* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Brachmann et al., 2005 Mol Cell Biol. 25, 2593
Survey
Document related concepts
Cytokinesis wikipedia , lookup
Endomembrane system wikipedia , lookup
Biochemical switches in the cell cycle wikipedia , lookup
Phosphorylation wikipedia , lookup
List of types of proteins wikipedia , lookup
Tyrosine kinase wikipedia , lookup
Purinergic signalling wikipedia , lookup
NMDA receptor wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Paracrine signalling wikipedia , lookup
Cannabinoid receptor type 1 wikipedia , lookup
Transcript
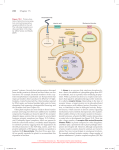
1. Fibroblast Chemotaxis: more about positive feedback loops. 2. Autoregulatory Mechanisms of Eukaryotic Chemotaxis System Components: Receptors, Gproteins, GEFs, PI3K, Kinases, phosphatases. How evolution has selected for components with autoregulation and integral feedback control. Fibroblasts chemotax toward growth factors 0 3 hrs 8 hrs QuickTime™ and a Photo - JPEG decompressor are needed to see this picture. 12 hrs 21 hrs PDGF-stimulated wound healing in mouse embryo fibroblasts PI3K p110 Family Members Class Ia 110 a p85 Ras binding binding C2 PIK 110 b 110 d Tissue Regulation Kinase All Tyr Kinase Tyr Kinase All + b/g Blood Tyr Kinase Cells Class Ib 110 g Blood Cells b/g Deletion of Class Ia PI3K genes in mouse embryo fibroblasts impairs PDGF-dependent cell migration. Brachmann et al., 2005 Mol. Cell. Biol. 25, 2593. 0 3 hrs 8 hrs QuickTime™ and a Photo - JPEG decompressor are needed to see this picture. 12 hrs 21 hrs WT PI3K-Ia Deletion Woundhealing 10ng/ml PDGF 80 3h Migrated Cells 70 8h 15h 21h 60 50 Ly294003 PI3K inhibitor 40 30 20 10 0 Wild type PI3K Ia deletion P85a-/-;p85b-/- Defect in PDGF-induced lamellipodia formation in MEFs defective in class Ia PI3K Brachmann et al., 2005 Mol Cell Biol 25, 2593 unstimulated Wild Type PI3K Ia deletion PDGF PDGF + WM Class Ia PI3K has multiple domains for signal input, allowing it to act as an ‘AND GATE’ or possibly an ‘OR GATE’ GTP CDC42 SH3 Cdc42 binding GPCR Membrane Tyr Kinase GTP bg P-Tyr P-Tyr Ras SH2 SH2 Ras C2 PIK Catalytic Binding p85 regulatory Class Ia PI 3-Kinase p110 catalytic p110b can also be activated by bg subunits of G proteins, but only when bound to a phosphoTyr protein (AND GATE). Growth Factor Class Ia PI3K mediates growth factordependent cortical actin formation Receptor Tyr Kinase P-Tyr Ras GTP p85 p110 SH2 PI3K PIP3 PH AKT PIP2 PIP3 Rac GTP GEF? Rac PTE N Cortical Actin Cell Migration Deletion of class Ia PI3K genes appears to impair (but not eliminate) Ras activation (as judged by impaired activation of the downstream protein kinase, Erk) Brachmann et al., 2005 Mol Cell Biol. 25, 2593 5 min PDGF [ng/ml]: 0 1 3 10 0 1 3 10 Erk-P Erk Wild Type MEFs PI3K Ia deleted Thus, as in Dictyostelium, there appears to be a positive feedback loop between PI3K and Ras in fibroblasts. Reduced PDGF-induced Rac activation in MEFs lacking class Ia PI3K Brachmann et al., 2005 Mol Cell Biol. 25, 2593 Control PDGF: - + Double KO - + Rac GTP GST-CRIB pulldown Rac p85a Erk-P Erk Overexpression of a Rac GEF (Vav2) induces lamellipodia formation in MEFs lacking Class Ia PI3K Brachmann et al., 2005 Mol Cell Biol. 25, 2593 Wild Type PI3K Ia deleted RhodaminePhalloidin (Actin) Vav2 PI3K is involved in both local Ras and local Rac positive feedback loops PDGF Receptor p85 Ras GTP ? p110 PIP3 + ? + Rac GTP PH GEF Conclusions 1. Growth Factor Receptors stimulate Class Ia PI3K through PhosphoTyr residues of receptors binding to SH2 domains, while GPCRs stimulate Class Ib PI3K through bg subunits binding to the catalytic subunit. 2. In both cases, PI-3,4,5-P3 is in a local positive feedback amplification loop involving Rac (and Ras?) that allows nonisotrophic localization of cortical actin, providing directionality to chemotaxis. How is perfect adaptation achieved in eukaryotic chemotaxis? Shutoff mechanisms must exist to adapt the system to a given level of stimulation, allowing a temporal increase in receptor stimulation to be sensed. The adaptation should be slow compared to the stimulation to insure significant directional migration prior to adaptation. What is known about shutoff mechanisms of GPCRs and Receptor Tyr Kinases? GPCR ACTIVATION Hormone Receptor a GDP bg Receptor a bg GDP GPCR ACTIVATION attractant Receptor a GDP Ligand-induced Conformational Change <100 msec msec to sec Receptor bg a GDP bg Receptor a bg GTP Receptor bg Effector 2 GTP PI 3-kinase etc. a GTP Effector 1 (Phospholipase C, etc.) Signal Termination, Downregulation and Reset to Basal State Receptor bg Effector 2 RGS Seconds a GTP Effector 1 Minutes a GDP Signal Termination, Downregulation and Reset to Basal State Receptor bg G-Receptor Kinase (GRK) RGS Seconds