* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Presentation
Vectors in gene therapy wikipedia , lookup
Gene expression wikipedia , lookup
Silencer (genetics) wikipedia , lookup
Biochemical cascade wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Endogenous retrovirus wikipedia , lookup
Paracrine signalling wikipedia , lookup
Metalloprotein wikipedia , lookup
Gene therapy of the human retina wikipedia , lookup
Secreted frizzled-related protein 1 wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Ligand binding assay wikipedia , lookup
Signal transduction wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Expression vector wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
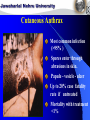
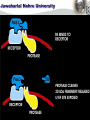
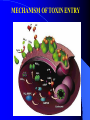
Centre for Biotechnology Jawaharlal Nehru University (JNU), New Delhi RECOMBINANT VACCINE AGAINST ANTHRAX ANTHRAX Geographical Distribution of Anthrax Epidemic Endemic Sporadic Probably Free Free Unknown Cutaneous Anthrax Most common infection (>95% ) Spores enter through abrasions in skin. Papule - vesicle - ulcer Up to 20% case fatality rate if untreated Mortality with treatment <1% GASTROENTESTINAL ANTHRAX Rare form of infection. Ingestion of insufficiently cooked, contaminated meat. Abdominal pain and fever. Fatal bacterium and toxemia then ensue. Mortality exceeds 50% if untreated. INHALATION ANTHRAX Inhaled spores phagocytosed by macrophages transported To regional lymphnodes. Spores germination followed by toxin release. Extensive necrotic haemorrhage. Death from sepsis and shock. Bacillus anthracis as a Biowarfare Agent Possible vehicle of mass death Weapon of mass destruction (WMD) Destructive capability of weaponized anthrax is equivalent to that of a nuclear bomb (Wein et.al. 2003) Poor Diagnosis [Webb et.al.] VACCINES AGAINST ANTHRAX TILL DATE, VACCINE BASED ON LIVE STERNE’S STRAIN IS THE MOST POPULAR VETERINARY VACCINE AGAINST ANTHRAX WORLDWIDE . RUSSIA USES LIVE SPORE VACCINE FOR HUMANS IN UK CURRENTLY AVAILABLE HUMAN VACCINE CONSISTS OF ALUM PRECIPITATED CELL FREE FILTRATE OF STERNE STRAIN. IN US THE VACCINE IS ALUMINIUM HYDROXIDE ADSORBED CELL FREE FILTRATE OF A NON-CAPSULATING STRAIN OF B. anthracis. HOWEVER, CURRENTLY AVAILABLE VACCINES HAVE CERTAIN DEGREE (5-10%) OF RESIDUAL VIRULENCE AS THE BACTERIUM PRODUCES BOTH LF AND EF COMPONENTS. Anthrax Vaccine Side Effects • • • • • • • Soreness, redness at the site of shot given Headache Muscle ache Fatigue Nausea Chills and Fever Allergic reactions Need for development of improved anthrax vaccine devoid of side effects ANTHRAX : Primarily a disease of animals, humans are accidental host. Bacillus anthracis pXO1 *pXO1:181 kb. Codes for Protective Antigen (PA), Lethal Factor (LF) & Edema Factor (EF). pXO2 VIRULENCE DETERMINANTS *pXO2: 96 kb. Codes for Poly D-Glutamic acid capsule. Bacillus anthracis (under microscope) Extra *chromosomal genetic material : plasmid . Anthrax spores are highly stable under adverse conditions. PA: MAIN IMMUNOGEN, PROVIDES PROTECTIVE IMMUNITY AGAINST ANTHRAX. MAIN COMPONENT OF ALL ANTHRAX VACCINES ANTHRAX TOXIN COMPONENTS Edema Factor (EF) Mw 89 kDa Adenylate cyclase Protective Antigen (PA) Mw 83 kDa Cell binding moiety Edema in Skin (Rabbits, Guinea Pigs) Increased Cyclic AMP Lethal Factor (LF) Mw 90 kDa Zn Metalloprotease Lethality (Rats, Mice, etc.) Macrophage Lysis MECHANISM OF TOXIN ENTRY THE PA 83 MONOMER DOMAIN 