* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Proteins
Non-coding RNA wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Molecular evolution wikipedia , lookup
Western blot wikipedia , lookup
Peptide synthesis wikipedia , lookup
Protein moonlighting wikipedia , lookup
Protein (nutrient) wikipedia , lookup
Epitranscriptome wikipedia , lookup
Bottromycin wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Gene expression wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Circular dichroism wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Point mutation wikipedia , lookup
Metalloprotein wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Protein adsorption wikipedia , lookup
Genetic code wikipedia , lookup
Expanded genetic code wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Protein structure prediction wikipedia , lookup
List of types of proteins wikipedia , lookup
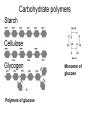
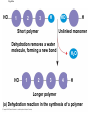
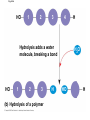
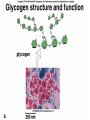
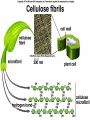
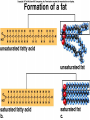
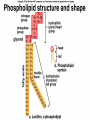
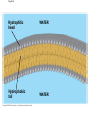
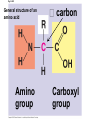
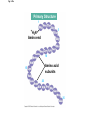
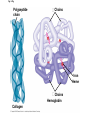
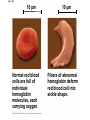
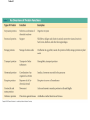
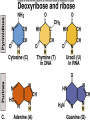
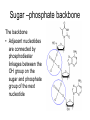
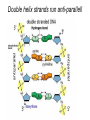
The Structure and Function of Macromolecules Part 2 Chapter 5 Emergent properties of macromolecules from smaller subunits • Within cells, small organic molecules are joined together to form larger molecules. • These large macromolecules may consist of thousands of covalently bonded atoms and weigh more than 100,000 daltons In this chapter we’ll study the structure and function of these macromolecules and their place in living organisms. Four major classes of macromolecules • • • • Carbohydrates Lipids Proteins Nucleic acids Most macromolecules are polymers, built from monomers • Carbohydrates, proteins, and nucleic acids are made of polymers, repeating subunits of smaller molecules called monomers. • Lipids are not polymers but they are macromolecules • Macromolecules - very large • Polymers - many (repeating) parts – Monomer - one unit Carbohydrate polymers Monomer of glucose Polymers of glucose Synthesis and digestion • Condensation/Dehydration reaction – synthesis - to build – Bonds monomers together – Release water molecule • Hydrolysis/Digestion – breaks down – hydrolysis (water breaking) – Adds water ions to the broken ends Condensation or dehydration reactions • Reaction that builds polymers from monomers by removing one molecule of water. • The cell uses energy to build polymers with the help of enzymes Fig. 5-2a HO 1 2 3 H Short polymer HO Unlinked monomer Dehydration removes a water molecule, forming a new bond HO 1 2 H 3 H2O 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer Hydrolysis/Digestion • Reverse reaction of Condensation • Enzymes help to speed up the reaction • Polymers are split by addition of water molecule • OH (Hydroxyl) is added to one monomer and a Hydrogen to the adjacent monomer. • Ex. In digestion large polymers are broken down and monomers are used to build new polymers needed by the body. Fig. 5-2b HO 1 2 3 4 Hydrolysis adds a water molecule, breaking a bond HO 1 2 3 (b) Hydrolysis of a polymer H H H2O HO H Carbohydrates……Sugars Monosaccharides have the molecular formula in multiples of CH2O • Glucose (C6H12O6) is the most common monosaccharide Monosaccharides are classified by 1. location of the carbonyl group – – Aldose –carbonyl is on the last carbon in the chain Ketose Carbonyl is located between two carbons 2. Number of carbons in the carbon skeleton Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings Fig. 5-3 Trioses (C3H6O3) Pentoses (C5H10O5) Hexoses (C6H12O6) Glyceraldehyde