: Polymer’s are combined monomers in a particular pattern Polymer (many) Monomer (single unit) Protein Amino acid Carbohydrate: Starch (less soluble) Glycogen (more soluble) Glucose Nucleic acid Nucleotide – 3 chemicals i) sugar - ribose (RNA) or deoxyribose (DNA) ii) phosphate – PO43iii) nitrogenous bases adenine A thymine T replaced by Uracil U guanine G cytosine C Double helix is governed by the Law of Complimentary Bases: If you have A on one side you must have T on the other side; etc. Pairing is based on two considerations: o Size of N-Bases o Orientation – hydrogen bonding (intermolecular attraction forces). No electron sharing; hence, not true bond. A & G are Double Ring Structures (“Purines”) OO T/U & C are Single Ring Structures (“pyridmidines”) O Pairs occur between O (pyridmidines) and OO (purines) BIOCHEMISTRY – DR. WONG BIOCHEMISTRY – WONG WEEK 9 – March 5th, 2006 Nucleic acid – Polymer i) P ii) Sugar iii) N- bases Repeating themselves in the same pattern DNA – take out 1 O atom from the ribose. Double helix More room to place the N-bases inside. One would think that DNA is less stable than RNA, but DNA is actually more stable. Law of Complimentary Bases Purines OO Pyrimidines O Adenine Thumine (Uracil) Guanine Cytosine This maintains the width of the DNA double helix. If you have a single ring on one side, there must be a double ring on the other side. Hydrogen Bonding - Intermolecular attraction forces which are weaker than ionic or covalent bond) H-------N H-------O a-a-a-a-a-a-a-a-a-a-a-a-a Hydrogen bonds between the alpha helix maintain strength of bond. Consider the size of structure when evaluating the strength of hydrogen bond. You aren’t responsible for this diagram, but in DNA this bond might look like this: N C C N C NH2 C N N C H Adenine Thymine LIPIDS I. Triglycerides (fat/oil) 1:3 ratio Glycerol + Fatty Acids (Carboxylic acids + at least 12 carbon long) C OH C OH C OH O HO-C-C-C-C-C-C-C-C-C-C-C-C O HO-C-C-C-C-C-C-C-C-C-C-C-C O HO-C-C-C-C-C-C-C-C-C-C-C-C You don’t have to put all the hydrogen’s on the molecules any more. However, put the bond marks on there. Condensation bond Glycolysis revisted: 1 glucose = “36 ATP” NAOH=3ATP FADH2=2ATP O22ATP Cristae is where the ETS is taking place. When Kreb cycle is producing NAOH/FADH2 in the matrix and during glycolysis. Glycolysis Outer membrane NADH Pyruvate NADH Inner membrane 1C 2C The longer the carbon chain you have, the more energy. Fatty acid could have 12c, 14c, 16c, 18c, 20c, 22c, 24c…. etc. even number. Always an II. Phospholipids - Major component of cell membrane 1glycerol:2fattyacid:1phosphate Glycerol: 2 F.A. and one of the F.A. is replaced by Phosphate. F.A. P PO43OH FA OH FA OH P Amphipathic compound- partially in water, partially in non-water molecule. Phospholipids are one of the few compounds that can do this. This special criteria is necessary for forming a cell membrane like ours. III. Wax - a long carbon chain. Alchohol + Fatty Acid (F.A.). Also form condensation – esther bond (they have smell). Water insulation is property of wax (for H20 insulation). 1:1 ratio IV. Lipid Vitamins- not all vitamins are fat soluble. Some are water soluble. H20 soluble: o Bo C Lipid soluble: o A - Retinol (for vision); body converts carotene to retinol because the body cannot eat Retinol directly (the liver breaks it down and prevents its absorption). o D - Calciferol o E – Trocopherol (anti-oxidant; captures free radicals) o K – this one has different forms. Sometimes it is given as an injection – affects blood clots. V. Steroids – OOOO four ring structure. 3 are hexagon, 1 is pentagon. Don’t worry about learning how to draw this. This is a broad class; including: cholestorol sex hormones o estrogen o testosterone o androgen aldostenone BIOCHEMISTRY – Dr. Wong 3/12/06 Three components of Nucleic Acid: Phosphate Sugar Nitrogenous Base Triglycerides (Fat/Oil): Glycerol + Fatty Acids (1:3) (Glycerine) Structural – manufactory of some other organic compound. Gluconeogenesis… Glycerol isn’t used for energy. The fatty acid IS used for energy. H+ (water soluble). Acids are water soluble. Carboxylic Acid is hydrophilic, atleast 12c or longer and of an even number. Name of Fatty Acid: 16:0 (The 16 means the number of C’s)(the 0 represents a saturated, single bond). O HO-C-C-C-C-C-C-C-C-C-C-C-C-C-C-C-C 1 2 3 4 5 6 7 8 9 (carbon molecules are numbered) When labeled with Greek letters, it skips the fx grp. Draw ALL the bonds Draw: 18:1∆15 15th (15) The 1 refers to one double bond -oxidation process O HO-C-C-C-C-C-C-C-C-C-C-C-C-C-C-C-C-C-C Remove bonds from the bottom. Don’t forget to put the functional group in here. Put triple bonds on last carbon If it is 18:3∆9,12,15, then the double bond is on the carbon’s between 9-10, 12-13, 15-16. When there is more than one double bond, it