* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Nucleophilic Substitution Swapping
Woodward–Hoffmann rules wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Marcus theory wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Stille reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
George S. Hammond wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
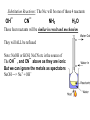
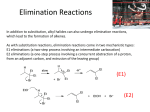
Nucleophilic Substitution Swapping Generic Equation: Swap R-X + Nu: R-Nu + :XThe problem lies in the mechanism. Importance of Alkyl Halides Precursor to many others NUCLEOPHILIC SUBSTITUTION Theory - nucleophile means ‘liking positive’ - a dipole is induced (by a Nu:) in the C-X bond and it is polar polar bond, why is it polar with chlorine? Answer: ∆E=0.4, electronegativity difference OH¯ CN¯ NH3 H 2O These are the 4 Nu: NaOH, NaCN the 2 moleculars are as is SN2 :NUCLEOPHILIC SUBSTITUTION MECHANISM MECHANISM: 2 Steps 1) The NU: electrons attacks the slightly positive carbon atom 2) The polar bond breaks unevenly (heterolytic) and Br- released Back attack Polar Bond Both eTo halogen 2 products Note/Points: -attack from back (electronegative halogen will forces this) -show the polar bond SN2 All 10 (primary)halides react as SN2 reactions. The 2 does NOT mean steps S = substitution N = Nucleophile 2 = second order (both reactants determine rate) Note: this is for primary halides, tertiary will under go SN1 as we will see later, secondary will do both – but we are not responsible to know about secondary reactions 1O Haloalkanes SN2 • https://www.youtube.com/watch?v=h5xvaP 6bIZI There are 4 Substitutions 1) WITH HYDROXIDE C2H5Br(l) + OH- (aq) -->C2H5OH(l) +Br-(aq) 2) WITH A CYANIDE C2H5Br(l) + CN- (aq/alc) —>C2H5CN + Br-(aq) 3) WITH 2 AMMONIAS C2H5Br(l) + 2NH3(aq/alc)—> C2H5NH2 + NH4Br 4) WITH H2O (solvolysis) solvent and reactant at same time C2H5Br(l) + H2O (l) —> C2H5OH(l) + HBr (aq) SN2 Energy Diagram • The SN2 reaction is a one-step reaction. • Transition state is highest in energy. Chapter 6 8 Substitution Reactions : The Nu: will be one of these 4 reactants OH¯ CN¯ NH3 H2O These four reactants will be similar in result and mechanism They will ALL be refluxed Note: NaOH or KOH, NaCN etc is the source of The OH¯ , and CN¯ above as they are ionic But we can ignore the metals as spectators NaOH ---> Na⁺ + OH⁻ NUCLEOPHILIC SUBSTITUTION AQUEOUS HYDROXIDE ALCOHOLS Reagent Conditions Product Aqueous* NaOH or KOH (aq) (OH¯) Reflux in aqueous solution (SOLVENT IS IMPORTANT) Alcohol Nucleophile hydroxide ion ( : OH¯) Equation e.g. C2H5Br(l) + Na OH-(aq) Spectator ——> C2H5OH(l) + Na Br-(aq) Spectator Mechanism STEP 1 Nu: attack STEP 2 :Br- forms and leaves * WARNING It is important to quote the solvent when answering questions. Elimination takes place when ethanol is the solvent - SEE LATER NUCLEOPHILIC SUBSTITUTION CYANIDE makes Nitriles AND makes chain LONGER Reagent Conditions Product Nucleophile Equation Alcohol/ Aqueous, NaCN / KCN Reflux in aqueous , alcoholic solution Nitrile (cyanide) cyanide ion (CN¯) e.g. C2H5Br + Na CN (aq/alc) ——> C2H5CN + Na CN-(aq) Mechanism 1-bromoethane Importance propanenitrile Br- extends the carbon chain by one carbon atom, called propanenitrile . NUCLEOPHILIC SUBSTITUTION AMMONIA makes an amine Reagent Conditions Product Nucleophile Equation Aqueous, alcoholic ammonia (in EXCESS, or 2 Ammonia’s) Reflux in aqueous , alcoholic solution under pressure Amine Ammonia (NH3 (aq/alc) ) e.g. C2H5Br + 2NH3 (aq / alc) ——> C2H5NH2 + NH4Br (i) C2H5Br + NH3 (aq / alc) (ii) C2H5NH3 + NH3 (aq / alc) ——> C2H5NH3 + HBr ——> C2H5NH2 + NH4Br _______________________________________________ C2H5Br + 2NH3 (aq / alc) ——> C2H5NH2 (l) + NH4Br (aq/alc) Mechanism Ethaneamine Bromide NUCLEOPHILIC SUBSTITUTION AMMONIA Why excess ammonia? The second ammonia molecule ensures the removal of the H to make an amine. A large excess ammonia ensures that further substitution doesn’t take place. Problem Amines are also nucleophiles (lone pair on N) and can attack another molecule of haloalkane to produce a 2° amine. This too is a nucleophile and can react further producing a 3° amine and, eventually an ionic quarternary ammonium salt. .. C2H5NH2 + C2H5Br ——> HBr + (C2H5)2NH diethylamine, a 2° amine .. (C2H5)2NH + C2H5Br ——> HBr + (C2H5)3N (C2H5)3N + C2H5Br ——> (C2H5)4N+ Br¯ triethylamine, a 3° amine tetraethylammonium bromide a quaternary (4°) salt NUCLEOPHILIC SUBSTITUTION WATER Makes Alcohols Details A