* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Working in the third dimension - biomed
Cell membrane wikipedia , lookup
Signal transduction wikipedia , lookup
Endomembrane system wikipedia , lookup
Programmed cell death wikipedia , lookup
Cell growth wikipedia , lookup
Cytokinesis wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cellular differentiation wikipedia , lookup
Tissue engineering wikipedia , lookup
Cell culture wikipedia , lookup
List of types of proteins wikipedia , lookup
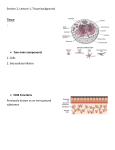
10 wissenschaft & praxis Working in the third dimension The application of 3D cell culture systems in life science In the 19th century Wilhelm Roux and Ross Granville Harrison independently published their first results of culturing tissue explants in warm saline solution (Ringer´s solution) for several days. They have extracted small pieces of tissue and maintained it for some days in the physiological salt solution and thereby introduced the first tissue culture technique to the scientific community. Nowadays this culture technique is still used but it is called explant culture. In the 1940s and 1950s the main application of cell culture techniques was using isolated cells for virus infection in a standardized environment to improve the knowledge of virus infection and to use the purified viruses for the production of vaccines. Thereby the aim of the scientists was to isolate special cell types from tissues and to culture them in standardized flasks with defined media as monolayer cultures, the typical way of two dimensional cell culture systems. In a parallel manner the techniques to investigate cells and tissues and to study tissue composition and morphogenesis developed further. Thereby, the importance of the microenvironment of the cells and the role of the interaction of the different cell types in tissues was investigated and scientists started the next step in cell culture techniques by introducing different matrices (coatings of the cell culture flasks or petri dishes) like collagens, laminin, and fibronectin to offer the cells a more physiological environment in the cell culture systems. The interaction of the cells with these extracellular matrix (ECM) components was demonstrated in their changed morphology and cellular behaviour including alterations in drug response and genetic information. The development of a huge number of different techniques to culture cells in a more physiological environment than two dimensional (2D) flat bottom plastic or glass dishes mirrors their potential to work as a link between cell culture systems and living tissues/organisms in all fields of science. Different techniques of 3D cell culture Organotypic explant cultures represent a basic technique to obtain cultured cells in their natural environment and are available as a source for primary isolated cells. An organotypic explant culture is composed of a thin slice of tissue cultured on a metal grid and/or an air-liquid interface in growth or differentiation medium. In a specific environment these primary isolated cells can grow out of this system. These cells can be used as primary cells for secondary reconstruction of organotypic cell culture systems. The cells reestablish cell-cell and cell-extracellular matrix (ECM) contacts to form aggregates (spheroids) of several hundreds of micrometers featuring tissue-specific characteristics. Different techniques to form these cellular spheroids were established during the last decades but the main components are gravity and accumulation (hanging drops, spinner flasks, gyratory rotation systems). The establishment of multicellular spheroids, consisting of different cell types, introduced co-culture techniques into spheroidal formation techniques (Figure 1). Figure 1.Formation of a multicellular spheroid. Cell suspension of stromal cells (green) is cultured in a hanging drop (A) to form a spheroid (B). This stromal spheroid is placed in a well and then an epithelial cell suspension is carefully overlayed (D). This technique results in a multicellular spheroid with a stromal core (green) surrounded by a differentiated surface epithelium (pink). These multicellular spheroids produce their own ECM but during formation the addition of laminin or collagens to the culture medium can improve the quality of the spheroids. Several approaches have been designed to reach well differentiated polarized epithelial cells in 3D cell culture systems and different techniques were established to mimic the natural micro-environment of the epithelial cells and epithelial