1: (RESIDUES 1-258) CONTAINS FURIN CLEAVAGE SITE WHICH DEFINES TWO SUB-DOMAINS: PA 20 FRAGMENT (RESIDUES 1-167) AND DOMAIN 1’ (RESIDUES 168-258). DOMAIN 2: (RESIDUES 259-487) PLAYS A ROLE IN MEMBRANE INSERTIONAND TRANSLOCATION. DOMAIN 3: (RESIDUES488-595) PLAYS A ROLE IN OLIGOMERISATION. DOMAIN 4: (RESIDUES 596-735) RECEPTOR BINDING DOMAIN. THE PA63 HEPTAMER • LOSS OF PA20 LEADS TO HEPTAMER FORMATION BY PA63. HEPTAMER IS WATER SOLUBLE AT NEUTRAL OR BASIC pH. HEPTAMER INSERTS INTO MEMBRANE AT ACIDIC pH FORMING CATION- SELECTIVE CHANNELS IN BOTH ARTIFICIAL LIPID BILAYERS AND CELLS. LETHAL FACTOR DOMAIN I : INVOLVED IN PA BINDING DOMAIN II : RESEMBLES ADP RIBOSYLATING TOXIN OF B. cereus, AUGMENTS SUBSTRATE RECOGNITION DOMAIN III : ALONGWITH DOMAIN 2 AND 4 HELPS IN HOLDING THE 16 RESIDUE LONG N-TERMINAL TAIL OF MAPKK BEFORE CLEAVAGE. POSSIBLY INVOLVED IN MEMBRANE INSERTION. DOMAIN IV : CATALYTIC SITE Zn CONTAINING CLONING, EXPRESSION AND PURIFICATION OF PA, LF AND EF FROM E. coli: CLONING IN EXPRESSION VECTORS PCR pExp pXO1 184kb PCR Amplified Gene References: 1. Gupta P, Waheed SM, Bhatnagar R. (1999) Expression and purification of the recombinant protective antigen of Bacillus anthracis. Protein Expr Purif. Aug;16 : 369-76. 2. Chauhan V, Singh A, Waheed SM, Singh S, Bhatnagar R. (2001) Constitutive expression of protective antigen gene of Bacillus anthracis in Escherichia coli. Biochem Biophys Res Commun. May 4;283 : 308-15 3. Gupta P, Batra S, Chopra AP, Singh Y, Bhatnagar R. (1998) Expression and purification of the recombinant lethal factor of Bacillus anthracis. Infect Immun. Feb;66 : 862-5. 4. Kumar P, Ahuja N, Bhatnagar R. (2001)Purification of anthrax edema factor from Escherichia coli and identification of residues required for binding to anthrax protective antigen. Infect Immun. Oct; 69 : 6532-6. LOCALIZATION OF E.coli EXPRESSED PA 220 kDa 97 kDa 66 kDa 46 kDa 30 kDa 21.5 kDa 14.3 kDa PURIFICATON OF PA 220kDa 116kDa 97kDa 66kDa 45kDa Purification of PA from E.coli Fractions Volume (ml) Protein (mg/ml) Activity (EC50)a Purification (fold)b Cell lysatec 50 115.84 75.580 1 Affinity purification 10 0.65 0.040 1890 FPLC 2 2.0 0.025 3023 EC50 is defined as the concentration of PA (μg/ml) along with LF ( 1 μ g / m l ) r e q u i r e d k i l l i n g 5 0 % o f t h e J 7 7 4 A . 1 c e l l s . Purification fold was determined by dividing EC50 for cell lysate with EC50 for fractions obtained from different columns. Cell lysate prepared from 2 litres of culture. BINDING OF PA TO CELL SURFACE RECEPTORS A Protein CPM PA(ng) PA/cell protein b (ng/mg) nPA 82679 ±1169 6.89 7.25 rPA 79986 ±1388 7.34 7.72 J774A.1 CELLS WERE INCUBATED WITH 1µg OF RADIOIODINATED PA (nPA AND rPA) FOR 3 HRS. AT 4C. a PROTEIN CONTENT OF THE CELLS PER WELL WAS 0.95 ± 0.05 mg AS DETERMINED BY LOWRY’S METHOD. b BINDING OF RECOMBINANT PA TO LF PA-LF complex PA63 LF PA20 LF (1µg) was incubated with trypsin – nicked PA (1µg) and samples were analyzed on a 4.5% native PAGE. The gel was stained with Coomassie-blue. BINDING OF LF TO RECEPTOR BOUND PA Protein (ng/mg) PA / cell protein b LF alone 0.180.02 nPA+LF 3.70 0.12 rPA+LF 3.55 0.15 J774A.1 CELLS WERE INCUBATED WITH 1µg OF RADIOIODINATED LF (nLF AND rLF) ALONG WITH PA 1µg/ML FOR 12 HRS. AT 4C. THE CELLS WERE WASHED WITH HANK’S BUFFERED SALINE SOLUTION AND SOLUBLIZED IN 100 mM NaOH. RADIOACTIVITY WAS COUNTED IN A GAMMA COUNTER. PROTEIN CONTENT OF THE CELLS PER WELL WAS 1.1 ± 0.05 mg AS DETERMINED BY LOWRY’S METHOD. MACROPHAGE LYSIS ASSAY 120 NATIVE PA ALONE 100 RECOMBINANT PA ALONE %age viability 80 60 NATIVE PA +LF 40 RECOMBINANT PA + LF 0.01 0.1 PA( g/ml) J774A.1 CELLS WERE INCUBATED WITH VARYING CONCENTRATIONS OF PA ALONE OR IN COMBINATION WITH LF (1g/ml) FOR 3HRS. AT 37C. PA FROM Bacillus anthracis, RECOMBINANT PA PA FROM Bacillus anthracis WITH LF, RECOMBINANT PA WITH LF. 20 0 0.001 BIOLOGICAL ACTIVITY OF PA PURIFIED FROM Bacillus anthracis AND E. coli DH5 CELLS. 