Ribose Glucose Galactose Dihydroxyacetone Ribulose Fructose Examples of Disaccharides Monosaccharide units Disaccharide Sources Maltose Germinating grains Used in brewing beer Milk, yogurt, ice cream glucose + glucose Sugar cane, sugar beets glucose + fructose Lactose Sucrose glucose + galactose Polysaccharides • Made of many monosaccharides joined by glycosidic linkages • The structure and function of a polysaccharide is determined by the monomers and the position of the glycosidic linkage Three important polysaccharides made of repeating units of glucose • Complex sugars - many sugar units • Starch – Glucose chain molecules – Energy storage in plants • Glycogen – Glucose chain molecule – Energy storage in animals • Cellulose – Glucose chain molecule – Structural molecule in plant cell walls Fig. 5-10 Chitin hard, insoluble... and yet somehow flexible (a) The structure of the chitin monomer. (b) Chitin forms the exoskeleton of arthropods. (c) Chitin is used to make a strong and flexible surgical thread. Chitin is polysaccharide Nacetylglucosamine (a natural derivative of glucose). Carbohydrate structural Isomers molecular formula C6H12O6 Lipids • Not polymers made of Glycerol molecule and 3 fatty acids called a triglyceride • Hydrophobic - Water fearing • Fats and steroids • Fats functions – Store twice as much energy as carbs – Protection, Cushion and insulate internal organs – Fats are stored in adipose cells – Examples include waxes, oils, fats and steroids Fig. 3.14 Saturated versus Unsaturated fats • Saturated fats – No double bonds between carbons – All possible Hydrogens attached to carbons – Solid at room temperature commonly produced by animals – Examples lard, butter, bacon grease – Linked to cardiovascular disease Unsaturated Fats • • • • • Have carbon=carbon double bonds In place of attached Hydrogens Liquid at room temperature Commonly produced by plants Examples are vegetable, corn and olive oils Phospholipids • Two regions with opposite properties • Forms plasma membrane • Phosphate ‘head’ is polar – Hydrophillic water loving – Phosphate group faces out – Towards Watery environment inside and outside cell • Fatty acid tails are non-polar – Hydrophobic - water fearing – Tails face each other – Forms a barrier Fig. 5-14 Hydrophilic head Hydrophobic tail WATER WATER Steroids • Lipids because they are hydrophobic • Carbon chains form 4 fused rings • Cholesterol – Component of cell membranes – Forms other steroids from it – Make into sex hormones • Estrogen • Testosterone Fig. 3.15 Anabolic steroids • • • • Mimic testosterone First used for anemia / muscle disease Abused by athletes Misuse can cause – Facial bloating/acne – Violent mood swings – Liver damage – Increase cholesterol levels – Reduce sex drive and fertility Proteins have many structures, resulting in a wide range of functions • Proteins are polymers made of amino acid monomers • Amino acid general structure- central carbon is bonded to a a carboxyl group, amine group, a Hydrogen and an R group which varies. • Peptide bonds link amino acids by dehydration synthesis Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings Fig. 5-UN1 General structure of an amino acid Amino group carbon Carboxyl group Proteins • Amino acids linked by peptide bonds • The function of a protein depends on the order and number of amino acids. Polypeptide (protein) formation Primary structure – Unique sequence of amino acids – There are 20 different amino acids – Change in order can cause disease • Sickle cell anemia • One amino acid changed Fig. 5-21a Primary Structure 1 +H 5 3N Amino end 10 Amino acid subunits 15 20 25 Secondary Structure • Secondary structure, found in most proteins refers to one of two threedimensional shapes as a result of Hydrogen bonding • Alpha helix is a coiled shape • Beta pleated sheet is an accordion shape Fig. 5-21c Secondary Structure pleated sheet Examples of amino acid subunits helix Tertiary Structure • Tertiary structure results in a complex globular shape due to interactions