* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Practical II - Faculty Websites
DNA damage theory of aging wikipedia , lookup
DNA polymerase wikipedia , lookup
DNA vaccination wikipedia , lookup
Microevolution wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Non-coding DNA wikipedia , lookup
United Kingdom National DNA Database wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Epigenomics wikipedia , lookup
Genomic library wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Metagenomics wikipedia , lookup
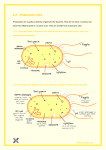
Genealogical DNA test wikipedia , lookup
Nucleic acid double helix wikipedia , lookup
Molecular cloning wikipedia , lookup
DNA supercoil wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
Helitron (biology) wikipedia , lookup
SNP genotyping wikipedia , lookup
Extrachromosomal DNA wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
Microsatellite wikipedia , lookup
Gel electrophoresis of nucleic acids wikipedia , lookup
Bisulfite sequencing wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Dharshani Nanayakkara Fall 2005 – BIOL 251 Lab Sec. 7, 8, & 9 Page 1 of 4 NOTE: THIS IS FROM LAST SEMESTER!!! POSTED IT ONLINE BECAUSE THERE ARE SOME GOOD QUESTIONS…SOME OF THE QUESTIONS ARE 251 SPECIFIC, SOME 351 SPECIFIC!! BIOL 251 Lab Practicum II – Study Guide REVIEW DAY/OPEN LAB – Friday, December 2, 2005 8.30 a.m. to 4.30 p.m. I will be there from 2.30 – 4.30 p.m. Please utilize this time to review all the demos. ***Make sure to record results of your conjugation and food microbiology plates. There are 3 additional study guides on the class website (http://www.unlv.edu/staff/wmojica/). I strongly recommend that you study these thoroughly. It might be helpful to refer to these study guides while looking at the demos. Practical II General Review Guide Practical II Tests Study Photos for Practical II Review the lab manual, lab handouts, quizzes and the lab reports, especially the review questions!!! The following questions cover only the following exercises: Polymerase Chain Reaction (study the PCR Handout) Bacterial Conjugation Food Microbiology ***You may check your answers with me during the open lab. 1. (351) Polymerase chain reaction is a method used to amplify a specific DNA sequence in vitro by repeated cycles of synthesis. Each PCR “cycle’ involves 3 steps. Complete the following table with information pertinent to each step. What Happens Temperature Time Step 1 Step 2 Step 3 2. (351) What are the three items that should be added in excess at the start of the PCR process? i. ii. Dharshani Nanayakkara Fall 2005 – BIOL 251 Lab Sec. 7, 8, & 9 Page 2 of 4 iii. 3. (351) Why is a primer needed at each “end” of the DNA to be amplified? 4. (351) Suppose that you want to amplify the following DNA sequence using PCR. First, you have to prepare two oligonucleotide primers – reverse and forward. If you have all the necessary raw material, including all four dNTPs, what would be your two primers? 5’ 3’ AAGTCCACCGTTAAGCGCCTAAATCCGCCCTAAATGCTTAAG TTCAGGTGGCAATTCGCGGATTTAGGCGGGATTTACGAATTC 3’ 5’ 5. (351) Why is PCR usually performed with Taq (Thermus aquaticus) DNA polymerase? 6. (351) What other reagents are necessary for PCR and why? 7. (351) What are some applications of PCR? 8. (351) What is the most common way of seeing the results of a PCR? 9. (351) Gel electrophoresis is a powerful tool for the separation of macromolecules. Once electric current is applied, what characteristics determine the rate of migration of a DNA molecule? 10. (351) The following figure shows an agarose gel. Three fragments of DNA (A, B, and C) have been loaded into wells toward the top of the gel, and the positive pole of the electric field is at the bottom. The sizes of the three DNA fragments are indicated below. (a) After a defined period of migration time, where will you see the three DNA fragments? _ DNA Fragment A B C Size (kb) 4000 500 1800 Dharshani Nanayakkara Fall 2005 – BIOL 251 Lab Sec. 7, 8, & 9 Page 3 of 4 + (b) What dye is usually used to stain the above gel and what would you do to get an image of the DNA bands? 11. (251) Define bacterial conjugation. What are the three steps that conjugation depends on? 12. (251) What are the most important characteristics of conjugative plasmids? 13. (251) In what region of the conjugative plasmid are the genes that control conjugation contained, and what are these genes responsible for? 14. (251) Suppose that you are asked to perform a conjugation experiment using the following bacterial strains: Donor – Escherichia coli Kanamycin resistant (Kmr), streptomycin sensitive (Sms), gentamicin resistant (Gmr) – the kanamycin and streptomycin genes are located on a conjugative plasmid and the gentamicin gene is located on the bacterial chromosome. Recipient – Pseudomonas fluorescens Kanamycin sensitive (Kms), streptomycin resistant (Smr), gentamicin sensitive (Gms) (a) What would be the first step in your conjugation experiment? Explain why you would perform this step? (b) Suppose that you plated the donor strain in the following selective media plates. In which plate(s) will it grow? Km+Sm (c) Km+Gm Km+Sm+Gm Suppose that you plated the recipient strain in the following selective media plates. In which plate(s) will it grow? Sm+ Gm Km+Sm Km+Sm+Gm Dharshani Nanayakkara Fall 2005 – BIOL 251 Lab Sec. 7, 8, & 9 (d) After mixing the donor and recipient strains, you provided sufficient time for the mating cells to make contact. After that, you plated this culture in the following selective media plates. In which plate(s) will the exconjugant strain grow? Km+Sm+Gm (e) Sm Km+Sm Using the data in the following table, calculate the frequency of conjugation between these two strains. Exconjugants Recipients 15. Page 4 of 4 Dilution Factor 10-4 10-7 CFU 103 221 CFU/ml 1.03 x 106 2.21 x 109 (251) In a conjugation experiment, the following results were obtained. Calculate the frequency of conjugation. Exconjugants Recipients Dilution Factor 10-4 10-6 CFU 87 198 CFU/ml 16. (Both) Does a heterotrophic plate count of a food sample accurately reflect the total amount of bacteria present in that sample? Why? 17. (Both) What is the difference between food poisoning and food intoxication? 18. (Both) What is a coliform? Give some examples. 19. (Both) (a) List a few genera of bacteria, molds, and yeasts that are associated with foods. (b) List a few bacterial species that you don’t want to find in your food. 20. (Both) Suppose that you wanted to determine the number of viable bacteria in a raw hamburger. You performed a heterotrophic plate count of your food sample as outlined in Figure 50.1 in your lab manual. If 180 colonies were counted from a 10-5 dilution of a 25- Dharshani Nanayakkara Fall 2005 – BIOL 251 Lab Sec. 7, 8, & 9 Page 5 of 4 gram sample and 1 ml was plated, calculate the number of bacteria per gram of your raw hamburger. If your friend were to eat a hamburger that is in the same packet from which you obtained the above sample, would you let him/her eat it?