1 Protective efficacy of the rPA against B. anthracis S. No. Group Conc. used 1. Unvaccinated control (challenged) Vaccinated control (unchallenged) Protective Antigen en from B. anthracis PBS 00/18 0 -- 50 g 18/18 100 -- 5g 12/18 66 -- 1g 6/18 33 -- 50 g 12/18 66 100 10 g 12/18 66 100 5 g 12/18 66 100 1g 6/18 33 50 2. 3. 4. Recombinant protective antigen Survivors/ % Relative Total survival efficacy Relative efficacy is defined as the percentage of rPA immunized animals survivingg after virulent spore challenge w.r.t., the Native PA from B. anthracis. Concentration of the anthrax spore vaccine used is 10 x 107 spores per ml. EXPRESSION, OPTIMIZATION AND PURIFICATION OF PA, LF and EF PAG E. coli with expression plasmid construct GROWTH CONDITION OPTIMIZATION FOLLOWED BY HIGH DENSITY FED BATCH CULTURE Ni-NTA AFFINITY CHROMATOGRAPHY & GEL FILTRATION PURIFIED PA Same way LF and EF were purified OVERPRODUCTION OF rPA METHOD OF FEEDING : pH-DO-stat FEED : 25xcomplex media (LB + 25% w/v glycerol) INCREASE IN BIOMASS : OD600>100units WET CELL WEIGHT: 195 grams/litre DRY CELL WEIGHT : 52 grams/litre PA : 20-30% of total cell protein PURIFICATION: Ni-NTA affinity chromatography and Gel : 90-95% pure Filtration Yield : 3-5 g/L equivalent to ~1million shots compared to currently available vaccines. Technology transfer of PA production Technology transferred to Panacea Biotec Ltd. A Pharmaceutical Company already producing vaccines for Polio and Hepatitis B. Scientists from Panacea Biotech Ltd. have been given extensive training in JNU for making recombinant vaccine. JNU scientists have gone and helped Panacea Biotech Ltd to produce 5 batches of recombinant vaccine in GMP facility of Panacea Biotech Ltd. Panacea Biotech Ltd., scientists have produced 5 batches of rPA for toxicity and efficacy studies under GMP. Toxicity studies on mice, and rats at Rallis India Ltd. Banglore have shown that recombinant anthrax vaccine (rPA) is not toxic. Pre–Exposure studies on efficacy have been completed. Phase-I/II, open labeled, randomized, placebo controlled, ascending dose trial to evaluate the safety and immunogenecity of recombinant protective antigen (rPA) anthrax vaccine have been initiated in Oct. 2004 and likely to be completed by Dec. 2005. immunogenecity and Immunogenicity of Anthrax toxin components PA :Good Immunogen PA+LF+EF: Better Immunogen. LF and EF cannot be added in the vaccine due to associated toxicity. Mutants defective in any one of the steps of intoxication may be added in vaccine with PA without causing toxicity. Generation of non toxic mutants of PA, LF, EF gene M M pExp M M Long PCR with Pfu turbo using adjacent, partially overlapping oligonucleotides encoding the desired mutation at the 5’ end of the primer. gene PCR amplified nicked plasmid M M M Methylated Plasmid template M : Mutation point M : Methyl group (CH3) DpnI Digetsion (degrades methylated template DNA while spares unmethylated PCR amplified product) Transformation into competent E. coli cells. Mutants confirmed by sequencing PA STRUCTURE: FUNCTIONALLY IMPORTANT RESIDUES DOMAIN 1: BINDING TO LF/EF DOMAIN 3: PA OLIGOMERIZATION DOMAIN2: MEMBRANE INSERTION AND TRANSLOCATION OF LF/EF RESIDUES OF PROTECTIVE ANTIGEN INVOLVED IN BINDING TO LF/EF Phe 202 Ile 207 Leu 203 Pro 205 DOMAIN 1b Ref: Chauhan V, Bhatnagar R. Identification of amino acid residues of anthrax protective antigen involved in binding with lethal factor. Infect Immun. 