a GTP Effector 1 Minutes a GDP Signal Termination and Reset to Basal State Inactive Receptor PPP Receptor PPP bg Arrestin Phosphatase G-Receptor Kinase (GRK) Seconds Effector 1 Receptor a bg GDP Dephosphorylation And rebinding of Ga and bg. (minutes) RGS a GTP Basal State Minutes a GDP Only activated receptors are phosphorylated and downregulated. This effect is slow (minutes) compared to activation (seconds). During this perturbation from steady state, PI3K activation occurs, driving directional motility. Integral Feedback Control Analogous to model in Yi, Huang, Simon&Doyle 2000 PNAS 97, 4649 If we assume that only activated receptors are phosphorylated (and thus inactivated) and that the phosphatase that dephosphorylates the GPCR operates at saturation and is less active than the G-protein Receptor Kinase (GRK), then the model is analogous to integral control of bacterial chemotaxis receptors. Inhibition of active chemotaxis receptors by demethylation is analogous to inactivation of active GPCRs by phosphorylation. This is a consequence of the fact that GRKs only phosphorylate receptors associated with active bg proteins. The rate of receptor phosphorylation is: dRP/dt = VPmax - VKmax(A)/(KK+A) (where A is the concentration of activated receptors, KK is the KM of the GRK for activated receptors, VPmax is the maximal activity of the phosphatase and VKmax is the maximal activity of the kinase, GRK ). Thus, the activity at steady state will be: Ast= KKVPmax/(VKmax-VPmax) This is the set point (y0 in the model above). y is defined as the difference between the activity at time t (y1) and the activity at steady state (y0). Thus, at steady state, y = 0. Increased ligand binding acutely increases u and elevates y1 to a value above y0, giving a transient positive value for y (resulting in PI3K activation). At steady state, (y = 0) the rate of phosphorylation and dephosphorylation are equal. If one assumes that GRK only acts on active receptors (whether or not ligand is bound) then the net rate of phosphorylation at any instantaneous time will be directly proportional to y (the transient excess in active receptors over the steady state value). When y = 0 phosphorylation and dephosphorylaiton cancel out. The fraction of phosphorylated receptors (x) at any time t is then determined by the number of receptors in the phosphorylated state at time zero, x0 (e.g. prior to the perturbation due to increased ligand binding) plus the number of receptors that get phosphorylated during the interval in which the system was perturbed. This latter term is the integral from the time at which the perturbation (e.g. ligand unbinding) occurred t=0 to time t of ydt. So x(t) = x0 + t ydt 0 Notice that y can be + or - depending on whether ligand decreases or increases. Thus dx/dt = y = k(u-x) - y0 At steady state, dx/dt=y=0 and y1=y0 Notice that since k and y0 are constants, an increase in u (rapid binding of ligand) is ultimately offset by a slow decrease in x so that at steady state k(u-x) = y0. P-Tyr Tyr-P Kinase Tyr-P SH2 Kinase Kinase Regulation of protein-Tyr kinases P-Tyr PI3K Autopho-transphorylation of low activity monomeric protein kinases in the ligand-induced dimer stabilizes the active state of each monomer, allowing further transphosphorylation at sites that recruit signaling proteins. P-Tyr Kinase Kinase Tyr-P SHP2 SH2 Tyr-P P-Tyr SH2 containing phosphoTyr phosphatases (e.g. SHP2) are preferentially recruited to activated receptors and play a dual role of transmitting additional signals (Ras activation) and turning off receptors. INSULIN RECEPTOR CATALTIC DOMAIN (INACTIVE) ATP Pocket Tyr 1158 Tyr 1162 Tyr 1163 Prior to stimulation, protein-Tyr kinases have floppy activation loops (region containing Tyr 1157, 1162 and 1163 of the insulin receptor). As a consequence the enzyme has a low probability of being in the active conformation (~1%). Despite this low activity, when brought in proximity with a another low activity Tyr kinase (due to growth factor binding), crossphosphorylation of respective activation loops can occur. Phosphorylation of the residues on this loop stabilizes the active conformation of the protein giving a ~100 fold increase in activity. Activated Insulin Receptor Peptide substrate Integral Control of Receptor Protein-Tyr Kinases The preferential dephosphorylation of activated Protein-Tyr kinases by SH2containing phosphatases provides a potential mechanism for integral control. In response to an acute elevation in the level of ligand, the receptor will be rapidly activated, but in the continuous presence of the ligand, the phosphatase will ultimately return the kinase to a steady state activity that is determined by the affinity of the phosphatase for the activated kinase, the Vmax of the