similar reaction to that with OH¯ takes place with water. It is slower as water is a poor nucleophile. Water may be looked at as HOH THE OH AND BR SWAP Equation C2H5Br(l) + H2O(l) ——> C2H5OH(l) + HBr(aq) We show the product as HBr and not just Br- because it is molecular and the H in the water is not an ion like Na+ or K+. As well, H3O+ forms, which we rewrite as H+ and the water from the H3O+ is part of the (aq) SN1 V SN2 There is very little difference in these reactions. Both have 2 steps and the same product , the same reactants. Hard to see any difference S = substitution (both will swap) N= Nucleophile (both will use the same Nu: 1 =Rate of reaction (depends on 1 reactant) called 1st order 2 =Rate of reaction (depends on 2 REACTANTS) called 2nd order Steric Hinderance • With a tertiary halogenoalkane, NU: attack is impossible. The back of the molecule is completely cluttered with CH3 groups. • It has to go by an alternative mechanism. Tertiary Haloalkenes SN1 Steric hindrance means the Nu: cannot approach and has no effect on the reactions first step The [Nu:] nucleophile has no effect. Only the [haloalkane] affect the reaction rate. Thus it is first order. rate = k [haloalkane] 1 Mechanism for 3o Halides SN1 SLOW (RDS) STEP Carbocation Intermediate FAST STEP http://ibchem.com/IB16/10.34.htm SN1 Energy Diagram • Forming the carbocation is an endothermic step. • Step 2 is fast with a low activation energy. Chapter 6 19 Film Clip 30 Haloalkanes SN2 (single step) https://www.youtube.com/watch?v=JmcVgE2 WKBE Secondary Halides • They undergo both SN1 and SN2 reactions so on an exam if you are given a secondary halide, either mechanism will receive full credit in marks LAST REACTION ELIMINATION 3 WAYS to Favor Elimination To favor elimination rather than substitution use: 1) More heat 2) More concentrated hydroxide in ALCOHOL 3) pure ethanol as the solvent Elimination • The :OH- is a BASE, in alcohol and takes a hydrogen (H+) to form water. This sets off a cascade where a C=C forms and the halide ion is lost. (as always Reflux) 24 Elimination Via OH- BASE Step 1: The :OH- is NOT a nucleophile, it is a BASE, and attacks H Step 2: The H+ leave and e- pair make C=C Step 3: The halogen (here as :Br-) leaves GENERIC CASCADING REACTION + H2O + :Br- An Example + H2O + :Br- ELIMINATION Reagent Alcoholic sodium (or potassium) hydroxide Conditions Product Mechanism Equation Reflux in alcoholic solution, ALL alcohol Alkene Elimination C3H7Br + NaOH(alc) ——> C3H6 + H2O + NaBr Mechanism the OH¯ ion acts as a base and picks up a proton the proton comes from a carbon atom next to that bonded to the halogen the electron pair left moves to form a second bond between the carbon atoms the halogen is displaced overall there is ELIMINATION of HBr. Complication:Cis / Trans with unsymmetrical halalkanes, you get mixture of products ELIMINATION Complication If the haloalkane is unsymmetrical a mixture of isomeric alkene products is obtained. but-1-ene (Minor Product) 2-bromobutane but-2-ene (Major Product) 2-bromobutane can exist as cis and trans isomers ELIMINATION v. SUBSTITUTION The products of reactions between haloalkanes and OH¯ are influenced by the solvent SOLVENT ROLE OF OH– MECHANISM PRODUCT WATER NUCLEOPHILE SUBSTITUTION ALCOHOL ALCOHOL BASE ELIMINATION ALKENE Modes of attack Aqueous soln OH¯ acts as a nucleophile Alcoholic soln OH¯ acts as a base (A BASE IS A PROTON ACCEPTOR) Both reactions take place at the same time but by varying the solvent you can influence which mechanism dominates. Elimination Clips • https://www.learnnext.com/CBSE/Class12/Chemistry/Haloalkanes-andHaloarenes/Chemical-Reactions-Of-AlkylHalides-Elimination-Reactions/L1804203.htm • Franklychem • https://www.youtube.com/watch?v=o3lk77s dFHY The ability of solvents to stabilize ions through solvation is directly associated with their polarity. Polar solvents, such as water, can stabilize the ions 3X more than alcohol (see chart) Methanol can stabilize ions through solvation, but water is best at controlling ions. The :OH- is a super strong ion and needs controlling. Alcohol controls less and is better to dissolve weak :Nu’s such as NH3 and CN, as they do not need to be controlled like OH- as they are weak ions Aprotic solvents ε Protic solvents ε Hexane 1.9 Acetic acid 6.2 Benzene 2.3 1-Methyl-2-propanol 11 Diethyl ether 4.3 Ethanol 34.3 Chloroform 4.8 Methanol 33.6 Hexamethylphosphora mide (HMPT) 30 Formic acid 58.0 Dimethyl formamide (DMF) 38 Water 80.4 Dimethyl sulfoxide (DMSO) 48