* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Preimplantation Genetic Testing An Overview
Cre-Lox recombination wikipedia , lookup
Deoxyribozyme wikipedia , lookup
SNP genotyping wikipedia , lookup
Skewed X-inactivation wikipedia , lookup
Pathogenomics wikipedia , lookup
Genomic imprinting wikipedia , lookup
Y chromosome wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Human genome wikipedia , lookup
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
Whole genome sequencing wikipedia , lookup
Genome evolution wikipedia , lookup
DNA paternity testing wikipedia , lookup
Genealogical DNA test wikipedia , lookup
Population genetics wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Molecular Inversion Probe wikipedia , lookup
Non-coding DNA wikipedia , lookup
Neocentromere wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
Human genetic variation wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
X-inactivation wikipedia , lookup
Genome editing wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Genomic library wikipedia , lookup
Genetic engineering wikipedia , lookup
Medical genetics wikipedia , lookup
Genetic testing wikipedia , lookup
Public health genomics wikipedia , lookup
History of genetic engineering wikipedia , lookup
Comparative genomic hybridization wikipedia , lookup
Microevolution wikipedia , lookup
Genome (book) wikipedia , lookup
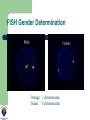
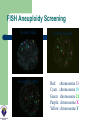
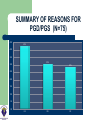
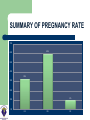
DEVELOPMENTS AND LANDMARKS OF GENETIC TESTING IN WEST AFRICA PROF. OLADAPO ASHIRU M.B.B.S, MS, PHD. HCLD/CC(ABB-USA), FASN, FNSEM, OFR 48th Annual General Meeting and Scientific Conference SOGON ASABA 2014. ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Conception requiring the complex handling or manipulation of male and female gametes in-vitro to facilitate pregnancy LANDMARKS IN ART 1969 Sperm Capacitation Chang & Yamagimachi 1976 IVF in London Steptoe & Edwards 1978 IVF baby London Steptoe & Edwards (First IVF Baby – Louise Brown) 1977 LHRH Schally & Guileman 1978 FSH +ve feedback Ashiru & Blake 1980 IVF in Melbourne, Australia Carl Wood 1982 IVF in Virginia Jones 1984 IVF in Lagos, Nigeria Ashiru & Giwa-Osagie – 1986/1989 1992 ICSI Jacques Cohen LANDMARKS IN PGD Jacobson CB & Barter RH 1960 - 1st prenatal diagnosis Saiki et al 1985 – PCR Alan Handyside and Robert Winston 1989 – Embryo Biopsy Verlinsky Y et al 1999 – Polar body testing LANDMARKS IN PGD 1990: Sexing Human Preimplantation Embryo, & Polar Body Analysis for Mendelian Disease; - First PGD Baby Born 1993: FISH Analysis for Sexing and for PGD of Aneuploidies 1996: PGD for Chromosomal Translocations 1999: PGD for Late Onset Common Disorders with Genetic Predisposition LANDMARKS IN PGD 2000: Pre-implantation HLA Typing 2002: 1st Thousand PGD Babies Born 2002 onwards: Many technological developments CGH, aCGH, NGS, SNP,qPCR TYPES OF PREIMPLANTATION GENETIC TESTING Preimplantation Genetic Diagnosis (PGD) – Targeted genetic testing of known genetic abnormality in the couple: Single gene disorder Structural chromosomal abnormality TYPES OF PREIMPLANTATION GENETIC TESTING Preimplantation Genetic Screening for Aneuploidy (PGS) – Screening for numerical abnormality of select chromosomes: 13, 18, 21, X, Y, etc Preimplantation Gender Determination or Gender Selection INDICATIONS FOR PGD Autosomal dominant disorders – One of the couple is a carrier of the genetic defect (e.g., Huntington Disease or dwarfism) Autosomal recessive disorders: – Both couple are carriers of the genetic defect (e.g., Sickle Cell Disease) INDICATIONS FOR PGD X-linked disorder – One of the couple is a carrier of a x-linked genetic defect (e.g., Hemophilia) Human leukocyte antigen (HLA) matching Structural chromosomal abnormality – One of the couple is a carrier of a chromosome abnormality (translocation, inversion, deletion, insertion) INDICATIONS FOR PGS (ANEUPLOIDY SCREENING) Advanced maternal age Recurrent pregnancy loss Repeated IVF failure Severe male factor infertility INDICATIONS FOR GENDER SELECTION One of the couple is a carrier of x-link disorder Family balancing BIOPSY TECHNIQUES • • • May be largely diagnostic No paternal/meiosis II/post-zygotic information May require later follow-up testing (d3/d5) • Accounts for meiosis II errors • No paternal/post-zygotic information • Most expensive option diagnostically • Biopsy most detrimental • Chromosomal mosaicism common • Associated with Low implantation • • • • • Highly recommended 3-10 (?) cells (high accuracy/reliability) Significance of mosaicism? Least expensive per patient (diagnosis) May require vitrification BIOPSY OBJECTIVE Methods of Genetic Analysis: PGD single gene disorder – – – – Whole genome amplification Polymerase chain reaction (PCR) Direct detection of specific mutation Genotyping of linked polymorphic markers to ensure: Bi-parental inheritance Infer the inheritance