between R groups, • Interactions include hydrogen bonds, ionic bonds, hydrophobic interactions, and van der Waals interactions • Strong covalent bonds called disulfide bridges may reinforce the protein’s structure Animation: Tertiary Protein Structure Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings Fig. 5-21e Tertiary Structure Quaternary Structure Quaternary Structure • Quaternary structure – Interaction between two or more polypeptide chains linked together to form one large protein. – Example: hemoglobin is a globular protein with quaternary structure composed of four chains – Single amino acid substitution causes sickle cell anemia Fig. 5-21g Polypeptide chain Chains Iron Heme Chains Hemoglobin Collagen Fig. 3.21 Fig. 5-22c 10 µm Normal red blood cells are full of individual hemoglobin molecules, each carrying oxygen. 10 µm Fibers of abnormal hemoglobin deform red blood cell into sickle shape. How is structure determined? • Order of amino acids specified by a gene recipe for a polypeptide • Proteins include – Structural – Storage – Contractile – Transport – Defensive – Signal proteins – ENZYMES! ADD Chaperonins • Chaperonins are protein molecules that assist in the proper folding of proteins within cells. • It provides protection against other particles in the cytoplasm while the protein folds. ADD Denaturation of Proteins • The function of a protein is determined by the sequence and spontaneous folding of the polypeptide chain. • Certain physical and chemical conditions – pH, salt concentration, temperature • Denaturation occurs when a protein unfolds, loses its shape and ability to function properly. • Can you think of a way we denature proteins? • The white (albumen) turns opaque during cooking because denatured proteins solidify. • This is why high fevers can be fatal • Proteins in the blood can become denatured from high body temperatures Table 5-1 Fig. 5-21d Abdominal glands of the spider secrete silk fibers made of a structural protein containing pleated sheets. The radiating strands, made of dry silk fibers, maintain the shape of the web. The spiral strands (capture strands) are elastic, stretching in response to wind, rain, and the touch of insects. Nucleic acids • • • • DeoxyriboNucleic Acid - DNA DNA is a recipe book for proteins Genes direct the order of amino acids Two types of nucleic acids – DNA – RNA - RiboNucleic Acid • Chemical code – Nucleic acid to protein language – RNA helps with this process Fig. 5-26-2 DNA 1 Synthesis of mRNA in the nucleus mRNA NUCLEUS CYTOPLASM mRNA 2 Movement of mRNA into cytoplasm via nuclear pore Nucleic Acids: DNA and RNA • DNA and RNA are polynucleotides made up of monomers called nucleotides • Each Nucleotide has three parts: – Nitrogenous base (adenine, thymine, guanine, cytosine and uracil (RNA only) – Pentose - five carbon sugar, deoxyribose in DNA and ribose in RNA – Phosphate group Fig. 5-27ab 5' end 5'C 3'C Nucleoside Nitrogenous base 5'C Phosphate group 5'C 3'C (b) Nucleotide 3' end (a) Polynucleotide, or nucleic acid 3'C Sugar (pentose) Nitrogenous bases • Pyrimidines-six membered ring of carbon and nitrogen – Cytosine C (DNA &RNA) – Thymine T (DNA) – Uracil U (RNA) • Purines-six membered ring fused to a five membered ring of carbon and nitrogen – Adenine A (DNA & RNA) – Guanine G (DNA & RNA) Fig. 3.26 Sugar –phosphate backbone The backbone • Adjacent nucleotides are connected by phosphodiester linkages between the OH group on the sugar and phosphate group of the next nucleotide Double helix strands run anti-parallell The Roles of nucleic acids DNA RNA • DNA is the genetic material organisms inherit from their parents • Each chromosome contains one long DNA molecule containing from several hundred to more than a thousand genes. • DNA programs all the cells activities by producing proteins as needed • DNA directs the synthesis of mRNA which then directs the production of amino acids Which of the following molecules would contain a polar covalent bond? A. Cl2 B. NaCl C. H2O D. CH4 E. C6H12O6