* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Protein replacement therapies
Gene regulatory network wikipedia , lookup
Ancestral sequence reconstruction wikipedia , lookup
Silencer (genetics) wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Gene expression wikipedia , lookup
Magnesium transporter wikipedia , lookup
Protein (nutrient) wikipedia , lookup
List of types of proteins wikipedia , lookup
Protein folding wikipedia , lookup
Paracrine signalling wikipedia , lookup
Protein structure prediction wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Interactome wikipedia , lookup
Expression vector wikipedia , lookup
Protein moonlighting wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Western blot wikipedia , lookup
Protein purification wikipedia , lookup
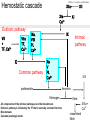
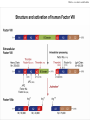
Manifestation of Novel Social Challenges of the European Union in the Teaching Material of Medical Biotechnology Master’s Programmes at the University of Pécs and at the University of Debrecen Identification number: TÁMOP-4.1.2-08/1/A-2009-0011 Manifestation of Novel Social Challenges of the European Union in the Teaching Material of Medical Biotechnology Master’s Programmes at the University of Pécs and at the University of Debrecen Identification number: TÁMOP-4.1.2-08/1/A-2009-0011 József Tőzsér Molecular Therapies- Lectures 5-6 PROTEIN REPLACEMENT THERAPIES TÁMOP-4.1.2-08/1/A-2009-0011 Protein-based therapies • Replacement of missing or abnormal proteins (e.g. Insulin) • Influencing signal transduction pathways (e.g. interferons) • Supplementation of growth hormones (e.g. PDGF) • Influencing the haemostatic system (e.g. tPA) • Degradation of molecules with enzymes (e.g. Asparaginase • Use of protein vaccines (e.g. recombinant HPV vaccine) TÁMOP-4.1.2-08/1/A-2009-0011 Examples of proteins replaced in therapy • • • • • • • Insulin (diabetes) Albumin (hypoalbuminaemia) Lactate (lactose intolerance) Factor VIII. (haemophilia) Factor IX. (haemophilia) Protein C (protein C deficiency) Beta-glucocerebrosidase (Gaucher’s disease) TÁMOP-4.1.2-08/1/A-2009-0011 Examples of protein therapeutic products • • • • • • • Humulin (insulin) Novolin (insulin) Flexbumin 25% (albumin) Lactaid (lactose) ReFacto (F VIII) BeneFix (F IX) Ceprotin (aktivált protein C) TÁMOP-4.1.2-08/1/A-2009-0011 Therapeutic proteins • The first therapeutic protein use was the utilization of cow pox „protein vaccine" against smallpox by Jenner (1796) • The first therapeutic protein used was insulin by Banting and Best (1922) • More than 200 peptide or protein has been approved in USA for use in therapy • Therapeutic proteins may come from different sources TÁMOP-4.1.2-08/1/A-2009-0011 Therapeutic proteins – sources Protein • • • • • • • Albumin Insulin Factor VIII Factor IX Kalcitonin Anti-venom β-glucorerebrosidase Original source Human blood Pig, bovine pancreas Human blood Human blood Salmon Horse, dunkey blood Human placenta TÁMOP-4.1.2-08/1/A-2009-0011 Therapeutic proteins – sources • Natural sourse is usually sparse and expensive • It is difficult to satisfy demands • Hard to isolate the product • May lead to immune intolerance (e.g. in case of animan proteins) • Potential viral and pathogen contaminations • Nowadays most of the protein drugs are made by recombinant techniques • Cheaper, safer, unlimited source TÁMOP-4.1.2-08/1/A-2009-0011 Insulin, the first protein replacement drug • In January 1922, Banting and Best used first insulin to treat a 14-years-old patient named Leonard Thomson • He became more ill as the consequence of the injection, but his blood glucose level decreased, therefore the improvement of the preparation technique was decided • 6 weeks later a better extract was able to decrease the blood glucose level from 520 mg/dL to 120 mg/dL within 24 hours. • Leonard lived for additional 13 years, he died of pneumonisa at the age 27 TÁMOP-4.1.2-08/1/A-2009-0011 Structure of insulin Two polypeptid chains, A.chain: 21 aminoacid residues B-chain: 30 aminoacid residues Chains are held together by a disulfide bridge Insulin gene is located at chromosome 11 TÁMOP-4.1.2-08/1/A-2009-0011 Protein therapeutics – from blood • The human body contains approx. 6 liters of blood • 60-70% of blood is plasma, 8-9%- proteins. Therefore it is an important protein source • Human plasma contains about 10,000 different proteins • About 20 proteins make up the 99% of the total protein content of plasma • Annually several million liters of outdated transfusion plasma is genereated, therefore it is an excellent and relatively abundant protein source TÁMOP-4.1.2-08/1/A-2009-0011 Examples of blood protein products • • • • • • Factor VIII (correction of haemophilia) Factor IX (correction of haemophilia) Albumin (correction of hypoalbuminaemia) Intravenous IgG (in infections) Antithrombin III (correction of coagulation disorder) Alpha I-PI (correction of emphysema) TÁMOP-4.1.2-08/1/A-2009-0011 Cohn fractionation • Cohn fractionation was originally developed in 1946. • In Cohn fractionation plasma proteins are selectively precipitated by using ethanol, salt, temperature change. • Separation of the fractions is achieved by centrifugation. TÁMOP-4.1.2-08/1/A-2009-0011 TÁMOP-4.1.2-08/1/A-2009-0011 Recombinant technologies • They were developed in the 1970s and 1980s • Paul Berg (1973): discovery of restriction enzymes • Herbert Boyer (1978): cloning of the human insulin gene into E. coli - Genentech • Fundamentally two major approaches – Expressin in isolated cells – Expression in transgenic plants or animals TÁMOP-4.1.2-08/1/A-2009-0011 Hemostatic cascade XIa Ca2+ Extrinsic pathway VII + TF, Ca2+ VIIa TF, Ca2+ Common pathway XI IX IXa, VIII PL Ca2+ X XII XIIa Intrinsic pathway Xa V PL Ca2+ prothrombin XIII thrombin fibrinogen All components of the intrinsic pathway are in the bloodstream. Extrinsic pathway is initiated by the TF that is normally excloded from the Bloodstream. Cascade (avalange) model. fibrin crosslinked XIIIa + Ca2+ TÁMOP-4.1.2-08/1/A-2009-0011 Haemophilias A and B • Hemophilias A and B are caused by deficiencies in factors VIII or IX, respectively • Affect ~1 in 5,000 males in Hemophilia A, ~1 in 30,000 males in Hemophilia B • Inherited as a recessive X-linked trait in both cases (Mother would be an unaffected carrier) • Treated by administration of factor VIII or factor IX concentrates • Recombinant factor VIII or IX • Gene therapy trials TÁMOP-4.1.2-08/1/A-2009-0011 TÁMOP-4.1.2-08/1/A-2009-0011 TÁMOP-4.1.2-08/1/A-2009-0011 Haemophilia A – gene therapy • Only FVIII level is 1 % of normal causes severe symptoms (spontaneous bleeding into joints, vital organs), therefore even low levels of proteins are beneficial • Tight control of FVIII production is not required • Broad therapeutic index minimizes the risk of overdose • Delivery to bloodstream does not require expression in the liver • Domain B is not required for function, in its absence the expression levels are higher TÁMOP-4.1.2-08/1/A-2009-0011