2002 Aug;70(8):4477-84 Residues of PA Involved In Membrane Insertion And Translocation of LF/EF Trp 346 Leu 352 DOMAIN 2 Ref: Batra S, Gupta P, Chauhan V, Singh A, Bhatnagar R. (2001) Trp 346 and Leu 352 residues in protective antigen are required for the expression of anthrax lethal toxin activity. Biochem Biophys Res Commun. 281:186-92 RESIDUES OF PROTECTIVE ANTIGEN NEEDED FOR OLIGOMERIZATION Leu 566 Ile 562 Ile 574 Phe552 DOMAIN 3 Ref: Ahuja N, Kumar P, Bhatnagar R.Hydrophobic residues Phe552, Phe554, Ile562,Leu566, and Ile574 are required for oligomerization of anthrax protective antigen.Biochem Biophys Res Commun. 2001 Sep 21;287(2):542-9. SIMILARITY BETWEEN EF AND LF SEQUENCES Score = 123 bits (309), Expect = 9e-27 Identities = 77/225 (34%), Positives = 123/225 (54%), Gaps = 3/225 (1%) Query: 63 Sbjct: 73 INNLVKTEFTNETLDKIQQTQDLLKKIPKDVLEIYSELGGEIYFTDIDLVEHKELQDLSE 122 + ++VK E E K + + LL+K+P DVLE+Y +GG+IY D D+ +H L+ LSE MKHIVKIEVKGEEAVKKEAAEKLLEKVPSDVLEMYKAIGGKIYIVDGDITKHISLEALSE 132 Query: 123 EEKNSMNSRGEKVPFASRFVFEKKRETPKLII-NIKDYAINSEQSKEVYYEIGKGISLDI 181 ++K + G+ +V+ K+ P L+I + +DY N+E++VYYEIGK +S DI Sbjct: 133 DKKKIKDIYGKDALLHEH YVYAKEGYEPVLVIQSSEDYVENTEKALNVYYEIGKILSRDI 192 Query: 182 ISKDKSLDPEFLNLIKXXXXXXXXXXXXFSQKFKEKLELNNKSIDINFIKENLTEFQHAF 241 +SK +FL+++ F +L+ + + F+++N E Q F Sbjct: 193 LSKINQPYQKFLDVLNTIKNASDSDGQDL--LFTNQLKEHPTDFSVEFLEQNSNEVQEVF 250 Query: 242 SLAFSYYFAPDHRTVLELYAPDMFEYMNKLEKGGFEKISESLKKE 286 + AF+YY P HR VL+LYAP+ F YM+K + E LK + Sbjct: 251 AKAFAYYIEPQHRDVLQLYAPEAFNYMDKFNEQEINLSLEELKDQ 295 QUERY: THE SEQUENCE OF EF FIRST DOMAIN SUBJECT: THE SEQUENCE OF LF FIRST DOMAIN HOMOLOGOUS STRETCH OF LF/EF NH2 147Val-Tyr-Tyr-Glu-Ile-Gly-Lys 153 LF NH2 136Val-Tyr-Tyr-Glu-Ile-Gly-Lys 142 EF The amino terminal region of LF and EF is required in binding to PA. Sequence analysis reveals that 1 to 300 amino acids have several homologous stretches. Maximum homology was observed at a stretch of seven residues (Val-Tyr-Tyr-Glu-Ile-Gly-Lys ). Therefore, in order to determine to the role of these residues each amino acid of this stretch was substituted with alanine. LF Structure: Residues Needed For Binding To PA Val 147 II PA binding binding PA Glu 150 Tyr 148 Ile 151 Tyr 149 Lys 153 Gly 152 Leu 189 II VIP2 like Leu 188 Asp 187 Phe190 III Helix bundle IV Catalytic centre MAPKK-2 Mutants of Domain 1 defective in binding to PA: Tyr 148 Grey residues: involved in binding Tyr 149 Glu 150 Yellow and green: dispensable ones Il e 151 Lys 153 Asp187 Phe190 References: 1. Singh A, Chauhan V, Sodhi A, Bhatnagar R. Asp 187 and Phe 190 residues in lethal factor are required for the expression of anthrax lethal toxin activity. FEMS Microbiol Lett. 2002 Jul 2; 212(2):183-6. 2. Gupta P, Singh A, Chauhan V, Bhatnagar R. Involvement of residues 147VYYEIGK153 in binding of lethal factor to protective antigen of Bacillus anthracis. Biochem Biophys Res Commun. 