structures. To study epithelial cells with their surface-covering characteristics polarized epithelial cell culture systems were established to provide an air-medium interface for apical polarization and an underlying porous substrate/membrane coated with specific ECM components for apical and baso-lateral polarization. In co-culture systems, supporting cell types can be placed below or in the epithelial-ECM system to enhance the mimicry of the natural microenvironment by providing direct or indirect (due to the pores in the membrane) cell-cell communication (Figure 2). Positive effects concerning polarization, differentiation and physiological behavior of cultured epithelial cells were demonstrated by adding conditioned medium gained from cell cultures of other cell types which underlay and support the epithelium in vivo. Figure 2.A co-culture system to demonstrate epithelial-stromal crosstalk. wissenschaft & praxis On the bottom of the well a collagen type IV matrix allows a coculture of smooth muscle cells and stromal cells. The membrane of the insert is coated with laminin, an important component of the basement membrane that enables epithelial polarization. The differentiated, polarized epithelial cells on the membrane can interact with the medium of the underlying co-culture of smooth muscle cells and stromal cells due to pores in the membrane. Thereby, communication of the different cell types is ensured in an indirect manner (purple arrows). This co-culture model mimics the interaction of endometrial epithelial surface cells and the cells of the underlying stroma and the myometrium. All of the cell types are responsive for stimulation with steroid hormones and their interaction influences steroid hormone receptor expression in the respective cell types. Epithelial cells grown in specific media have the potential to form spheroids with luminal formation due to apoptosis resembling e.g. cysts or glandular structures (Figure 3). The differentiation of epithelial cell layers into different types of epidermal cells (keratinocytes) is the key to receive artificial skin constructs supported by different types of partial ECM-coated scaffolds and co-cultured fibroblasts. Figure 3.Formation of a glandular structure with multicellular spheroid technique. Cell suspension of epithelial cells (red) is cultured in a hanging drop (A) to form a spheroid (B). This epithelial spheroid consists of polarized epithelial cells. During culturing time a lumen in the core of the spheroid develops due to apoptosis. The spheroid is placed in a well (C) and then a stromal cell suspension is carefully overlayed (D). This technique results in a multicellular spheroid with an epithelial core (red) with a lumen, surrounded by stromal cells (green). These multicellular spheroids can be used to study glandular morphogenesis and the interaction of glandular epithelial and stromal cells. For expanding primary cells or as vehicle for adhesive cells during sensitive procedures including cell-based assays, cryopreservation and electroporation, microcarriers were developed. The surface of a microcarrier (small sphere, Ø < 500µm) reaches up to 500 cm²/g due to the curved surface providing small niches for cells to adhere. In these niches cells produce their own ECM or the microcarrier is coated with different proteins and/or other ECM components to supply cell adhesion and growth. The extracellular matrix (ECM) In living organisms most of the cells are embedded and surrounded by the ECM, their own three dimensional microenvironment with a highly organized, complex and dynamic molecular composition. The ECM is sensed and organized by cells through binding events between proteins on the cell surface and ECM proteins. Cells dynamically reorganize their microenvironment due to signaling mole- cules activating ECM-cleaving proteins (e.g. metalloproteinases), and modifying the composition of ECM, both regulated by integrin-mediated signaling pathways. These remodeling processes are necessary for cell migration and proper tissue homeostasis and have a pronounced role during pathological and developing states. The highly organized meshwork of the ECM also serves as three dimensional substructures for cell adhesion and movement, and offers storage for growth factors, chemokines and cytokines, important factors for tissue morphogenesis. This highly dynamic biological network cannot be reconstituted by any synthetic model in vitro. Therefore, nearby using simple single ECM component gels (laminin, collagen), two different opportunities for working with a