of normal copy of the gene (Allele) Methods of Genetic Analysis: Structural chromosomal abnormality Fluorescent in situ hybridization (FISH) PCR-based testing – – Whole genome amplification Genotyping of linked polymorphic markers METHODS FISH Gender Determination Orange: X chromosome Green: Y chromosome FISH Aneuploidy Screening Normal Male Trisomy 21 Male Normal Female Red: chromosome 13 Cyan: chromosome 18 Green: chromosome 21 Purple: chromosome X Yellow: chromosome Y ARRAY COMPARATIVE GENOMIC HYBRIDIZATION (aCGH) • A molecular cytogenetic technique • Detects 'copy number changes' of chromosomes on a genome • Embryo DNA is compared with a known normal DNA specimen utilising thousands of specific genetic markers • More accurate results (fewer false normal or false abnormal results) • Error rate of 2% compared to 5 - 10% of FISH aCGH versus Conventional CGH • Better resolution (as low as 5 – 10 kilobytes of DNA sequences) • Cloned DNA fragments with exact chromosomal location replaces reference metaphase spread • Better detection of aberrations • Better identification of micro deletions and duplications • Increases the possibility of mapping changes directly onto the genomic sequence ARRAY COMPARATIVE GENOMIC HYBRIDIZATION (aCGH) - METHOD Same principle as conventional CGH Labelling of DNAs (control and test) with 2 different 'flurophores' (green and red) Green flurophore (cyanine 3) for test / patient and red flurophore (cyanine 5) for control / reference are used as 'probes' Competitive cohybridisation of probes onto nuclei acid targets (cloned genomic fragments (BACs / plasmids), cDNAs or oligonucleotides) with thousands of genetic markers. Digital imaging / specialised microscopes to capture and quantify fluorescence intensities of hybridised flurophores. ARRAY COMPARATIVE GENOMIC HYBRIDIZATION (aCGH) – RESULT Interpretation of results. Ratio of the fluorescence intensities is proportional to the ratio of 'copy number of DNA sequences' in the test and reference genomes. Altered Cy 3 : Cy 5 ratio indicates a loss or gain of the patient DNA at that specific genomic region If Cy 3 : Cy 5 ratio is equal on one probe (equal intensities of the flurophores), patient's genome is interpreted as having equal quantity of DNA as in the reference sample. ARRAY COMPARATIVE GENOMIC HYBRIDIZATION (aCGH) Array CGH remains the ‘gold standard’ for PGS with the greatest clinical experience to date RCTs show great promise for PGS Numerous RCTs are ongoing Relatively easy to set up Use of a reference laboratory recommended as a first step DETECTION OF WHOLE CHROMOSOME ANEUPLOIDIES IN SINGLE CELLS Trisomy 13 male (47,XY,+13) Sequencing Results 24Sure Results PGD/PGS SINCE 2009 (MART DATA) Chromosomes Aneuploidy (CA) Single gene disorders (Sickle cell – SC) Family balancing (sex selection – FB) So far over 75 cases have been done SUMMARY OF REASONS FOR PGD/PGS (N=75) 45% 43% 40% 35% 32% 30% 23% 25% 20% 15% 10% 5% 0% CA FB SC SUMMARY FOR PGS (N= 32) SUMMARY OF EMBRYO BIOPSED FOR CHROMOSOMES ANEUPLIODY (N=300) 59% 60% 50% 40% 30% 59% of embryos biopsied were abnormal 15% No result rate 26% 20% 15% 10% 0% Normal Abnormal No Result ? Number and quality of cells tested SICKLE CELL DIAGNOSIS (PGD) N = 17 SUMMARY OF EMBRO BIOPSED FOR SICKLE CELL DIAGNOSIS (N= 133) 50% 41% 34% 40% 25% 30% 20% 10% RESULT… 0% Normal Abnormal No Result 41% of biopsied embryos were homozygous and heterozygous normal 34% of embryos undiagnosed FAMILY BALANCING – SEX SELECTION (N=26) SUMMARY OF GENDER ANALYSIS IN FAMILY BALANCING 100% 100% 90% 80% 70% 60% 47% 47% 50% 40% 30% 20% 10% 0% Male Total Female No difference in the proportion of male and female embryos SUMMARY OF PREGNANCY RATE 70% 59% 60% 50% 40% 32% 30% 20% 9% 10% 0% CA FB SC SUMMARY OF CLINICAL PREGANCY RATE 35 35 30 30 25 20 15 9 10 5 0 CA (%) FB (%) Clinical Rate SS (%) SETTING UP A PGD LAB Two ways IVF centre and PGD centre in the same institute – preferred Transport PGD ORGANIZATION OF THE PGS/PGD CENTRE Highly successful IVF unit Patients need genetic and specific PGD counselling Biopsy performed by trained embryologist Diagnosis performed by molecular biologist/cytogeneticist Patient information leaflets and consents Excellent communication between IVF centre and diagnosis lab Join the PGD Consortium WHAT MAKES A GOOD PGD CENTRE COMMUNICATION Excellent IVF Platform Excellent Diagnostics Laboratory Integration of Services Commitment to Follow-up TRANSPORT PGD FUTURE OF PGD/PGS • PGD – New technology to allow diagnosis of more disorders – Whole genome amplification – SNP microarrays, array-CGH, quantitative real time PCR (qPCR) – Blastocyst biopsy and vitrification FUTURE OF PGD/PGS • PGS – Randomised controlled trials to see if valid procedure with clinical significance – NOT cleavage stage biopsy, NOT FISH – Try polar body or trophectoderm biopsy – SNP, array-CGH, or sequencing BEETHOVEN “I RECOGNIZE NO SUPERIORITY IN MANKIND OTHER THAN GOODNESS.” THE TEAM THANK YOU FOR LISTENING!