2001 Jan 12;280(1):158-63. PA BINDING DEFECTIVE MUTANTS OF EF Domain 3 Domain 2 Linker Ca 2+ Calmodulin Domain 1 PA binding domain MUTATED RESIDUES Val136 Tyr137 Tyr138 Glu139 Ile140 Gly141 Lys142 BINDING/TOXICITY + + / + + –– –– / –– –– –– –– / –– –– + + /+ + –– –– / –– –– + + / + + –– –– / –– –– Ref: Kumar P, Ahuja N, Bhatnagar R. 2001. Purification of anthrax edema factor from Escherichia coli and identification of residues required for binding to anthrax protective antigen. Infect Immun. Oct;69(10):6532-6 THERMOSTABILIZATION OF PA • COSOLVENT MEDIATED: MgSO4 and Trehalose are the best among the studied cosolvents. Additive None Sorbitol Xylitol Trehalose Sodium Citrate MgSO4 Molarity PA activity retained after 48 hrs of incubation at 37oC 1.0 2.0 1.5 1.0 ~5 ~10 ~18 ~74 ~67 3.0 ~83 Ref: Radha C, Salotra P, Bhat R, Bhatnagar R. Thermostabilization of protective antigenthe binding component of anthrax lethal toxin. J.Biotechnol. 1996, Oct 1;50(2-3):235-42. Gln277Ala and Phe554Ala increase thermal stability. Activity of PA mutants retained after 48 hrs of incubation at 37oC in comparison with native PA. S.NO. MUTANT 1. Gln277Ala RESIDUAL ACTIVITY AFTER 48 HRS. OF INCUBATION AT 37 OC ~45% 2. Phe554Ala ~90% 3. Native PA 0% Ref: Singh S, Ahuja N, Chauhan V, Rajasekaran E, Mohsin Waheed S, Bhat R, Bhatnagar R. Gln277 and Phe554 residues are involved in thermal inactivation of protective antigen of Bacillus anthracis. Biochem Biophys Res Commun. 2002 Sep 6;296(5):1058-2 Transgenic plants as a source of Edible vaccine against Anthrax • Cloned and expressed in Tobacco plants. Ref: Aziz MA, Singh S, Anand Kumar P, Bhatnagar R. Expression of protective antigen in transgenic plants: a step towards edible vaccine against anthrax. Biochem Biophys Res Commun. 2002 Dec 6;299(3):345-51. •Transgenic Tomatoes are in early stage of development. Identification of the transgene in genomic DNA by PCR amplification 1 2 3 4 5 6 7 8 9 10 11 1.5kb Genomic DNA extracted from tobacco leaves was used as template. Primers flanking 1.5 Kb region within the PA gene were used to carry out the reaction. Molecular analysis Protective antigen expression determined using immunoblot analysis. Functional efficacy established using cytotoxicity assay. Immunoblot detection of protective antigen with antisera raised against purified recombinant PA 209 kDa 1 2 3 4 5 120 kDa 78 kDa 47.7 kDa Lane 1-3: PA expressed in transgenic plants Lane 4 : rPA, Lane 5: Negative control i.e. Total soluble protein extracted from untransformed tobacco plants Functional assay of plant expressed PA Total soluble protein from different plant samples was incubated along with 1ug/ml LF. The percentage killing of RAW264.7 cells ranged between 26% to 98% owing to different expression levels in different plants percentage killing of macrophage cells 120 100 80 60 40 20 0 1 2 3 4 5 6 7 8 9 10 Number Of plant from which TSP was isolated Tomato Callus Differentiating On Selection Medium Putative Transgenic Tomato Plants at Bottle Stage Putative Tomato Transgenic Plants Transferred To Pots CONCLUSIONS We have PCR amplified PA gene. Overexpressed in suitable Vectors. Bioprocess optimized upto near industrial production. Recombinant PA was found to be fundamentally identical to native antigen. Thermostabilization of PA has been achieved. Technology or producing recombinant vaccine transferred to M/s. Panacea Biotec Ltd. Non-toxic variant of PA, LF, & EF generated for next generation vaccine. biologically PA gene was expressed in Tobacco & Tomatoes. Clinical trials are being conducted. & Thank You