complex3D ECM in cell culture systems are practiced: on the one hand the natural-derived ECM of Engelbreth-Holm-Swarm mouse sarcoma (e.g. MatrigelTM) and on the other hand synthetic peptide hydrogels with certain, synthetically produced, defined ECM components. Figure 4. A complex 3D co-culture system to demonstrate epithelialstromal crosstalk with the surface and the glandular epithelium. On the bottom of the well a collagen type IV matrix allows to culture smooth muscle cells in their natural environment. In the insert a scaffold of ECM components is placed to provide an ECM meshwork for glandular spheroids and surrounding stromal cells. On the top of the scaffold a differentiated layer of polarized epithelial cells mimics the surface epithelium. This model can be used to study the interaction of the surface and glandular epithelial cells with the stroma and the smooth muscle cells (purple arrows). The application of a natural-derived ECM like MatrigelTM and its role on cellular activity in 3D cell culture systems has to be considered carefully. The composition and detailed amounts of ingredients of this artificial ECM are not disclosed by the manufacturer probably because of the high variability of formula depending on the extracted ECM from various Engelbreth-Holm-Swarm mouse sarcomas. Information for the percentage of laminin, collagen IV and entactin is available as well as for a selection of growth factors. Since the early 1980s different studies elucidated the main components and their effects on cell culture systems. To overcome the problems with undefined natural-derived, and therefore heterogenic in composition, artificial ECMs, the application of purified basement membrane components in a synthetic ECM (e.g. BD PuraMatrixTM Hydrogel) overcomes this problem but interactions of different factors and ECM components may be lost. Therefore, a study was carried out with an in-depth proteomic analysis of MatrigelTM demonstrating the high complexity of ECM composition due to the identification of 1302 different proteins in the 11 12 wissenschaft & praxis standard MatrigelTM and 1246 proteins in the growth factor reduced MatrigelTM. Based on these results, the replacement of MatrigelTM in different cell culture systems and experimental assays will be a time-consuming and expensive issue and due to the fact that the roles of several proteins are still unclear it will be very elaborate to find appropriate alternative compositions. However, the composition of the ECM in a 3D cell culture system depends on the cell/tissue type and has to be adjusted to the different cell types to provide the best microenvironment for the specific cells/tissues. Tissue architecture and cell polarity A further challenge nearby the complexity of ECM composition is the correct cellular polarity to mimic natural tissue architecture in vitro. Physiological cell polarity has to be considered especially in epithelial cell culture systems because the entire organization of an epithelial cell is polarized with an asymmetrical distribution of organelles and proteins including transmembrane receptors and other factors within the cell important for tissue differentiation and function. Development of polarized epithelial tissues depends on dynamic interactions of the genome and the microenvironment (ECM) resulting in specific alterations of gene expression patterns of single epithelial cells. For the assembly of individual cells into 3D tissue structures cell adhesion per se is crucial in both, the living organism and in vitro systems. Various types of adhesive molecules are involved in the development and maintaining of epithelial structure and function. Furthermore, proper tissue morphogenesis depends on close interactions of physical cell adhesion events and intercellular signaling, both related to physiological cell polarity. Bissell demonstrated in her innovative work concerning 3D cell culture systems of human breast cancer cells that formation of acinuslike structures in vitro depends on (a) ECM composition (including cell-cell-contacts) and (b) proper polarity of the epithelial cells. These factors have profound effects on cell behavior and gene expression patterns and have to be considered carefully for the choice of the applied 3D cell culture system. Gene expression and drug screening assays In pioneering experiments Bissell and collaborators demonstrated that the assembly of the different laminin components of the basement membrane has a crucial role in induction of apical polarity in epithelial cells because nearby other factors even