phosphatase and the Vmax of the kinase for transphosphorylation. Analogous to the set point for bacterial chemotaxis receptors one can show that: Ast = KM-SHP2VKinmax/(VSHP2max - VKinmax) This simplified system does not reset to the same steady state as prior to receptor stimulation since VKinmax is dependent on receptor ligation. Modeling predicts an overshoot followed by return to a steady state that depends on ligand occupation. This is in agreement with observations at intermediate times (0 to 30 min.) following PDGF stimulation Exclusive ubiquitinylation, of activated protein-Tyr kinases (due to SH2-containing E3 ligases (e.g cbl)), leads to receptor internalization, providing a second mechanism of longer term shut-off that also models as integral feedback control. Integral Control of PI3K PI3K, when activated, phosphorylates lipids at a high rate but also autophosphorylates (on regulatory and catalytic subunits) at a slow rate, leading to inactivation. Assuming that the phosphatase that dephosphorylates PI3K is saturated by substrate, this could also lead to integral control of this enzyme. Parallels between low molecular weight G protein (Ras, Rac Rho) regulation and heterotrimeric G protein regulation GDP/GTP Exchange Factor (GEF) activate: analogous to GPCR GEF (SOS) basal Ras-GDP GTPase Activating Protein Analogous to RGS slow GAP Ras-GTP Effector Effectors such as Raf (Ser/Thr kinase) or PI3K bind to activated Ras Heterotrimeric and low molecular weight GTP binding proteins have been retained and expanded during evolution because they have unstable activated states and can spontaneously return to inactive states. Inactivation can also be accelerated by GAPs. Signal Transduction in Eukaryotic cells is usually initiated by recruitment of signaling proteins to the plasma membrane. We have discussed three major mechanisms for acute and reversible protein relocation in response to cell stimulation. These mechanisms have the potential to amplify small signals. More importantly, recruiting signaling proteins from a 3-dimensional space (cytosol) to a 2-dimensional space (membrane) provides a mechanism for facilitating unfavorable multimeric interactions. Protein phosphorylation to create docking site Membrane Tyr P-Tyr Activation of GTP-binding proteins Membrane GDP-Ras GTP-Ras Generation of lipid second messengers Membrane PI-4,5-P2 PI-3,4,5-P3 PH SH2 Ras GEF Stochiometric PI3K Small Amplification AKT Large Amplification Sarraste Plasma Membrane 1 1 5 4 3 R28 3 The PH domain of BTK binds the head group of PI-3,4,5-P3 and crystallizes as a dimer with the two binding pockets on the same surface. However, in solution, it behaves as a monomer. Moving signaling proteins from the three dimensional environment of the cytosol to the two dimensional environment of the plasma membrane decreases the entropy difference between a monomeric and dimeric state. Many signaling proteins may have evolved very weak free energies of homo or hetero-dimerization to insure that dimerization only occurs when confined on a two dimensional surface. Signal Dimers Monomers Membrane Pólya's Random Walk Constants http://mathworld.wolfram.com/PolyasRandomWalkConstants.html Let p(d) be the probability that a random walk on a d-D lattice returns to the origin. Pólya (1921) proved that (1) p(1) = 1; p(2) = 1 but (2) p(d) < 1 for d > 2. Watson (1939), McCrea and Whipple (1940), Domb (1954), and Glasser and Zucker (1977) showed that (3) p(3) = 1 - 1/u(3) = 0.340537…. where u(3) = 3/(2p)3 p p p _____dxdydz______ -p -p -p 3-cosx-cosy-cosz Finch, S. R. "Pólya's Random Walk Constant." §5.9 in Mathematical Constants. Cambridge, England: Cambridge University Press, pp. 322331, 2003. Domb, C. "On Multiple Returns in the Random-Walk Problem." Proc. Cambridge Philos. Soc. 50, 586-591, 1954. Glasser, M. L. and Zucker, I. J. "Extended Watson Integrals for the Cubic Lattices." Proc. Nat. Acad. Sci. U.S.A. 74, 1800-1801, 1977. McCrea, W. H. and Whipple, F. J. W. "Random Paths in Two and Three Dimensions." Proc. Roy. Soc. Edinburgh 60, 281-298, 1940. Montroll, E. W. "Random Walks in Multidimensional Spaces, Especially on Periodic Lattices." J. SIAM 4, 241-260, 1956. Sloane, N. J. A. Sequences A086230, A086231, A086232, A086233, A086234, A086235, and A086236 in "The On-Line Encyclopedia of Integer Sequences." http://www.research.att.com/~njas/sequences/. Watson, G. N. "Three Triple Integrals." Quart. J. Math., Oxford Ser. 2 10, 266-276, 1939. Eric W. Weisstein. "Pólya's Random Walk Constants." From MathWorld--A Wolfram Web Resource. http://mathworld.wolfram.com/PolyasRandomWalkConstants.html Further websites for random walks http://www.rwc.uc.edu/koehler/biophys.2ed/java/walker.html http://www.krellinst.org/UCES/archive/modules/monte/node4.html