* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 12 - Membrane Transport
Model lipid bilayer wikipedia , lookup
Cell encapsulation wikipedia , lookup
SNARE (protein) wikipedia , lookup
P-type ATPase wikipedia , lookup
Cytoplasmic streaming wikipedia , lookup
Magnesium transporter wikipedia , lookup
Membrane potential wikipedia , lookup
Cytokinesis wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Signal transduction wikipedia , lookup
Cell membrane wikipedia , lookup
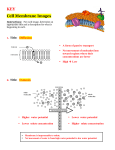
Lec-4 Membrane Transport 2 Lecturer: Dr. Twana A. Mustafa OSMOSIS OSMOSIS Definition: The diffusion of water down its concentration gradient (that is, an area of higher water concentration to an area of lower water concentration) thru a semi-permeable membrane is called Osmosis. Concept: Because solutions are always referred to in terms of concentration of solute, water moves by osmosis to the area of higher solute concentration. Despite the impression that the solutes are “pulling,” or attracting, water, osmosis is nothing more than diffusion of water down its own concentration gradient across the membrane. Osmotic pressure: is the pressure that is required to stop osmosis. It is the pressure necessary to prevent osmosis into a given solution when the solution is separated from the pure solvent by a semipermeable membrane. The greater the solute conc. of a solution, the greater its osmotic pressure. (HYDROSTATIC PRESSURE = OSMOTIC PRESSURE) An osmole is one mole of dissolved particles in a solution. E.g. glucose when dissolved in solution does not dissociate, so 1 mole of glucose is also 1 osmole of glucose. On the other hand, NaCl dissociates into 2 ions (Na and Cl) so is taken as 2 moles. Osmolarity is the number of osmoles of solute per liter of solution. Simply put, osmolarity is a measure of total solute conc. given in terms of number of particles of the solute in 1 liter of solution. The osmolarity of body fluids is usually expressed in milliosmoles per liter (mOsm/L). (The normal osmolarity of body fluid is 300 mOsm.) It is usually employed in clinical settings. Osmolality is the number of milliosmoles of solute per kg of solvent. It is usually calculated in laboratories using an osmometer. • Hyper = above • Iso = same • Hypo = below tonic refers to the shape of the cell Isotonic Solution NO NET MOVEMENT OF H2O (equal amounts entering & leaving) Hypotonic Solution CYTOLYSIS copyright cmassengale Hypertonic Solution PLASMOLYSIS 8 Active transport • Primary active transport:the transporter itself is an ATPase that cause the breakdown of ATP and phosphorylate itself. Therefore, change the affinity of the transporters solute binding site. • Secondary active transport:use of an ion concentration gradient across a membrane as the energy source. The flow of the ion provides energy for the uphill movement of the actively transported solutes. Na+/K+ Pump 10 Ca-ATPase • In the plasma membrane, the active transport of Ca is from cytosol into extracellular fluid. • In the organelle membranes, the active transport of Ca is from cytosol into organelle. • The extracellular [Ca] is 1 mM, while the intracellular [Ca] is 0.1 uM. H+ Pumps Diverse examples of carrier-mediated transport. Figure 4-15 3.2 Secondary Active Transport Coupled transport. Energy needed for “uphill” movement obtained from “downhill” transport of Na+. Hydrolysis of ATP by Na+/K+ pump required indirectly to maintain [Na+] gradient. 15 Figure 4-13 Secondary active transport uses the energy in an ion gradient to move a second solute. Figure 4-14 Cotransport: the ion and the second solute cross the membrane in the same direction. Countertransport: the ion and the second solute move in opposite directions. Secondary active transport co-transport (symport) out in Na+ glucose Co-transporters will move one moiety, e.g. glucose, in the same direction as the Na+. counter-transport (antiport) out in Na+ H+ Counter-transporters will move one moiety, e.g. H+, in the opposite direction to the Na+. 2 Methods of Glucose Transport • 2 mechanisms are separate – Passive transport at the basal surface – Active transport at the apical surface • Caused by the tight junctions Na+-Driven Transport • Na+ driven symport – Used to move other sugars and amino acids • Na+ driven antiport – Also very important in cells – Na+-H+ exchanger is used to move Na+ into the cell and then moves the H+ out of the cell • Regulates the pH of the cytosol Figure 4-22 Figure 4-24 4. Bulk Transport (Endocytosis and Excytosis) Movement of many large molecules, that cannot be transported by carriers. Exocytosis: A process in which some large particles move from inside to outside of the cell by a specialized function of the cell membrane Endocytosis: Exocytosis in reverse. Specific molecules can be taken into the cell because of the interaction of the molecule and protein receptor. 24 Exocytosis Vesicle containing the secretory protein fuses with plasma membrane, to remove contents from cell. 25 Endocytosis Material enters the cell through the plasma membrane within vesicles. 26 Types of Endocytosis Phagocytosis - (“cellular eating”) cell engulfs a particle and packages it with a food vacuole. Pinocytosis – (“cellular drinking”) cell gulps droplets of fluid by forming tiny vesicles. (unspecific) Receptor-Mediated – binding of external molecules to specific receptor proteins in the plasma membrane. (specific) 27 Example of Receptor-Mediated Endocytosis in human cells 28 Figure 4-21 Alternative functions of endocytosis: 1. Transcellular transport 2. Endosomal processing 3. Recycling the membrane 4. Destroying engulfed materials