* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Organometallic Compounds
Survey
Document related concepts
Enantioselective synthesis wikipedia , lookup
Elias James Corey wikipedia , lookup
George S. Hammond wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Discodermolide wikipedia , lookup
Stille reaction wikipedia , lookup
Persistent carbene wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Transcript
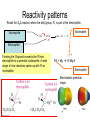
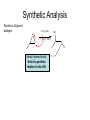
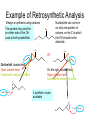
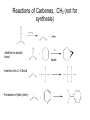
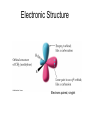
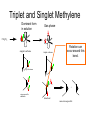
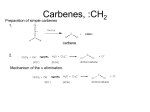
Organometallic Compounds Chapter 15 Carbon Nucleophiles: Critical in making larger organic molecules. Review some of the ones that we have talked about…. Cyanide ion: CN- + RX RCN RCH2NH2 Synthetic thinking: Disconnect + NH2 CN- Br Acetylide anions: strong base RC CH RX RC RC C: CR Synthetic Thinking: This offers many opportunities provided you can work with the two carbon straight chain segment. Ph X Ph Ph Ph Ph Ph Enolate anions: O or base O X O O OEt H RX O O OEt OEt H H R Try to see what factors promote the formation of the negative charge on the carbon atoms: hybridization, resonance. We examine two types of organometallics: RMgX, a Grignard reagent, and RLi, an organolithium compound Preparation d-d+ dd+ Solvated by ether, aprotic solvent Basicity Recall that a carbanion, R3C:-, is a very strong base. So also Grignards and alkyl lithiums. Ethane, a gas. Bottom Line: Grignards are destroyed by (weak) protic acids: amines, alcohols, water, terminal alkynes, phenols, carboxylic acids. The Grignard, RMgX, is converted to a Mg salt eventually and RH. The liberation of RH can serve as a test for protic hydrogens. Reactivity patterns Recall the SN2 reaction where the alkyl group, R, is part of the electrophile. Nucleophile Nucleophile Nu:- + R-X Nu - R + X- Electrophile Forming the Grignard converts the R from electrophile to a potential nucleophile. A wide range of new reactions opens up with R as nucleophile. - + RX + Mg R-Mg-X Electrophile Electrostatic potential maps. + - Recall Reactions of Oxiranes with Nucleophiles Recall opening of oxirane with a strong, basic nucleophile. CH3OO H H H H OH H H CH3 H3CO CH3 The next slides recall the diversity of nucleophiles that may be used. Observe that there is limited opportunity of creating new C-C bonds, welding together two R groups. We seem to be somewhat lacking in simple carbon based nucleophiles. Recall Synthetic Applications nucleophile Only reaction with the acetylide anion offers the means of making a new C-C bond and a larger molecule. Problem is that a terminal alkyne is needed. A Grignard has a reactive, negative carbon. Now examine reaction of Grignard and oxirane ring. Net results Newly formed bond The size of the alkyl group has increased by 2. Look at this alcohol to alcohol sequence R-OH R-X R-Mg-X R-CH2-CH2-OH. The functionality (OH) has remained at the end of the chain. We could make it even longer by repeating the above sequence. Note attack on less hindered carbon Now a substituted oxirane… Newly formed bond Synthesis Example Retrosynthesize the following OH OH O CH2CH=CH2 CH2=CH - CH2MgBr Recall reaction of a nucleophile with an (oxirane) epoxide to give a HO-CC-Nu pattern. Back side attack gives anti opening. Trans geometry suggests trying an oxirane. What should the nucleophile be? The allyl group should be the nucleophile. This is done by using a Grignard (or Gilman). Gilman Reagent (Lithium diorganocopper Reagents) Li R-X Preparation of Gilman Reagents CuI R-Li R2CuLi Gilman Reactions of Gilman Reagent Coupling Reaction Used to create new C – C bonds.. Overall result. R-X + R’-X R – R’ Necessary details Li As before: Next step: CuI R-Li R-X R2CuLi electrophile R'-X R2CuLi R - R' Restrictions on the process. Caution. R group which goes into Gilman may be methyl, 1o (best not 2o or 3o), allylic, vinylic (unusual), aryl Alkyl (not 3o), vinylic nucleophile Particularly useful, reaction with vinyl halides to make an alkene. trans Note that the stereochemistry of the alkene is retained. Gilman and oxiranes 1. R2CuLi HO O 2. H2O, HCl R R of the Gilman reagent is the nucleophile, typical of organometallics. Because in basic media (acid destroys Gilman) oxygen of oxirane can not be protonated. Less hindered carbon of oxirane is attacked. Synthetic Analysis Similar to Grignard analysis. 1. R2CuLi HO O 2. H2O, HCl R Newly formed bond. Note its position relative to the OH. Example of Retrosynthetic Analysis Design a synthesis using oxiranes The oxirane ring could be on either side of the OH. Look at both possibilities. Ph Nucleophile can come in on only one position of oxirane, on the C to which the OH should not be attached… OH OH OH or Ph On the left, located here. Open oxirane here. Nucleophile makes this bond. Ph On the right, located here. Open oxirane here. Nucleophile makes this bond. O (PhCH2)2CuLi 2 synthetic routes available O Ph LiCu(CH2CH3)2 Synthesis Example Carry out the following transformation in as many steps as needed. Br O OCH3 O target OH Br O OCH3 Remember oxidation of a secondary alcohol can produce a ketone. OCH3 Note pattern of a nucleophile (OCH3) then CC then OH. Use an epoxide. Epoxides can come from alkenes via peracids. Alkenes can come from halides via E2. Carbenes, :CH2 Preparation of simple carbenes 1. carbene 2. Mechanism of the a elimination. Reactions of Carbenes, :CH2 (not for synthesis) Addition to double bond. Insertion into C-H bond Formation of ylide (later) liquid Simmons Smith Reaction (for synthesis, addition to alkenes to yield cyclopropanes) CH2I2 + Zn(Cu) ICH2ZnI Carbenoid, properties similar to carbenes. Electronic Structure Electrons paired, singlet Triplet and Singlet Methylene Dominant form in solution Gas phase CH2N2 singlet carbene triplet carbene Rotation can occur around this bond. pi electrons CH2 + stereospecific addition diradical non-stereospecific