BIO 6.3 Carbon - Steinbach Science
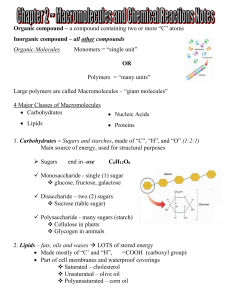
... The largest carbs are polysaccharides, polymers composed of many monosaccharide subunits (e.g., starch, glycogen, cellulose) Lipids are organic compounds that have a large portion (much greater than 2 to 1) ...
... The largest carbs are polysaccharides, polymers composed of many monosaccharide subunits (e.g., starch, glycogen, cellulose) Lipids are organic compounds that have a large portion (much greater than 2 to 1) ...
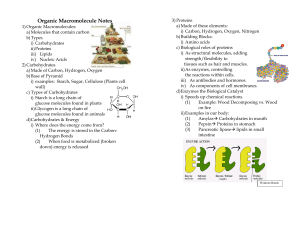
Ex. glucose, fructose and galactose: these are isomers
... B. Polypeptides: very long chains of amino acids. The amino acids in the chains interact with each other, forming different types of structures: 1.__________________________ 2.__________________________ 3.__________________________ C. The ___________________of a protein is greatly influenced by cond ...
... B. Polypeptides: very long chains of amino acids. The amino acids in the chains interact with each other, forming different types of structures: 1.__________________________ 2.__________________________ 3.__________________________ C. The ___________________of a protein is greatly influenced by cond ...
Slide 1 - Denton ISD
... Thymine. In RNA Uracil replaces Thymine. structurally similar to fats but a phosphate group replaces one of the fatty acid chains, thus giving this molecule a POLAR and NONPOLAR region. Structurally form two layers to create the plasma membrane vital to all cells. ...
... Thymine. In RNA Uracil replaces Thymine. structurally similar to fats but a phosphate group replaces one of the fatty acid chains, thus giving this molecule a POLAR and NONPOLAR region. Structurally form two layers to create the plasma membrane vital to all cells. ...
Lecture 3 (BY 14)
... • Breaks polymers into smaller units • Enzymes split molecules into two or more parts • An -OH group and an H atom derived from water are attached at exposed sites ...
... • Breaks polymers into smaller units • Enzymes split molecules into two or more parts • An -OH group and an H atom derived from water are attached at exposed sites ...
Organic Compounds
... Unsaturated Fatty Acids Liquids at room temperature Carbons have double bonds, therefore they are not saturated with Hydrogen atoms Examples: - Olive Oil - Corn Oil ...
... Unsaturated Fatty Acids Liquids at room temperature Carbons have double bonds, therefore they are not saturated with Hydrogen atoms Examples: - Olive Oil - Corn Oil ...
Carbon Compounds
... Made of monomers called AMINO ACIDS Amino Acids are joined by peptide bonds, therefore another name for a protein is POLYPEPTIDE There are 4 levels of structural organization ...
... Made of monomers called AMINO ACIDS Amino Acids are joined by peptide bonds, therefore another name for a protein is POLYPEPTIDE There are 4 levels of structural organization ...
Organic compounds Carbon compounds are also called organic
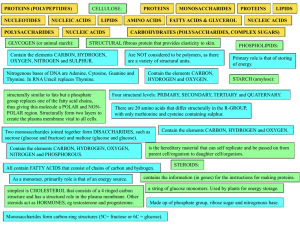
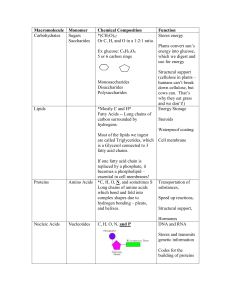
... very large and made up of smaller building blocks or sub-units. Four groups of organic molecules that are related to the survival of living things are; carbohydrates, lipids, nucleic acids and proteins. Carbohydrates are compounds made up of carbon, hydrogen and oxygen atoms in a 1:2:1 ratio. They a ...
... very large and made up of smaller building blocks or sub-units. Four groups of organic molecules that are related to the survival of living things are; carbohydrates, lipids, nucleic acids and proteins. Carbohydrates are compounds made up of carbon, hydrogen and oxygen atoms in a 1:2:1 ratio. They a ...
Carbon compounds class web14
... each of the 4 macromolecules. Put the name of the macromolecule in the first column, the monomers it is composed of in the second, and the function in the cell in the third. ...