the stiffness of the surrounding ECM influences the formation of cells and cell behavior. Thereby it was demonstrated that the ECM affects the chromatin structure, and the respective gene expression patterns in vivo and in vitro. This revolutionary discovery represents the fact that cells cultured in 2D systems have clearly different gene expression patterns than the same cells cultured in 3D cell culture systems. This insight opened a new challenge for basic research in all scientific fields in cell culture models because results achieved with 2D-cultured cells cannot be compared to results from 3D models. In different 3D cancer cell culture models added drugs or supplemented hormones induced effects never observed in the respective 2D models. Furthermore, the important role of tumor stromal cells was introduced by using co-culture systems that produced divergent results compared to single cell cultures. Organs on a chip An advanced application of 3D cell culture systems is demonstrated in microfabrication technologies and microfluidics approaches to create 3D cell-culture microenvironments allowing the simulation of tissue-tissue interactions on a chip comparable to a USB stick in size. These microengineered models have the potential to work as models for systemic diseases, to improve the knowledge of physiological processes and to replace animal testing in research. The application of 3D cell culture systems in research has profoundly improved the knowledge of cell-cell and cell-ECM communication and their interdependence in morphogenesis, differentiation and development as well as cellular response to different stimuli. Nevertheless, the high complexity of the 3D cell culture systems has to be considered in every study design and in the comparison of the outcome of different studies, bearing in mind that even a minimal change in the ECM may lead to changes in the cellular response, morphology and behavior. n Imaging methods for 3D cell culture The step from 2D to 3D cell culture systems implicated the development and new application of imaging techniques to facilitate life cell imaging even in the third dimension. Microscopy techniques that were established for thin, optically transparent cultures are poorly suited for imaging 3D cell cultures because of their thickness and scattering characteristics leading to optical distortion. Confocal microscopy is the best choice to monitor 3D cell culture systems routinely but it poorly defines along the z-axis. Furthermore, the penetration depth is limited compared to two-photon or multi-photon microscopy, whereas the high power laser required for this technique might harm the cells. Therefore, to monitor the dynamic changes to cells in 3D microenvironments non- invasive techniques like optical projection tomography as well as light sheet microscopy can be used and offer new insights in cellular behaviour and tissue rearrangement in vitro. Maga Cordula Bartel Veterinärmedizinische Universität Wien Institut für Anatomie, Histologie und Embryologie Cordula Bartel arbeitet seit 2009 an der Etablierung eines dreidimensionalen Zellkultursystems der caninen Gebärmutterschleimhaut zur Untersuchung der Veränderungen der verschiedenen involvierten endometrialen Zelltypen durch den Einsatz von ovariellen Steroidhormonen. Dieses Modell dient sowohl zur Grundlagenforschung der Reaktionen der Zellen auf unterschiedliche Steroidhormoneinflüsse als auch der Untersuchung pathobiologischer Vorgänge und ermöglicht die Reduktion von Tierversuchen in diesem Forschungsgebiet. wissenschaft & praxis References and recommended literature: Baker M: Tissue models: a living system on a chip. Nature 2011, 471(7340):661-665. Birgersdotter A, Sandberg R, Ernberg I: Gene expression perturbation --a growing case for three-dimensional (3D) culture systems. Seminars in cancer biology 2005, 15(5):405-412. Bissell MJ, Barcellos-Hoff MH: The influence of extracellular matrix on gene expression: is structure the message? J Cell SciSuppl 1987, 8:327-343. Hall HG, Bissell MJ: Characterization of Murine Mammary Cytoskeletal Elements - Response to Collagen Substratum. Journal of Cell Biology 1983, 97(5):A316-A316. Inman JL, Bissell MJ: Apical polarity in three-dimensional culture systems: where to now? J Biol 2010, 9(1):2. Kenny PA, Lee GY, Myers CA, Neve RM, Semeiks JR, Spellman PT, Lorenz K, Lee EH, Barcellos-Hoff MH, Petersen OW et al: The morphologies of breast cancer cell lines in three-dimensional assays correlate with their profiles of gene expression. MolOncol 2007, 1 %(1):84-96. Kim JB: Three-dimensional