Biology Study Guide for Section (Macromolecules) Test
... Energy-The main function of carbohydrates in animal cells. Sugars- Similar to starch, cellulose is a complex carbohydrate. This compound makes up cellulose. Nitrogen- Carbohydrates contain all C,H,O but no nitrogen. Amino acids are the building blocks of protein. Glucose-is also called a monosacchar ...
... Energy-The main function of carbohydrates in animal cells. Sugars- Similar to starch, cellulose is a complex carbohydrate. This compound makes up cellulose. Nitrogen- Carbohydrates contain all C,H,O but no nitrogen. Amino acids are the building blocks of protein. Glucose-is also called a monosacchar ...
Biomolecules Fill in the crossword puzzle by using
... is a polysaccharide made of glucose units hooked together found in plant ...
... is a polysaccharide made of glucose units hooked together found in plant ...
Worksheet Answer Key
... polypeptide chain. Can be alpha helix or beta sheet. Proteins whose shape are dominated by this interaction are called fibrous. ...
... polypeptide chain. Can be alpha helix or beta sheet. Proteins whose shape are dominated by this interaction are called fibrous. ...
Chemical Compounds in Cells and in Our Food
... Structure of Proteins • Made of amino acids – There are 20 different amino acids in living things • These amino acids link together to form a large molecule of 50-3000 amino acids in one protein. • Change one aa, change whole protein ...
... Structure of Proteins • Made of amino acids – There are 20 different amino acids in living things • These amino acids link together to form a large molecule of 50-3000 amino acids in one protein. • Change one aa, change whole protein ...
Macromolecules Notes Macromolecules Notes
... • Made up of individual “building blocks” called MONOMERS Macromolecule #1 Carbohydrates ...
... • Made up of individual “building blocks” called MONOMERS Macromolecule #1 Carbohydrates ...
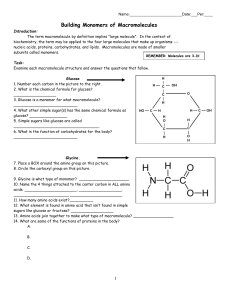
Building Monomers of Macromolecules
... Building Monomers of Macromolecules Introduction: The term macromolecule by definition implies "large molecule". In the context of biochemistry, the term may be applied to the four large molecules that make up organisms --nucleic acids, proteins, carbohydrates, and lipids. Macromolecules are made of ...
... Building Monomers of Macromolecules Introduction: The term macromolecule by definition implies "large molecule". In the context of biochemistry, the term may be applied to the four large molecules that make up organisms --nucleic acids, proteins, carbohydrates, and lipids. Macromolecules are made of ...
Organic Molecules - Mr. Swords` Classes
... - A key source of energy for the body. -They turn into energy for all cells. ...
... - A key source of energy for the body. -They turn into energy for all cells. ...
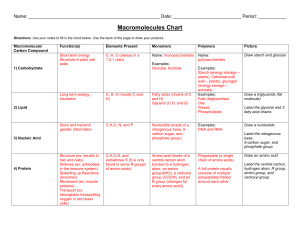
macromolecule_sheets
... 2. How does the number of H atoms in a carbohydrate compare to the number of oxygen atoms? 3. What is the simplest type of carbohydrate? 4. What two functional groups are found in monosaccharides? 5. Name three common monosaccharides and give the molecular formula for each. 6. What suffix is commonl ...
... 2. How does the number of H atoms in a carbohydrate compare to the number of oxygen atoms? 3. What is the simplest type of carbohydrate? 4. What two functional groups are found in monosaccharides? 5. Name three common monosaccharides and give the molecular formula for each. 6. What suffix is commonl ...
Vocabulary review
... b. stores energy in its bonds c. compounds made primarily of carbon atoms d. process used to break down a polymer e. building blocks of DNA f. cluster of atoms that influences the characteristics of a molecule g. building blocks of protein h. place where a substrate fits into an enzyme ...
... b. stores energy in its bonds c. compounds made primarily of carbon atoms d. process used to break down a polymer e. building blocks of DNA f. cluster of atoms that influences the characteristics of a molecule g. building blocks of protein h. place where a substrate fits into an enzyme ...
File
... • Key function to store energy, make up cell membranes, insulate/protect/ and make hormones • Lipid= 3 fatty acids (long chain of carbons with hydrogen atoms attached) AND glycerol (alcohol with 3 carbon atoms) • Commonly called fats and oils ...