... each of the 4 macromolecules. Put the name of the macromolecule in the first column, the monomers it is composed of in the second, and the function in the cell in the third. ...
NATURE`S BIOLOGICAL BUILDING BLOCKS
... Composition: mostly carbon and hydrogen with some oxygen Includes fatty, greasy and waxy compounds relatively insoluble in water, but soluble in organic solvents such as ether and benzene. Neutral Fats - high energy molecules consisting of glycerol and fatty acids. Phospholipids - neutral fat with o ...
... Composition: mostly carbon and hydrogen with some oxygen Includes fatty, greasy and waxy compounds relatively insoluble in water, but soluble in organic solvents such as ether and benzene. Neutral Fats - high energy molecules consisting of glycerol and fatty acids. Phospholipids - neutral fat with o ...
The Mac Daddies of Molecules
... Made from monosaccharides (single sugars) which form polysaccharides (many sugars) Examples: glucose, fructose, starch, sugars We get energy from carbs! ...
... Made from monosaccharides (single sugars) which form polysaccharides (many sugars) Examples: glucose, fructose, starch, sugars We get energy from carbs! ...
Organic Compounds Overview - Kenwood Academy High School
... • In animals: – Glucose – Glycogen (a polysaccharide that provides longer energy storage.) – Chitin (compound in exoskeletons) ...
... • In animals: – Glucose – Glycogen (a polysaccharide that provides longer energy storage.) – Chitin (compound in exoskeletons) ...
Organic Macromolecule Notes
... a) Made of these elements: i) Carbon, Hydrogen, Oxygen, Nitrogen b) Building Blocks: i) Amino acids c) Biological roles of proteins i) As structural molecules, adding strength/flexibility to tissues such as hair and muscles. ii) As enzymes, controlling the reactions within cells. iii) As antibodies ...
... a) Made of these elements: i) Carbon, Hydrogen, Oxygen, Nitrogen b) Building Blocks: i) Amino acids c) Biological roles of proteins i) As structural molecules, adding strength/flexibility to tissues such as hair and muscles. ii) As enzymes, controlling the reactions within cells. iii) As antibodies ...
biochem study guide
... coupled reactions cytosine dehydration synthesis denatured proteins disaccharide disulfide bond ...
... coupled reactions cytosine dehydration synthesis denatured proteins disaccharide disulfide bond ...
Carbon-Based Molecules
... These are nonpolar molecules that includes fats, oils, and cholesterol. Can be used for some energy uses, some are used for cell structure. Fatty acids – chains of carbon/hydrogen atoms. ...
... These are nonpolar molecules that includes fats, oils, and cholesterol. Can be used for some energy uses, some are used for cell structure. Fatty acids – chains of carbon/hydrogen atoms. ...
Chapter 3: Biochemistry
... is released b) energy is used by the cell to drive chemical reactions II. Molecules of Life A. Carbohydrates 1. Function: 2. Monomer: Monosaccharides: a) b) ...
... is released b) energy is used by the cell to drive chemical reactions II. Molecules of Life A. Carbohydrates 1. Function: 2. Monomer: Monosaccharides: a) b) ...
Biochemistry File - Northwest ISD Moodle
... 4. Proteins – polymers of amino acids joined by peptide bonds Used to build cells, transport molecules, and control the rate of reactions Made of “C”, “H”, “O”, and “N” 20 different amino acids ...
... 4. Proteins – polymers of amino acids joined by peptide bonds Used to build cells, transport molecules, and control the rate of reactions Made of “C”, “H”, “O”, and “N” 20 different amino acids ...
2.3 Outline
... • Simple sugars such as glucose are a major source of energy in cells. • _________________________ are double sugars formed when two _________________________ are joined. • Sucrose, or common table sugar, is a disaccharide that consists of both glucose and fructose. • _________________________ such ...
... • Simple sugars such as glucose are a major source of energy in cells. • _________________________ are double sugars formed when two _________________________ are joined. • Sucrose, or common table sugar, is a disaccharide that consists of both glucose and fructose. • _________________________ such ...
Macromolecule
... Stores energy Or C, H, and O in a 1:2:1 ratio. Plants convert sun’s Ex glucose: C6H12O6 energy into glucose, 5 or 6 carbon rings which we digest and use for energy ...
... Stores energy Or C, H, and O in a 1:2:1 ratio. Plants convert sun’s Ex glucose: C6H12O6 energy into glucose, 5 or 6 carbon rings which we digest and use for energy ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.