tissue culture models in cancer biology. Seminars in cancer biology 2005, 15(5):365-377. Lee GY, Kenny PA, Lee EH, Bissell MJ: Three-dimensional culture models of normal and malignant breast epithelial cells. Nature methods 2007, 4(4):359-365. Page H, Flood P, Reynaud EG: Three-dimensional tissue cultures: current trends and beyond. Cell Tissue Res 2012. Pampaloni F, Reynaud EG, Stelzer EH: The third dimension bridges the gap between cell culture and live tissue. Nat Rev Mol Cell Biol 2007, 8(10):839-845. Spencer VA, Xu R, Bissell MJ: Gene expression in the third dimension: the ECM-nucleus connection. J Mammary Gland BiolNeoplasia 2010, 15(1):65-71. Tibbitt MW, Anseth KS: Hydrogels as extracellular matrix mimics for 3D cell culture. BiotechnolBioeng 2009, 103(4):655-663. Williamson A, Singh S, Fernekorn U, Schober A: The future of the patient-specific Body-on-a-chip. Lab on a chip 2013. Xu R, Boudreau A, Bissell MJ: Tissue architecture and function: dynamic reciprocity via extra- and intra-cellular matrices. Cancer Metastasis Rev 2009, 28(1-2):167-176. Yamauchi N, Yamada O, Takahashi T, Imai K, Sato T, Ito A, Hashizume K: A three-dimensional cell culture model for bovine endometrium: regeneration of a multicellular spheroid using ascorbate. Placenta 2003, 24(2-3):258-269. Zhu HY, Wang JX, Tong XM, Xu WH, Jiang LY, Jing XY, Yang LY, Zhou F, Zhang SY: Three-dimensional cultures of human endometrial cells on Matrigel mimic in vivo morphology. Chinese medical journal 2012, 125(5):863-868. Fragen beantworten und CPD-Punkte sammeln Ab sofort erscheint in jeder Ausgabe der Fachzeitschrift „biomed austria“ ein Fachartikel, dem vier Fragen angeschlossen sind. Die (hoffentlich richtigen) Antworten auf diese Fragen können ausschließlich elektronisch über ein Formular auf unserer Homepage übermittelt werden. Sind drei der vier Fragen richtig beantwortet, erhalten Sie 1 CPD-Punkt bzw. 2 CPD-Punkte, wenn es sich wie diesmal um einen englischsprachigen Artikel handelt. Am Ende des Jahres wird ein Zertifikat über die erfolgreiche Teilnahme am Literatur studium mit der entsprechenden Anzahl an CPD-Punkten ausgestellt. Es können pro Jahr fünf CPD-Punkte mittels Literaturstudium bei biomed austria erworben werden – pro CPD-Periode sind das 15 CPD-Punkte, für die Sie keine Kosten aufwenden müssen. Dieses Service gibt es ausschließlich für Mitglieder von biomed austria. Die elektronische Eingabe der Antworten unter: www.biomed-austria.at/fortbildung Bitte beachten Sie, dass bis zu vier Antworten richtig sein können! 1. What does the term “conditioned medium” stand for? a. A cell culture medium with defined composition to work in standardized conditions. b. Cell culture medium collected from one cell culture to s upply cells in another cell culture system with soluble factors g ained from the other cell type. c. Conditioned medium is collected from specific cell types to provide a more natural environment for other cell types without direct cell-cell contact of the two different cell types. d. A possibility to work with a co-culture system in an indirect way. 2. Cellular characteristics can be changed in 3D cell culture systems due to the application ofdifferent extracellular matrices. Which characteristics can be changed? a. Morphology b. Genetic information c. Drug response d. Migration 3. What are the features of a microcarrier as an improved cell culture technique? a. Cells can not adhere to the smooth surface of a microcarrier and thereby suspension cell culture techniques are improved. b. Microcarriers can be used as vehicles to expand cell cultures. c. In the niches of the microcarriers cells can produce their own extracellular matrix. d. Microcarrier can be coated with different ECM proteins and thereby cell adherence is improved. 4. Why can the extracellular matrix (ECM) in a 3D cell culture system be crucial for tissue specific cell differentiation? a. The ECM components improve cell adhesion and thereby support specific cell differentiation. b. In epithelial cells polarization is crucial for correct tissue architecture and thereby the interactions of the ECM components with the cells as well as cell-cell-contacts in 3D cell culture systems induce cell polarization comparable with the in vivo situation. c. Because cellular differentiation depends on dynamic interactions of the genome and the cellular microenvironment (ECM). d. Information of the defined composition of the ECM used in a cell culture system is always provided by the manufacturer and thereby, cellular differentiation can be induced by using the respective ECM. 13