is called Polyunsaturated Fatty Acid (PUFA). In order of Best to Worse FA: Unsaturated Polyunsaturated (creates a lot of free radicals from the broken double bonds) Saturated 12:2∆7,10 Most Omega Fatty Acids are W3, W6, W9, W12, W15. Metabolism of Triglycerides (Fatty Acids) I. -Oxidation (cutting process) o Cutting occurs between and carbon. This is the oxidative degradation of saturated fatty acids in which two-carbon units are sequentially removed from the molecule with each turn of the cycle. II. Product acety-CoA This occurs within the Mitochondria. I) 16c/2 – 1 = (7)x5 (1 NADH, 1 FADH2) = 35 ATP II) 16/2 8 acety-CoA x 12 ATP = 96 ATP I + II: 131 ATP – 2 ATP (two subtracted for the energy required to push the fatty acid to the mitochondrial matrix) = 129. Biochemistry Lectures by Dr. Wong 3/19/06 Diabetes: Type I. Type II. o Insulin causes the blood glucose level to go down. Brings glucose into the cells for metabolism Convert glucose (monosaccharide) glycogen (polysaccharide). Compare and contrast different kinds of sugars: Starch vs. Glycogen Straight Chain Branched, globular H2O insoluble H2O soluble Body can convert glycogen to glucose. It can use: Glucagons Adrenaline/Epinephrine B-cells in the Pancreas are responsible for producing insulin. A-cells in the Pancreas are responsible for producing glucagons Adrenal (medulla portion) glands are responsible for producing epinephrine. Potentiation and synergism mean the same thing: A-1 B-1 A+B2>more. Drug-Drug interaction. Liver Glycogen Glucose Maintain blood sugar (glucose) level to provide enough glucose to the brain. Muscle Glycogen Glucose Provide fuel to generate energy. In exercise, first you burn off muscle glycogen, then liver glycogen, then lipid. If you only exercise In Diabetes, the cells cannot use glucose for energy. Only lipids. It depends on lipids, but the the Fatty Acids produce much more acetyl-CoA that kreb cycle cannot handle. This results in an accumulation of acetyl-CoA. The body does NOT like this. With too much acetyl-CoA, the body finds a way to get rid of it. It comes out in the urine and in the breath. Every 2 acetyl-CoA combines together forming a 4C molecule. This is called acetoacetic acid. There are two functional groups in this: Ketone, Carboxylic Acid. The Ketone is liver toxic. The Carboxylic acid is acidic. This can produce “acidosis”. Down-regulation = glucose receptors turn inward and ignore insulin. In the respiratory system, the acetoacetic acid is removed of O=C-O (CO2). It results in propanone (acetone). This causes the loss of NADH (“3ATP”)Acetone is the second component of the Ketone bodies. The ketone is converted to alcohol by placing a Hydrogen next to it. With the addition of another Hydrogen, it becomes octet. This is called -hydroxybutyric acid (butanoic acid). Butyric and Butanoic is the same thing. It is called because it’s on the second carbon molecule. Clinical testing of urine (with strip) measures the butanoic acid in the urine. Ketone Bodies: Acetoacetic Acid (found in blood and urine) Acetone (breath) -hydroxybutyric acid (urine) Neurotransmitter – GABA affects the CNS. It has an inhibitory effect on CNS. For patients with convulsions. These convulsions could be caused by: Not produce enough GABA GABA receptors are/have been damaged GABA stands for (gama)-amino-butyric-acid. Acetyl choline is an excitatory neurotransmitter in the CNS. FYI. TYPE I TYPE II Juvenile on-set Adult on-set IDDM – Insulin depedent diabetes mellitus NIDDM – Non-insulin dependant diabetes mellitus Insulin dependent Insulin independent Moderate genetic predisposition Strong genetic predisposition Prevalence 10-20% >80% Nutritional status undernourished Obesity Ketosis Type I. Common Type II. Rare Oral hypoglycemic Responsive Unresponsive Insulin level Low (damaged on bcells) Normal to high (down regulation on receptors) BIOCHEMISTRY Dr. Wong 3/26/06 Class of Chemicals: Glycosaminoglycans (GAGs) – combination of protein and carbohydrate together. Composed of: Glucosamine Glucouronic Acid – provide flexibility for the joints. Metabolism occurs in the liver for detoxification (to make the chemical more hydrophilic): o Phase I – Oxidation; cytochrome P450 o Phase II – Conjugation (coupling); glucuronic acid Phase II makes it hydrophilic. When drugs go through phaze II metabolism, the drug is considered inactive. Functions o For body ground substance Connective tissues Cartilage Ligament Bone matrix Examples made up of Glycosaminoglycans: Keratin (nails, cornea) Dermatan (skin; valves in circulatory system) Hyaluronic acid (shockabsorber) Heparin (anti-coagulating agent) Metabolism of Protein (a.a) Peptide bond Protein to individual a.a. Metabolism: I. Anabolism (build up) 2. Abdomen (break downs) -NH2 NH3 + CO2 Urea (hydrophilic) in urine. O H2N – C –NH2 Urea + H2O