... • Key function to store energy, make up cell membranes, insulate/protect/ and make hormones • Lipid= 3 fatty acids (long chain of carbons with hydrogen atoms attached) AND glycerol (alcohol with 3 carbon atoms) • Commonly called fats and oils ...
Name - MsOttoliniBiology
... in the immune system) Speeding up Reactions (enzymes) Movement (ex: muscle proteins) Transport (ex: hemoglobin transporting oxygen in red blood cells) ...
... in the immune system) Speeding up Reactions (enzymes) Movement (ex: muscle proteins) Transport (ex: hemoglobin transporting oxygen in red blood cells) ...
CHM 105 - Test 3 Review
... 5. To which of the four classes of amino acid does cysteine belong? 6. Draw the tripeptide: alanine–lysine–aspartic acid. What type of bond links the amino acids? Draw an arrow to this bond. 7. What are the four levels of protein structure? What level is given for the tripeptide above? 8. What is de ...
... 5. To which of the four classes of amino acid does cysteine belong? 6. Draw the tripeptide: alanine–lysine–aspartic acid. What type of bond links the amino acids? Draw an arrow to this bond. 7. What are the four levels of protein structure? What level is given for the tripeptide above? 8. What is de ...
Macromolecules and Enzymes
... • When the circumstances are just right, enzymes go to work and the substrates bind ...
... • When the circumstances are just right, enzymes go to work and the substrates bind ...
Unit 3 Biochemistry - The Naked Science Society
... fibers for plant structures. Humans can’t digest (fiber). Most abundant organic molecule. Chitin—glucose polymer for exoskeletons of some crustaceans & insects. ...
... fibers for plant structures. Humans can’t digest (fiber). Most abundant organic molecule. Chitin—glucose polymer for exoskeletons of some crustaceans & insects. ...
Biological Molecules
... cellulose Starch (plants) and glycogen (animals) are formed so as not to interfere with cell chemistry and osmosis. They can be quickly broken down to glucose and used as energy. (hydrolysis) ...
... cellulose Starch (plants) and glycogen (animals) are formed so as not to interfere with cell chemistry and osmosis. They can be quickly broken down to glucose and used as energy. (hydrolysis) ...
CH 2.3-Carbon Compounds
... More Carbohydrates - Polysaccharides - Also called complex carbohydrates - Made of many linked monosaccharides - Can be broken down to release energy - Can be used for structure ...
... More Carbohydrates - Polysaccharides - Also called complex carbohydrates - Made of many linked monosaccharides - Can be broken down to release energy - Can be used for structure ...
Organic Molecule Notes
... -used for… -ex: Maltose (Sugar used in alcohol) =1 glucose+1 glucose --used for… -ex: Lactose (Milk sugar) =1 glucose+1galactose -produced by animals Long term Energy Storage Molecules Starch=primary storage form of energy in plants ...
... -used for… -ex: Maltose (Sugar used in alcohol) =1 glucose+1 glucose --used for… -ex: Lactose (Milk sugar) =1 glucose+1galactose -produced by animals Long term Energy Storage Molecules Starch=primary storage form of energy in plants ...
Chapter 2
... • Solutions above 7 are called basic, because they have less H+ ions than pure water ...
... • Solutions above 7 are called basic, because they have less H+ ions than pure water ...
Cell Building Blocks
... The simplest carbohydrates are monosaccharides, which are small straight-chain aldehydes and ketones with many hyçjroxyl groups addcd, usually one on each carbon except the functional group. Other carbohydrates are composed of monosaccharide units and break down under hydrolysis. These may be classi ...
... The simplest carbohydrates are monosaccharides, which are small straight-chain aldehydes and ketones with many hyçjroxyl groups addcd, usually one on each carbon except the functional group. Other carbohydrates are composed of monosaccharide units and break down under hydrolysis. These may be classi ...
Carbon compounds - Sonoma Valley High School
... compounds that can be bonded together to form larger ones. • 3 important ones are: – sugars – amino acids ...
... compounds that can be bonded together to form larger ones. • 3 important ones are: – sugars – amino acids ...
File - Principles of Biology 103
... 11. The nonadjacent regions that form to create specific domains is termed: C. Tertiary structure 12. A nucleotide contains: A. A five carbon ring bonded to a nitrogen base and a phosphate group(s) 13. The breakdown of large molecules by the enzymatic addition of water is an example of what kind of ...
... 11. The nonadjacent regions that form to create specific domains is termed: C. Tertiary structure 12. A nucleotide contains: A. A five carbon ring bonded to a nitrogen base and a phosphate group(s) 13. The breakdown of large molecules by the enzymatic addition of water is an example of what kind of ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.